-
结直肠癌是严重威胁人类生命健康的常见消化道恶性肿瘤[1]。我国结肠癌发病率呈逐年上升趋势,发病年龄逐渐年轻化,目前位居我国恶性肿瘤死因的第4位。手术治疗是有效地治疗结肠癌的方法,同时采取联合放疗、化疗等方法提升治疗效果,提高患者的生存率。但是,由于结肠癌具有对辐射较强的抗性,严重影响了放疗效果。因此,有效增加结肠癌的辐射敏感性,减少对正常组织的放射损伤,从而提高结肠癌患者的放疗效果和治愈率尤为重要。
褪黑素是由哺乳动物和人类大脑组织中的松果体合成与分泌的一种吲哚类物质,于1958年首次被发现[2]。褪黑素参与机体许多重要的生理进程,包括控制生物节律、抗氧化、抗炎、抗衰老、调节免疫和内分泌的功能[3-4]。大量文献报道,褪黑素对不同肿瘤具有抗瘤的活性,例如乳腺癌[5]、肺癌[6]、食管癌[7]、胰腺癌[8]以及前列腺癌[9]。褪黑素对不同的结肠癌类型也有抗瘤特性[10-11]。有研究结果表明,褪黑素有可能作为新型辐射增敏剂应用到临床[12-13]。但是,褪黑素联合放疗应用于结肠癌治疗的研究并不多,在放疗前使用褪黑素对结肠癌会产生什么效应并不明了。本研究通过观察褪黑素联合γ射线照射对体外结肠癌HCT 116细胞生长的影响,以及对荷瘤裸鼠的抑瘤效应,阐明褪黑素对结肠癌细胞辐射敏感性的影响,为以后临床上提高结肠癌的放疗效果提供实验依据。
-
137Cs γ射线照射源(加拿大原子能公司);流式细胞仪和电泳仪(美国Bio-Rad公司)。IMDM培养液(美国HyClone公司);褪黑素(美国Sigma公司);碘化丙啶(propidium iodide, PI)和磷脂结合蛋白V-异硫氰酸荧光素(美国Beckman公司);低熔点琼脂糖(美国Bio-Rad公司)。
