-
痛风是长期血尿酸增高、尿酸盐晶体广泛沉积所引起的炎症性疾病。高尿酸血症是痛风发生的重要生化基础。痛风患者的早期临床表现不典型,多为无症状性高尿酸血症,随着关节及软组织中尿酸盐的沉积增多,形成痛风性关节炎,继而尿酸盐结晶析出形成痛风石,晚期可造成关节破坏及功能障碍等严重后果[1-2]。第一跖趾关节是痛风最常见的且具有特征性的好发部位,因此笔者选择此部位进行研究。影像学检查作为痛风诊断的重要手段,方法众多,但是每种检查方法都有其自身的局限性,特别是对早期痛风性关节炎的诊断存在一定的局限性[3]。双能量CT是近年来新出现的一种检测痛风患者尿酸盐结晶的有效的新方法,其灵敏度和特异度都很高[4-5]。宝石能谱CT的物质分离技术可以把任何一种物质解离成另外两种物质的组合,即基物质对,如尿酸-钙、水-碘等,这种技术在一定程度上实现了定性分离及定量分析[6-7]。笔者通过定量分析高尿酸血症患者足部第一跖趾关节及关节旁软组织的基物质浓度,以期了解高尿酸血症患者不同影像学表现的尿酸沉积情况,并通过与血尿酸的相关性研究,探讨能谱CT对高尿酸血症患者尿酸盐沉积的定量分析价值。
-
收集2016年10月至2017年6月在广州医科大学附属惠州医院就诊的高尿酸血症患者84例,入组需同时满足以下条件:①血尿酸值超过正常值至少1年,且近1个月内检查仍超标;②近3个月内没有接受标准量降尿酸药物治疗。排除条件:类风湿关节炎、脊柱关节炎、骨关节炎等非痛风关节炎患者,入组时血尿酸浓度正常且无高尿酸血症史。所有患者检查前均签署了知情同意书。
本研究患者经临床诊断可分为无症状高尿酸血症24例、疑似急性痛风性关节炎发作35例、慢性痛风25例。根据痛风不同时期的影像学表现,将患者进行再分组:影像学正常组36例(A组)、单纯骨质破坏组23例(B组)、骨质破坏+尿酸盐结晶形成组25例(C组)。本研究患者中未发现单纯尿酸盐结晶形成者。
-
采用美国GE公司Discovery CT750 HD能谱CT机进行扫描。扫描参数:GSI-Helical模式,电压80 kVp/140 kVp,螺比0.984:1,探测器准直64×0.625 mm,球管转速0.8 s/转,显示视野25 ~ 35 cm,扫描和间隔层厚0.625 mm,重建层厚1.25 mm。扫描部位:双侧足部第一跖趾关节区,双侧尽量对称。
-
所有数据分析及图像处理在美国GE公司AW4.6工作站上使用GSI Viewer软件进行处理,应用能谱CT的物质分离功能,同时获得尿酸(钙)基图和钙(尿酸)基图。在尿酸(钙)基图中,调节窗宽、窗位,窗位选择1170~1200 mg/cm3,窗宽350~400。寻找关节周围高密度的尿酸盐沉积区。选择双足第一跖趾关节内侧软组织(距离邻近骨皮质>2 mm)、第一跖趾关节面相邻皮质骨及松质骨进行测量。ROI选取高密度区内部,面积为6~10 mm3,测量选择最高峰值数据。由2名高年资主治医师分别对每个部位测量3次,取其平均值。选择尿酸与钙作为基物质对,测量配对基物质浓度。
能谱CT诊断尿酸盐沉积的参考标准:尿酸盐结晶在尿酸(钙)基图上表现为高密度,而在钙(尿酸)基图上表现为低密度。尿酸盐的尿酸(钙)浓度范围为1200~1500 mg/cm3。
-
采用SPSS 23.0软件进行统计学分析,数据以x±s表示。3组患者之间的年龄、血尿酸值及基物质浓度比较采用单因素方差分析。组内两两比较,方差齐者采用LSD法,方差不齐者采用Tamhane's T2检验。对所有高尿酸血症患者的血尿酸值与其第一跖趾关节骨皮质、骨松质及关节旁软组织尿酸(钙)浓度的相关性分别进行线性回归分析。P<0.05表示差异有统计学意义。
-
3组患者的年龄分别为A组(39.1±11.1)岁、B组(48.5±13.9)岁、C组(43.2±12.5)岁,B组最大,A组最小,3组间差异有统计学意义(LSD法,P=0.019);血尿酸值分别为A组(489.5±90.6)μmol/L,B组(494.7±77.2)μmol/L、C组(581.3±69.2)μmol/L,C组最高,A组最低,3组间差异有统计学意义(LSD法,P<0.001)(表 1)。
组别 例数 年龄/岁 血尿酸值/(μmol/L) 尿酸(钙)基物质浓度(mg/cm3) 关节旁软组织 骨松质 骨皮质 影像学正常组(A组) 36 39.1±11.1 489.5±90.6 1169.6±7.9 1134.3±13.3 1324.8±31.4 单纯骨质破坏组(B组) 23 48.5±13.9 494.7±77.2 1193.1±16.3 1145.8±23.3 1335.6±37.3 骨质破坏+尿酸盐结晶形成组(C 组) 25 43.2±12.5 581.3±69.2 1308.2±85.9 1145.8±30.9 1320.4±43.5 F值 4.139 9.49 66.073 1.076 2.685 χ2值 - - 61.698 3.464 - P值 0.019 0 0 0.177 0.346 注:表中,"_"原表示此项采用LSD法检验,无χ2值。 表 1 3组高尿酸血症患者的年龄、血尿酸值、尿酸(钙)基物质浓度的比较(x)
Table 1. Comparison of the levels of uric acid(calcium) in cortical bone, cancellous bone, and periarticular soft tissue of the first metatarsophalangeal joint in 3 groups(x)
-
3组患者第一跖趾关节关节旁软组织尿酸(钙)浓度分别为A组(1169.6±7.9)mg/cm3、B组(1193.1±16.3)mg/cm3、C组(1308.2±85.9)mg/cm3,C组最高,A组最低,3组间差异有统计学意义(χ2=61.698,P<0.001)。骨皮质尿酸(钙)浓度分别为A组(1324.8±31.4)mg/cm3、B组(1335.6±37.3)mg/cm3、C组(1320.4±43.5)mg/cm3。骨松质尿酸(钙)浓度分别为A组(1134.3±13.3)mg/cm3、B组(1145.8±23.3)mg/cm3、C组(1145.8±30.9)mg/cm3。3组间骨皮质、骨松质尿酸(钙)浓度差异均无统计学意义(骨皮质:LSD法,P=0.346;骨松质:χ2=3.464,P=0.177)(表 1)。
-
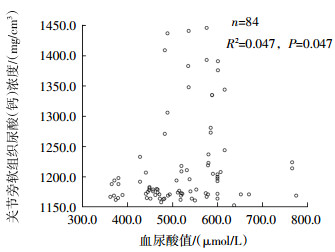
3组患者尿酸(钙)浓度与血尿酸值的相关性分析结果见表 2。结果显示,第一跖趾关节关节旁软组织的尿酸(钙)浓度与血尿酸值有关联(图 1),显著性P值=0.047(<0.05),表示两者存在线性关系;B值=0.187(>0),表示两者呈线性正相关,即血尿酸值越高,关节旁软组织的尿酸(钙)浓度越高;决定系数R2为0.047,表示血尿酸值可解释关节旁软组织尿酸(钙)浓度变异性的4.7%,另外95.3%的变异性不能用血尿酸值来解释。第一跖趾关节骨皮质、骨松质的尿酸(钙)浓度与血尿酸值均没有关联,显著性P值分别为0.408、0.810(均>0.05)。

图 1 84例高尿酸血症患者第一跖趾关节关节旁软组织尿酸(钙)浓度与血尿酸值的相关性散点图
Figure 1. Correlation between uric acid (calcium) concentration in the first metatarsophalangeal joint and serum uric acid in 84 patients with hyperuricemia
因变量 R2 调整后R2 B值 T值 显著性(P值) 关节旁软组织的尿酸钙浓度 0.047 0.036 0.187 2.018 0.047 骨皮质的尿酸钙浓度 0.008 -0.004 0.038 0.832 0.408 骨松质的尿酸钙浓度 0.001 -0.011 -0.007 -0.241 0.81 表 2 84例高尿酸血症患者的血尿酸值与其第一跖趾关节骨皮质、骨松质及关节旁软组织尿酸(钙)浓度的线性回归分析结果
Table 2. Correlation of serum uric acid with uric acid (calcium) concentration in cortical bone, cancellous bone and periarticular soft tissue of the first metatarsophalangeal joint in 84 hyperuricemia patients
-
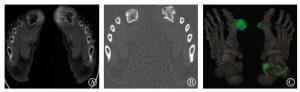
利用尿酸(钙)基图可以明确显示出更多、更小的尿酸盐结晶(图 2中A)。钙(尿酸)基图可以清晰地显示骨质破坏情况,包括破坏骨边缘形成的硬化带(图 2中B)。在尿酸(钙)基图的基础上,使用伪彩色对高尿酸浓度区进行标注可以更加直观地显示出尿酸结晶沉积的分布(图 2中C)。
-