-
人结肠癌HCT 116细胞(由本实验室保存)用含10%胎牛血清的IMDM培养液在5% CO2的37℃恒温培养箱中培养,待细胞生长浓度约为80%时进行传代培养。选择处于对数生长期的人结肠癌HCT 116细胞进行实验。
-
雄性BALB/c-nu裸鼠24只,6~8周龄,体重(22±1)g,由北京维通利华实验动物技术有限公司提供。所有动物于无特定病原体条件下,于恒温(23℃±2℃)、恒湿(45%~50%)、无菌净化屏障系统内,饲养于中国医学科学院放射医学研究所实验动物中心,实验动物使用许可证编号为SYXK(津)2014-0004。动物实验方案由中国医学科学院放射医学研究所实验动物伦理福利委员会审核(批号DWLL-20180112)。
-
将人结肠癌HCT 116细胞分为4组,即:空白对照组、褪黑素组、照射组和褪黑素+照射组。空白对照组:不给予任何处理;褪黑素组:褪黑素给药浓度为1 mmol/L,给药时间为2 h;照射组:利用137Cs γ射线照射源进行6 Gy照射;褪黑素+照射组:在照射前2 h给予褪黑素,给药浓度为1 mmol/L,给药2 h后进行6 Gy照射。照射时间为6 min 20 s, 剂量率为0.95 Gy/min。
-
体外克隆形成实验照射剂量为0、2、4、6、8 Gy,细胞周期、细胞凋亡和DNA损伤实验照射剂量为6 Gy,剂量率为0.95 Gy/min。体内实验裸鼠照射前给予3.5%水合氯醛(0.3 g/kg)腹腔麻醉,待裸鼠麻醉后放入铅盒里,将右下肢足部固定于铅盒外进行局部照射,照射剂量为2 Gy。