近年来,随着生活水平的提高及饮食习惯的改变,痛风、无症状高尿酸血症的发病率明显上升,且有明显年轻化的趋势[8]。目前确诊痛风的“金标准”是通过关节穿刺找到特征性的尿酸钠结晶,但该检查为有创性检查,操作难度大,绝大部分患者不愿意接受。因此,通过影像学方法进行无创性的早期关节内尿酸盐沉积的检测日益成为目前临床工作的关注点。目前常用的影像学检查(X射线、超声、常规CT和MRI)在痛风诊断的应用中各有其优缺点,特异度和灵敏度都不够高[3]。双能量CT是近年来出现的检测尿酸盐沉积的一种新方法,其灵敏度及特异度都很高。宝石能谱CT通过高低电压(80 kVp/140 kVp)的瞬时(0.5 ms)切换获得两组吸收投影数据,转换成物质密度投影数据,经过重建求解出两种基物质的密度的空间分布,这样就将组织的CT值转化为基物质的密度值,实现了物质的分离和定量分析[6-7]。初步研究结果表明双能量CT对痛风的诊断有重要意义[7, 9-14]。
本研究中影像学正常的A组患者的平均年龄为(39.1±11.1)岁,影像学异常的B组和C组患者的平均年龄分别为(48.5±13.9)岁和(43.2±12.5)岁,其中经临床确诊的最年轻痛风患者仅20岁,发病年龄明显年轻化,与文献中报道的结果一致[8]。B组和C组的发病年龄均大于A组,笔者考虑是因为随着年龄的增长,人体的代谢率下降,40岁以后的高尿酸血症患者出现影像学异常的风险更大。在血尿酸值的对比中,C组明显高于B组和A组,B组和A组的平均值接近,笔者认为是因为C组患者经临床确诊均为慢性痛风患者,并已有明显尿酸结晶形成,虽然部分患者接受了治疗,但血尿酸值仍然维持在高水平,而B组和A组患者均未发现尿酸结晶形成,血尿酸值相对C组较低。
基物质图定量分析能够区分骨皮质、尿酸盐沉积灶、肌肉、皮下组织中尿酸含量的差别,从而能在尿酸基图上显示尿酸盐沉积区,并定量测量其基物质浓度。能谱CT基物质图中尿酸(钙)浓度并非真正的尿酸浓度,而是用尿酸的密度值来表示。尿酸盐结晶沉积的越多,反应带的尿酸(钙)浓度越高,密度也越大。所以尿酸盐结晶的密度很高。若无尿酸盐沉积,则表现为软组织正常的密度。笔者选择尿酸与钙配对基物质图像,是因为尿酸盐的主要成分是尿酸,钙是人体骨骼的主要成分,是人体中含量最多的物质之一[15],而尿酸盐沉积的关节的主要构成组织为尿酸结晶与邻近关节组成骨。本研究中,关节旁软组织尿酸结晶在尿酸基图上可以被明确地显示出来,这对利用尿酸基图寻找外周关节旁可能存在的尿酸结晶沉积提供了帮助。同时,在基物质图的基础上可根据尿酸浓度的差异使用伪彩色来对高尿酸浓度的尿酸结晶进行标记,从而更加清晰地显示尿酸结晶的有无、大小、部位以及分布状况。钙基图上可以很好地显示骨质破坏边缘的情况。沉积在关节旁软组织内的尿酸结晶具有明显的骨质破坏特性,是痛风性关节炎引起骨质破坏发展的一个决定因素[16]。因此,在使用尿酸基图检查尿酸盐沉积的同时,可以在钙基图上清晰显示出尿酸盐沉积邻近关节骨质的侵蚀破坏情况。
需要注意的是,尽管本研究中3组患者的第一跖趾关节关节旁软组织的尿酸(钙)浓度之间的差异有统计学意义,但骨皮质、骨松质的尿酸(钙)浓度之间的差异均无统计学意义。我们推测在尿酸盐沉积发生发展的过程中,可能最先沉积于关节旁的软组织中。因此,可以通过监测关节旁软组织的尿酸(钙)浓度的变化从而较灵敏地反映高尿酸血症患者关节周围的尿酸水平。
单纯骨质破坏组中部分是无症状的高尿酸血症患者,在尿酸(钙)基图中并没有找到明确的尿酸盐沉积高密度区,也没有发现骨质破坏旁软组织增厚或肿胀,考虑可能是其他疾病所致。
血尿酸值在人体组织中的实际影响一直是临床工作关注的重点,我们尝试运用线性回归方法对3组中所有高尿酸血症患者的血尿酸值与其第一跖趾关节骨皮质、骨松质及关节旁软组织的尿酸(钙)浓度的相关性作分析比较,这在国内同类研究文献中未见报道。本研究结果显示所有高尿酸血症患者的血尿酸值与其第一跖趾关节关节旁软组织的尿酸(钙)浓度呈线性正相关,即血尿酸值越高,患者第一跖趾关节关节旁软组织的尿酸(钙)浓度越高,但决定系数R2仅为0.047,即关节旁软组织的尿酸(钙)浓度变异性的4.7%是由血尿酸值引起的,另外95.3%的变异性并不是由血尿酸值引起。另外,第一跖趾关节骨皮质、骨松质的尿酸(钙)浓度与血尿酸值之间均无相关性。我们认为这个结论能进一步印证前文提到的推测,即第一跖趾关节关节旁软组织的尿酸(钙)浓度的变化能较灵敏地反映高尿酸血症患者关节周围的尿酸水平,可作为监测的重点部位。
高尿酸血症患者在控制饮食以及服用降尿酸药物后血尿酸值可以保持在正常范围,如果临床症状不典型,在不借助于其他检查的情况下,就很容易引起误诊。本研究中部分无症状高尿酸血症患者在影像学检查中已出现骨质破坏的情况,同时尿酸(钙)浓度也较高,此类患者应该引起临床的重视,以免病情恶化。而能谱CT定量测量的基物质浓度具备一定时间段上的稳定性,对痛风的诊疗比血尿酸值具备更大的临床参考价值。