-
将人结肠癌HCT 116细胞以1000个/孔接种到6孔细胞培养板中,待细胞贴壁后,按实验分组处理,继续培养10 d,至出现肉眼可见的克隆。弃掉培养基,用吉姆萨染液染色。肉眼进行克隆计数,以每团细胞数>50个作为1个克隆。
-
将人结肠癌HCT 116细胞以1×105个/孔接种到6孔板中,待细胞贴壁后,按实验分组处理。培养24 h后采用胰酶消化法收集细胞,用PBS洗涤,1800×g离心5 min。将细胞悬于500 μL 含300 μg/mL RNase A和10 μg/mL蛋白酶 K的10 μg/mL PI溶液中,4 ℃避光反应30 min,使用流式细胞仪的FACS caliber及 ModFit LTTM软件对10 000个细胞的周期分布进行检测。
-
将人结肠癌HCT 116细胞以1×105个/孔接种到6孔板中,待细胞贴壁后,按实验分组处理。分别培养24 h和48 h后采用胰酶消化法收集细胞,用PBS洗涤,1800×g离心5 min。加入500 μL结合缓冲液含[10 μL 磷脂结合蛋白V-异硫氰酸荧光素(Annexin V-FITC,0.5 μg/mL)和5 μL PI(5 μg/mL)],室温避光反应30 min。同时以不加Annexin V-FITC及PI的一管作为阴性对照。使用流式细胞仪的FACS caliber及 ModFit LTTM软件对10 000个细胞进行凋亡检测。
-
将1×105个人结肠癌HCT 116细胞接种到60 mm培养皿中,待细胞贴壁后,按实验分组处理。用PBS稀释成5×105个/mL的细胞悬液,加入低熔点琼脂糖胶混合,铺展到预冷的载玻片上。将凝固的载玻片浸在4℃碱性水解液(含2.5 moL NaCl、10 mmoL Tris base、1%十二烷基肌氨酸钠、30 mmoL乙二胺四乙酸二钠、10% 二甲基亚砜、1% Triton X-100)中作用2 h。然后在4℃下进行碱性电泳20 min,电流量为200 mA。载玻片用2.5 µg/L PI染液避光染色20 min,在倒置荧光显微镜下通过图像分析系统进行图像测量和数据分析。一个典型的受损细胞即“彗星”图像包括了头部和尾部,以尾部DNA含量、尾长、尾矩、Olive尾矩等参数作为细胞DNA损伤的分析指标。每个受测样品观察100个细胞。
-
将处于对数生长期的人结肠癌HCT 116细胞经胰酶消化,离心(1000×g离心8 min)后弃上清,用无血清培养液离心(1000×g离心8 min)洗涤2次,制备成1×107个/mL的细胞悬液。BALB/c-nu裸鼠右后腿皮下注射200 μL细胞悬液,裸鼠接种结肠癌细胞后约10 d形成肿瘤,当肿瘤最大直径长至3 mm时进行简单随机分组。
-
每个实验组为6只裸鼠,空白对照组:不给予任何处理;褪黑素组:于腹腔注射褪黑素,给药浓度为10 mg/kg,连续3 d;照射组:接受2 Gy/次照射,隔日照射,共3次;褪黑素+照射组:于腹腔注射褪黑素,给药浓度为10 mg/kg,连续3 d,第3天给药后1 h接受2 Gy/次照射,隔日照射,共3次。