本研究存在的不足包括没有将血尿酸值正常组作为对照组,研究的部位较为局限,研究组样本量不够大,血尿酸值与影像学检查所得第一跖趾关节关节旁软组织尿酸(钙)浓度的相关程度较低,并没有与尿酸-羟基磷灰石基物质对作对比分析,这些不足仍有待在今后的研究中进一步改进。如能应用能谱CT对慢性痛风患者治疗前后的尿酸结晶沉积情况进行定期监测,将会对临床治疗方案提供更可靠的监测手段[17]。
综上所述,能谱CT不仅可以定性地显示出更多、更小的尿酸结晶,还可以对患者的尿酸(钙)基物质浓度进行量化比较,通过对第一跖趾关节关节旁软组织尿酸(钙)浓度的监测,结合血尿酸值能有效地评价患者的尿酸代谢水平,对临床痛风的早期诊断及定期治疗疗效监测有重要价值。
能谱CT对高尿酸血症患者尿酸盐沉积的定量分析价值
Quantitative analysis value of uric acid deposition in patients with hyperuricemia by Gemstone spectal CT
-
摘要:
目的探讨能谱CT对高尿酸血症患者第一跖趾关节及关节旁软组织尿酸盐沉积的定量分析价值。 方法选取2016年10月至2017年6月就诊并经临床证实的高尿酸血症患者84例。根据影像学表现分为影像学正常患者36例(A组)、单纯骨质破坏患者23例(B组)、骨质破坏+尿酸盐结晶形成患者25例(C组)。采用宝石能谱CT对A、B、C 3组患者的足部行宝石能谱CT扫描,分别测量3组患者第一跖趾关节骨皮质、骨松质及关节旁软组织的尿酸(钙)的基物质浓度。3组患者的年龄、血尿酸值和基物质浓度的比较,采用单因素方差分析。组内两两比较,方差齐者采用LSD法,方差不齐性采用Tamhane's T2检验。运用线性回归方法分析所有高尿酸血症患者的血尿酸值与其第一跖趾关节骨皮质、骨松质及关节旁软组织尿酸(钙)浓度的相关性。 结果3组患者的年龄[A组:(39.1±11.1)岁,B组:(48.5±13.9)岁,C组:(43.2±12.5)岁](LSD法,P=0.019)、血尿酸值[A组:(489.5±90.6)μmol/L,B组:(494.7±77.2)μmol/L,C组:(581.3±69.2)μmol/L](LSD法,P < 0.001)及第一跖趾关节关节旁软组织尿酸(钙)浓度[A组:(1169.58±7.88)mg/cm3,B组:(1193.13±16.35)mg/cm3,C组:(1308.20±85.89)mg/cm3](χ2=61.698,P < 0.001)之间的差异均有统计学意义。第一跖趾关节骨皮质[A组:(1324.8±31.4)mg/cm3,B组:(1335.6±37.3)mg/cm3,C组:(1320.4±43.5)mg/cm3](LSD法,P=0.346)、骨松质尿酸(钙)浓度[A组:(1134.3±13.3)mg/cm3,B组:(1145.8±23.3)mg/cm3,C组:(1145.8±30.9)mg/cm3](χ2=3.464,P=0.177)之间的差异均无统计学意义。所有高尿酸血症患者的血尿酸值与第一跖趾关节关节旁软组织尿酸(钙)浓度呈线性正相关,与第一跖趾关节骨皮质、骨松质的尿酸(钙)浓度没有相关性。 结论能谱CT基物质图可以定量测量关节内外基物质浓度,明确尿酸盐沉积情况,为临床判断病情提供依据。 Abstract:ObjectiveTo evaluate the quantitative analysis value of gemstone spectral CT for diagnosing urate deposition in the first metatarsophalangeal joint and periarticular soft tissue of patients with hyperuricemia. MethodsThis work involved 84 patients with hyperuricemia who underwent foot gemstone spectral CT from October 2016 to June 2017. The patients were divided into three groups:36 patients (group A) with normal imaging, 23 patients(group B) with bone destruction, and 25 patients(group C) with bone destruction