-
定期观察瘤体体积的变化,于处理后第0、3、7、11、15天用游标卡尺测量肿瘤结节的最长径(a)和最短径(b)。根据公式V=1/6π(ab2) 计算肿瘤体积,取平均值并绘制肿瘤生长曲线。于第15天处死裸鼠,取瘤体称重,计算抑瘤率。抑瘤率=(空白对照组平均瘤重−实验组平均瘤重)/空白对照组平均瘤重×100%
-
采用SPSS 16.0软件进行统计学分析。计量资料符合正态分布,数据用
$\bar x$ ±$s$ 表示,在方差齐的条件下,两组间比较采用t检验。P<0.05表示差异有统计学意义。 -
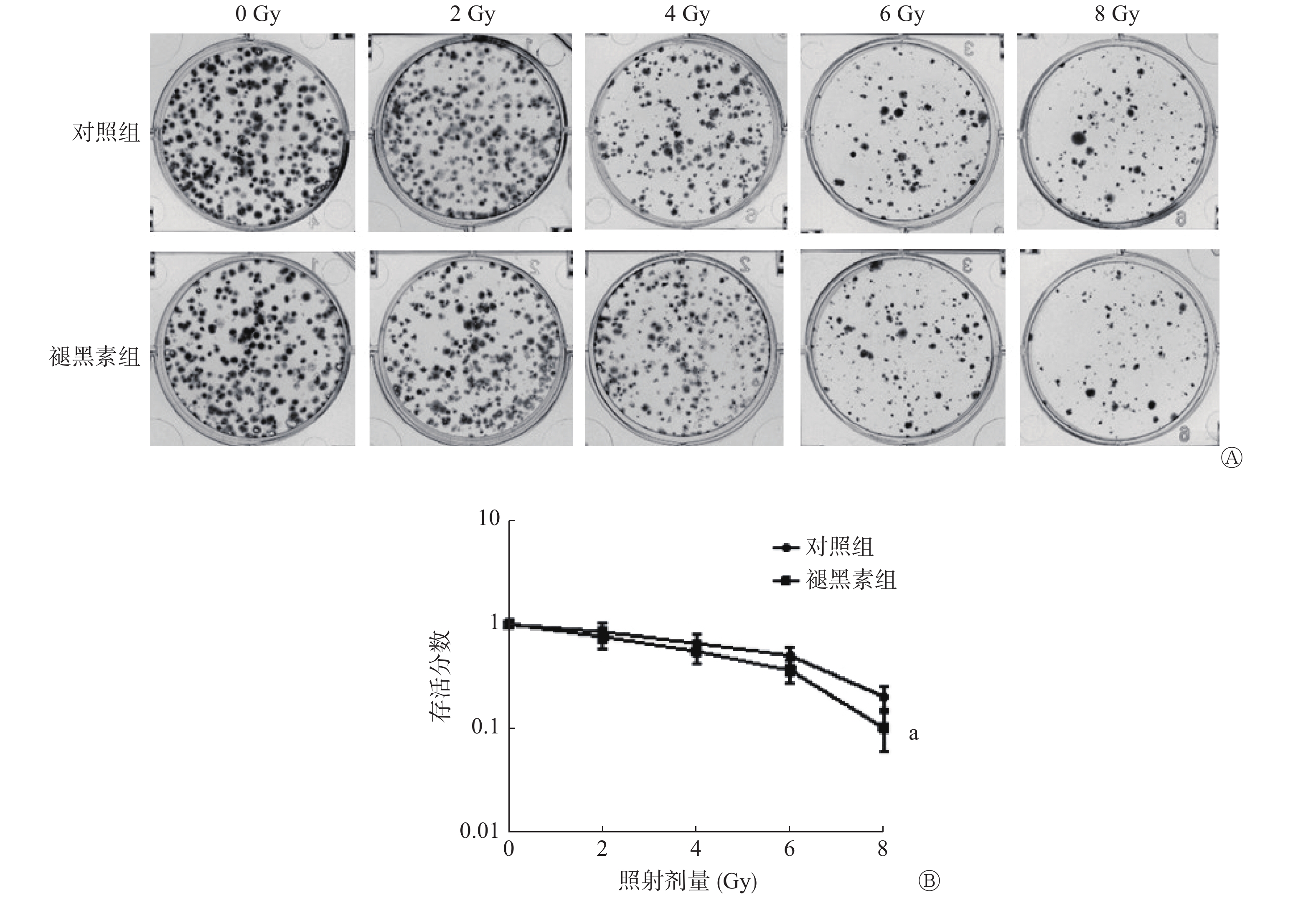
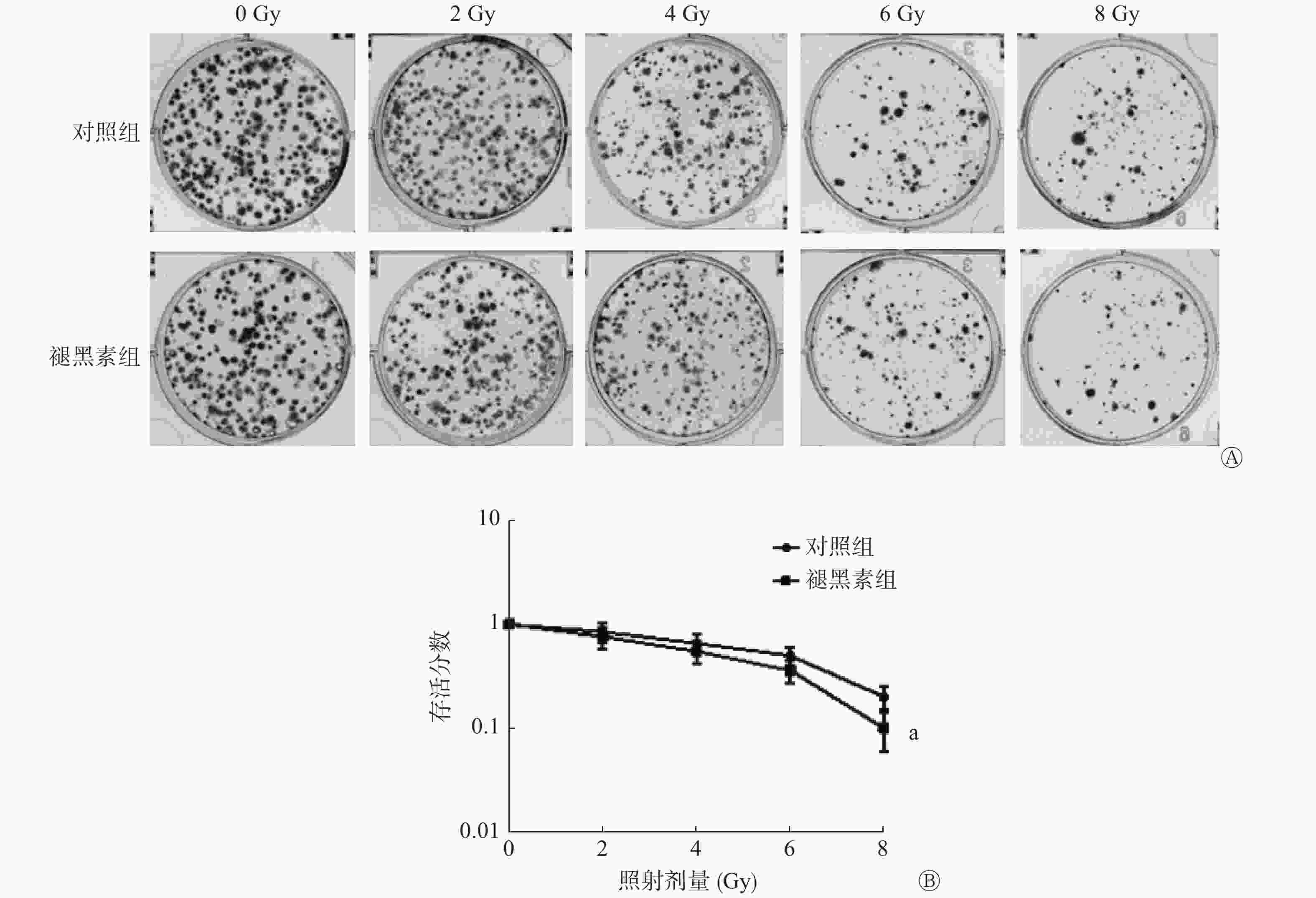
用克隆形成实验检测接受0、2、4、6、8 Gy照射后人结肠癌HCT 116细胞的增殖能力。其结果如图1中A所示,与对照组相比,照射前给予褪黑素后,随着照射剂量的增加,克隆形成数目显著减少。克隆存活曲线显示,照射前给予褪黑素,人结肠癌HCT 116 细胞的存活分数明显小于对照组,且差异有统计学意义(t=3.83,P=0.005)(图1中B)。以上结果表明,照射前给予褪黑素可有效抑制人结肠癌HCT 116细胞的增殖,提高人结肠癌HCT 116细胞的辐射敏感性。
-
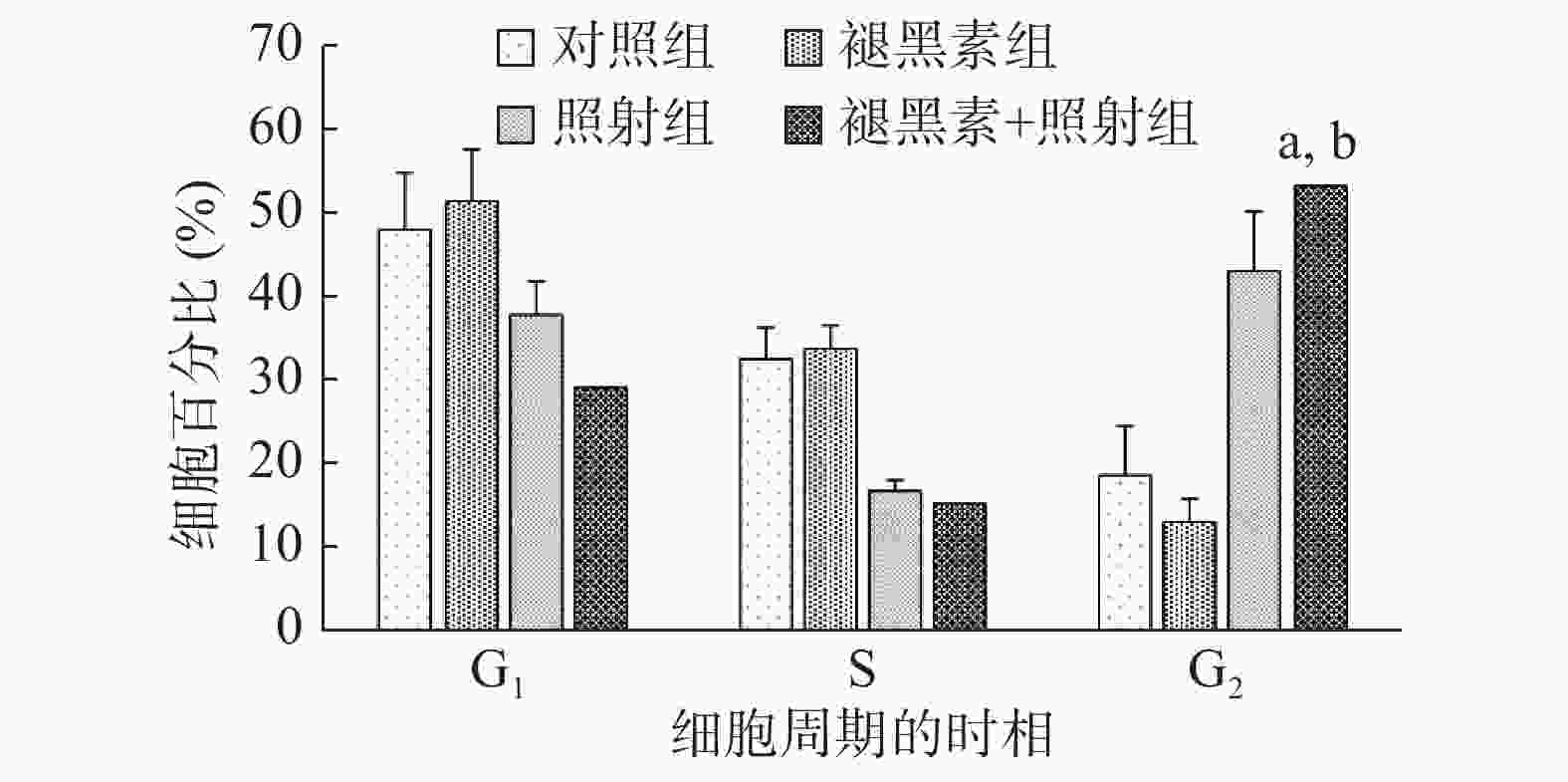
从流式细胞仪的分析结果可见,对照组和褪黑素组的人结肠癌HCT 116细胞主要阻滞在细胞周期的G1期;褪黑素+照射组在照射后阻滞在G2期的人结肠癌HCT 116细胞的比例大大增加(53.04%±4.67%),与照射组(42.83%±7.10%)和褪黑素组(12.95%±0.96%)相比,差异均有统计学意义(t=2.94、20.66,P=0.017、P<0.01)(图2)。以上结果表明,照射前给予褪黑素使人结肠癌HCT 116细胞从G1期逃离,发生G2期周期阻滞。
-
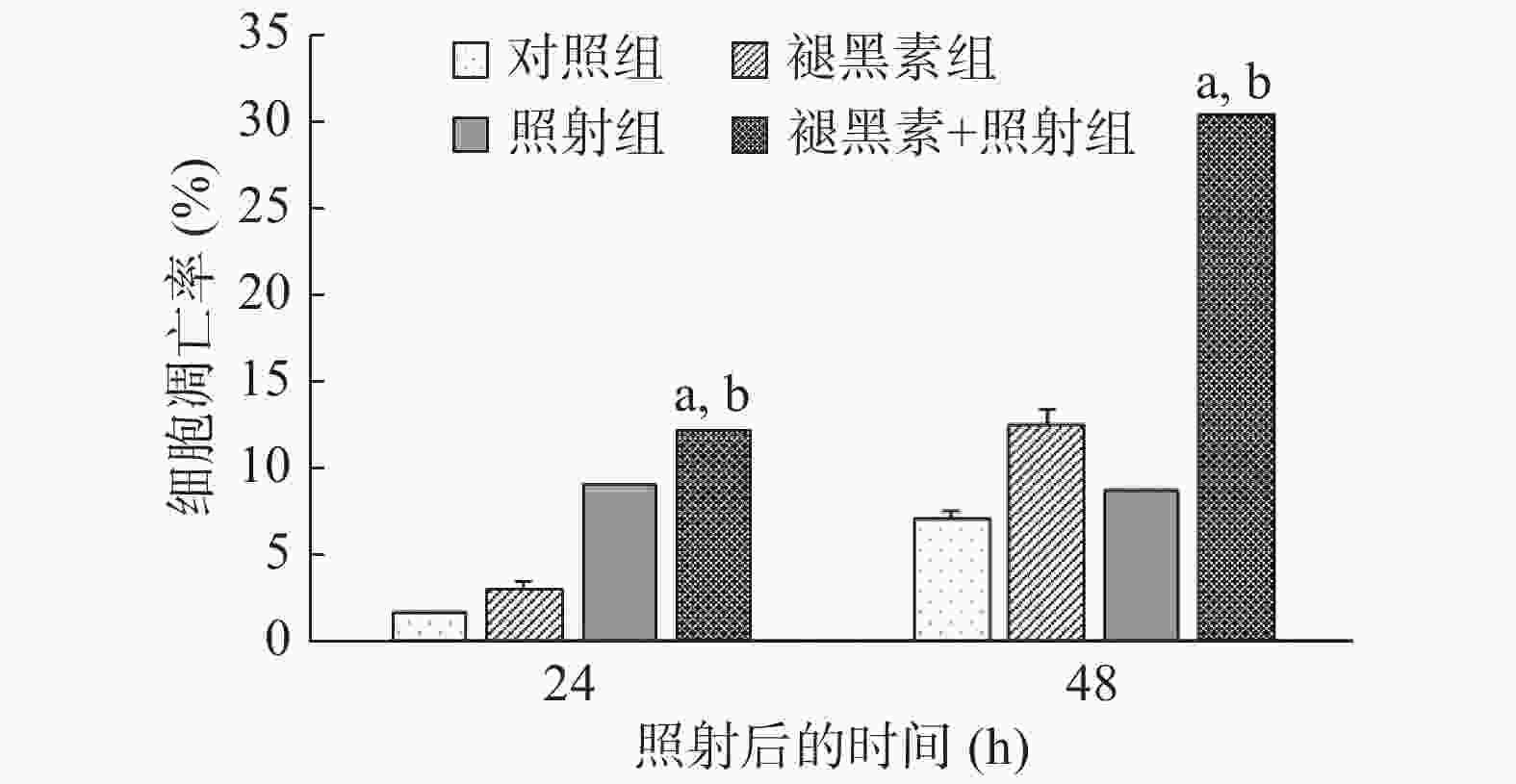
与对照组相比,褪黑素组和照射组在处理后24 h和 48 h,人结肠癌HCT 116细胞的凋亡数量均明显增加;褪黑素+照射组人结肠癌HCT 116细胞的凋亡率最高,分别达到(12.15±0.41)%和(30.57±1.91)%,与照射组(9.00%±0.70%、8.69%±0.71%)和褪黑素组(3.03%±0.42%、12.56%±0.89%)相比,差异均有统计学意义(t=7.46、17.75、29.12和14.80,均P<0.01)(图3)。以上结果表明,照射前给予褪黑素促使大量人结肠癌HCT 116细胞发生细胞凋亡。
-
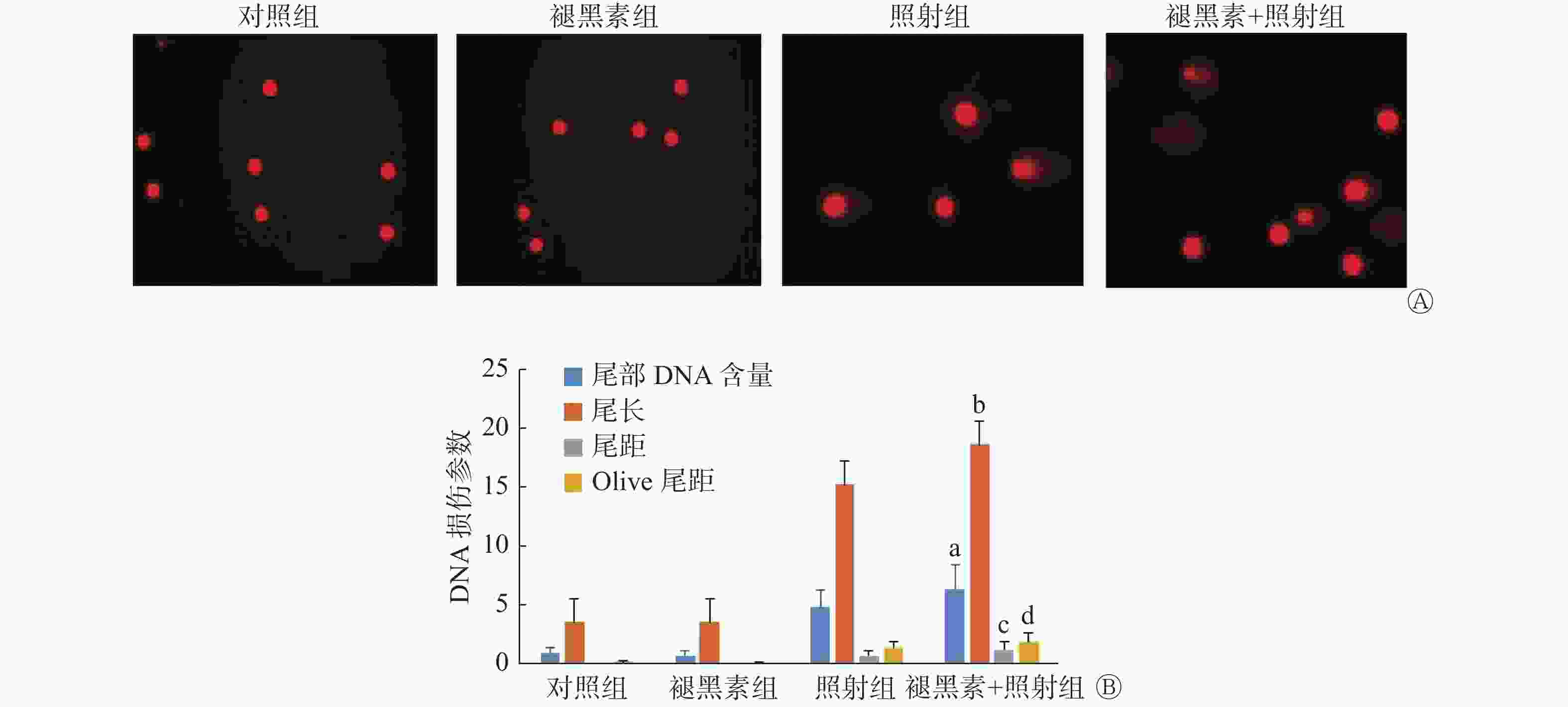
如图4中A所示,对照组和褪黑素组人结肠癌HCT 116细胞没有显示出拖尾样的彗星图像,而照射组细胞呈现明显的拖尾彗星,褪黑素+照射组细胞拖尾程度较照射组严重。