and uric acid crystallization. The basal concentrations of uric acid(calcium) in the first metatarsophalangeal joint cortical bone, cancellous bone and periarticular soft tissue were measured using gemstone spectral CT scanning. The age, serum uric acid value, and basal concentration of uric acid(calcium) were compared by univariate ANOVA among the three groups. In intra-group comparison, LSD method was used for the data with equal variance, and Tamhane's T2 test was used for the data with unequal variance.Correlation of serum uric acid value with the uric acid(calcium) level of the first metatarsophalangeal joint cortical bone, cancellous bone, and periarticular soft tissue in all hyperuricemia patients was analyzed by linear regression. ResultsSignificant differences in age, serum uric acid value, and uric acid (calcium) concentration were found for periarticular soft tissues of the first metatarsophalangeal joints(P < 0.05). No differences were observed between the uric acid(calcium) concentration in the first metatarsophalangeal joint cortical bone and cancellous bone. The following values were obtained for age:group A(39.1±11.1) years old, group B (48.5±13.9) years old, and group C (43.2±12.5) years old (LSD, P=0.019); for serum uric acid value:group A(489.5±90.6) μmol/L, group B(494.7±77.2) μmol/L, and group C(581.3±69.2) μmol/L(LSD, P < 0.001); for the uric acid (calcium) concentration in the periarticular soft tissues of the first metatarsophalangeal joints:group A (1169.58±7.88) mg/cm3, group B (1193.13±16.35) mg/cm3, and group C (1308.20±85.89) mg/cm3(χ2=61.698, P < 0.001); for the uric acid(calcium) concentration in the first metatarsophalangeal joints:group A (1324.8±31.4) mg/cm3, group B (1335.6±37.3) mg/cm3, and group C (1320.4±43.5) (LSD, P=0.346); for the uric acid(calcium) concentration in the cancellous bone of the first metatarsophalangeal joints:group A (1134.3±13.3) mg/cm3, group B (1145.8±23.3) mg/cm3, and