彗星图像分析显示,褪黑素+照射组人结肠癌HCT 116细胞的尾部DNA含量、尾长、尾矩和Olive尾矩均明显高于照射组(t=4.72、4.16、4.74、4.50,均P<0.01)和褪黑素组(t=20.27、22.80、13.81和18.85,均P<0.01),差异均有统计学意义(图4中B)。以上结果表明,照射前给予褪黑素后人结肠癌HCT 116细胞DNA损伤修复能力下降,导致DNA损伤加重。
-
从肿瘤的生长曲线看出,对照组荷瘤裸鼠的肿瘤从处理的第7天后迅速增长,褪黑素组和照射组肿瘤较对照组生长速度缓慢;褪黑素+照射组肿瘤生长明显减慢,至第15天肿瘤体积显著小于照射组和褪黑素组,差异均有统计学意义(t=3.51和2.72,P=0.006和P=0.021)(图5中A)。
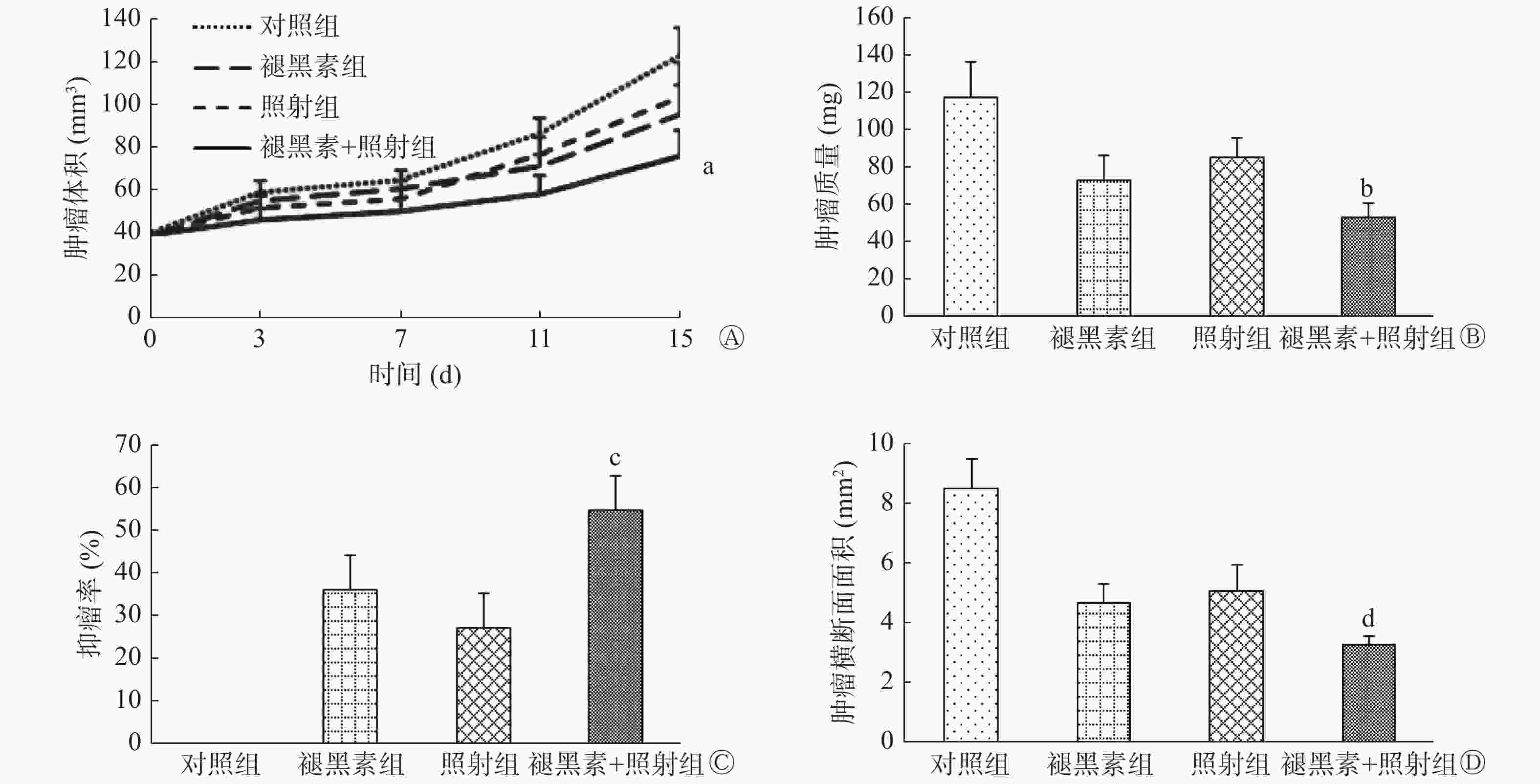
图 5 褪黑素联合照射后的各组荷瘤裸鼠肿瘤的生长
Figure 5. Melatonin suppressed the tumor growth in tumor-bearing nude mice following exposed to radiation
由图5中B~C所示,与对照组相比,照射组、褪黑素组和褪黑素+照射组的肿瘤质量明显减少,而褪黑素+照射组瘤重最轻,抑瘤率最高(54.7%±8.0%),远远高于照射组和褪黑素组(t=7.50和4.12,均P<0.01)。此外,由图5中D所示,褪黑素+照射组瘤体的横断面面积明显减小,与照射组、褪黑素组比较,差异均有统计学意义(t=4.72和4.68,P=0.003和P=0.002)。以上结果表明,照射前给予褪黑素提高了裸鼠结肠癌移植瘤的辐射敏感性,有效抑制了肿瘤的生长。