group C (1145.8±30.9) mg/cm3 (χ2=3.464, P=0.177). The serum uric acid value was linearly positively correlated with the uric acid(calcium) concentration in periarticular soft tissues of the first metatarsophalangeal joints, but not with the uric acid(calcium) concentration in cortical bone and cancellous bone of the first metatarsophalangeal joints. ConclusionGemstone spectral CT can measure quantitatively the internal and external base material concentrations of joints and define the crystallization of uric acid salt, which can provide a basis for clinical judgment. -
Key words:
- Hyperuricemia /
- Uric acid salt deposition /
- Gemstone spectral CT /
- Quantitative analysis
-
表 1 3组高尿酸血症患者的年龄、血尿酸值、尿酸(钙)基物质浓度的比较(x)
Table 1. Comparison of the levels of uric acid(calcium) in cortical bone, cancellous bone, and periarticular soft tissue of the first metatarsophalangeal joint in 3 groups(x)
组别 例数 年龄/岁 血尿酸值/(μmol/L) 尿酸(钙)基物质浓度(mg/cm3) 关节旁软组织 骨松质 骨皮质 影像学正常组(A组) 36 39.1±11.1 489.5±90.6 1169.6±7.9 1134.3±13.3 1324.8±31.4 单纯骨质破坏组(B组) 23 48.5±13.9 494.7±77.2 1193.1±16.3 1145.8±23.3 1335.6±37.3 骨质破坏+尿酸盐结晶形成组(C 组) 25 43.2±12.5 581.3±69.2 1308.2±85.9 1145.8±30.9 1320.4±43.5 F值 4.139 9.49 66.073 1.076 2.685 χ2值 - - 61.698 3.464 - P值 0.019 0 0 0.177 0.346 注:表中,"_"原表示此项采用LSD法检验,无χ2值。 表 2 84例高尿酸血症患者的血尿酸值与其第一跖趾关节骨皮质、骨松质及关节旁软组织尿酸(钙)浓度的线性回归分析结果
Table 2. Correlation of serum uric acid with uric acid (calcium) concentration in cortical bone, cancellous bone and periarticular soft tissue of the first metatarsophalangeal joint in 84 hyperuricemia patients
因变量 R2 调整后R2 B值 T值 显著性(P值) 关节旁软组织的尿酸钙浓度 0.047 0.036 0.187 2.018 0.047 骨皮质的尿酸钙浓度 0.008 -0.004 0.038 0.832 0.408 骨松质的尿酸钙浓度 0.001 -0.011 -0.007 -0.241 0.81 -
[1] Roddy E, Zhang W, Doherty M. The changing epidemiology of gout[J]. Nat Clin Pract Rheumatol, 2007, 3(8):443-449. DOI:10.1038/ncprheum0556. [2] 中华医学会风湿病学分会.原发性痛风诊断和治疗指南[J].中华风湿病学杂志, 2011, 15(6):410-411. DOI:10.3760/cma.j.issn.1007-7480.2011.06.013.
Chinese Rheumatology Association. Guidelines for the diagnosis and treatment of primary gout[J]. Chin J Rheumatol, 2011, 15(6):410-413. DOI:10.3760/cma.j.issn.1007-7480.2011.06.013.[3] 王旭, 刘斌.痛风性关节炎的影像学诊断及进展[J].国际医学放射学杂志, 2012, 35(3):251-254. DOI:10.3784/j.issn.1674-1897. 2012.03.Z0309.
Wang X, Liu B. Imaging diagnosis and progress of gouty arthritis[J]. Int J Med Radiol, 2012, 35(3):251-254. DOI:10.3784/j.issn.1674-1897. 2012.03.Z0309.[4] Bongartz T, Glazebrook KN, Kavros SJ, et al. Dual-energy CT for the diagnosis of gout:an accuracy and diagnostic yield study[J]. Ann Rheum Dis, 2015, 74(6):1072-1077. DOI:10.1136/annrheumdis-2013-205095. [5] Finkenstaedt T, Manoliou A, Toniolo M, et al. Gouty arthritis:the diagnostic and therapeutic impact of dual-energy CT[J]. Eur Radiol, 2016, 26(11):3989-3999. DOI:10.1007/s00330-016-4237-2. [6] 李小虎, 刘斌, 余永强, 等.能谱CT的原理与临床应用价值[J].中国医疗器械信息, 2011, 17(10):1-5. DOI:10.3969/j.issn.1006-6586. 2011.10.001.
Li XH, Liu B, Yu YQ, et al. The Principle and Clinical Value of Spectral CT[J]. China Med Dev Info, 2011, 17(10):1-5. DOI:10.3969/j.issn.1006-6586. 2011.10.001.[7] Li X, Wang X, Yu Y, et al. Detection of uric acid depositing in tophaceous gout using a new dual energy spectral CT technology[J]. J Xray Sci Technol, 2014, 22(4):541-549. DOI:10.3233/XST-140444. [8] 张琳, 闫雅更, 董凤丽, 等.饮食饮酒及肥胖对无症状高尿酸血症发生痛风的影响[J].军医进修学院学报, 2012, 33(2):135-136, 147. DOI:10.3969/j.issn.1672-4860.2014.01.009.
Zhang L, Yan YG, Dong FL, et al. Effects of diet, drinking and obesity on development of gout in patients with asymptomatic hyperuricemia[J]. J Chinese PLA Postgrad Med Sch, 2012, 33(2):135-136, 147. DOI:10.3969/j.issn.1672-4860.2014.01.009.[9] 刘炜, 薛华丹, 曾学军, 等.双能量CT检测痛风患者尿酸盐沉积的初步应用[J].中国医学科学院学报, 2010, 32(6):645-648. DOI:10.3881/j.issn.1000-503X.2010.06.011.