-
肿瘤的辐射抗性一直是临床上肿瘤放疗中遇到的难题。本研究通过观察褪黑素对辐射后结肠癌HCT 116细胞的抑瘤效应,探讨褪黑素对结肠癌辐射敏感性的影响。经典的克隆形成实验由于可准确检测到存活细胞而成为辐射敏感性判定的“金标准”[14]。本研究首先采用克隆形成法观察了体外结肠癌细胞辐射敏感性的变化。结果发现,照射前给予褪黑素导致人结肠癌HCT 116细胞的克隆形成数目显著减少,有效抑制了细胞的增殖。该结果提示,褪黑素可提高结肠癌细胞的辐射敏感性。
细胞周期是影响辐射敏感性的重要因素之一。肿瘤细胞对于电离辐射的敏感性依赖于其所处的细胞周期:M期细胞是对辐射最敏感的细胞;G2期细胞通常对辐射较敏感;G1期细胞对辐射不太敏感;晚S期细胞通常具有较大的辐射耐受性[15]。本研究结果发现,照射前给予褪黑素后,处于G2期的结肠癌HCT 116细胞比例大大增加,G1期和S期细胞明显减少。人结肠癌HCT 116细胞从辐射耐受的G1期逃离,阻滞于对辐射较敏感的G2期,因此结肠癌细胞表现出辐射敏感性的增强。
细胞凋亡与肿瘤细胞的辐射敏感性密切相关,凋亡反应的增加提示细胞具有更强的辐射敏感性。有研究报道,褪黑素通过促使人胃癌细胞和结肠癌细胞发生凋亡而发挥抗肿瘤效应[16-17]。与上述研究结果一致, 本研究结果发现,照射前给予褪黑素诱导大量的结肠癌细胞发生凋亡。该结果提示,经褪黑素处理后的结肠癌细胞对辐射较为敏感。
DNA是电离辐射最重要的靶点,DNA损伤修复能力也是影响辐射敏感性的重要因素之一。有文献报道,褪黑素可降低结直肠腺