Liu W, Xue HD, Zeng XJ, et al. Application of Dual-energy Computed Tomography for Detecting Uric Acid Deposition in Patients with Gout[J]. Acta Acad Med Sin, 2010, 32(6):645-648. DOI:10.3881/j.issn.1000-503X.2010.06.011.[10] 盛雪霞, 曹志宏, 闵志刚, 等.双源CT诊断痛风性关节炎准确性的Meta分析[J].实用放射学杂志, 2015, 31(6):974-977. DOI:10.3969/j.issn.1002-1671.2015.06.023.
Sheng XX, Cao ZH, Min ZG, et al. Accuracy of dual-energy CT in diagnosing gouty arthritis:a Meta-analysis[J]. J Pract Radiol, 2015, 31(6):974-977. DOI:10.3969/j.issn.1002-1671.2015.06.023.[11] 任洁, 周毅, 吴会霞, 等.双能CT检查在痛风性关节炎中的应用价值[J].南方医科大学学报, 2015, 35(3):384-386. DOI:10.3969/j.issn.1673-4254.2015.03.14.
Ren J, Zhou Y, Wu HX, et al. Value of dual-energy computed tomography in the diagnosis of gouty arthritis[J]. J South Med Univ, 2015, 35(3):384-386. DOI:10.3969/j.issn.1673-4254. 2015.03.14.[12] 窦小锋, 张波, 田为中.双源CT双能量成像在痛风患者尿酸盐沉积诊断中的应用价值[J].现代医用影像学, 2015, 24(4):550-552.
Dou XF, Zhang B, Tian WZ. Value of Dual-energy Computed Tomography in the Diagnosis of Patients with Gout[J]. M M I Bimonthly, 2015, 24(4):550-552.[13] 张娅梅, 陈谦, 殷信道, 等.双源CT双能量成像在急性期和非急性期痛风患者尿酸盐沉积诊断中的对比研究[J].中国医学计算机成像杂志, 2016, 22(6):537-541. DOI:10.3969/j.issn.1006-5741.2016.06.009.
Zhang YM, Chen Q, Yin XD, et al. Comparative Study of Urate Deposition in Acute and Non-acute Gout Patients by Dual-energy Computed Tomography[J]. Chin Comput Med Imag, 2016, 22(6):537-541. DOI:10.3969/j.issn.1006-5741.2016.06.009.[14] 郑玲, 周长圣, 张龙江, 等.双源双能量CT检测痛风石的初步经验[J].中国临床医学影像杂志, 2011, 22(2):105-107. DOI:10.3969/j.issn.1008-1062.2011.02.009.
Zheng L, Zhou CS, Zhang LJ, et al. Preliminary experience of dual source, dual energy CT in detection of gout[J]. J Chin Clin Med Imaging, 2011, 22(2):105-107. DOI:10.3969/j.issn.1008-1062. 2011. 02.009.[15] 李小虎, 余永强, 王万勤, 等. CT能谱成像对肾结石成分分析的初步研究[J].中华放射学杂志, 2011, 45(12):1216-1219. DOI:10.3760/cma.j.issn.1005-1201.2011.12.034.
Li XH, Yu YQ, Wang WQ, et al. Spectral CT imaging in the evaluation of composition of kidney stones[J]. Chin J Radiol, 2011, 45(12):1216-1219. DOI:10.3760/cma.j.issn.1005-1201.2011.12. 034.[16] Dalbeth N, Clark B, Gregory K, et al. Mechanisms of bone erosion in gout:a quantitative analysis using plain radiography and computed tomography[J]. Ann Rheum Dis, 2009, 68(8):1290-1295. DOI:10.1136/ard.2008.094201. [17] Manger B, Lell M, Wacker J, el al. Detection of periarticular urate deposits with dual energy CT in patients with acute gouty arthritis[J]. Ann Rheum Dis, 2012, 71(3):470-472. DOI:10.1136/ard.2011. 154054. -





 下载:
下载: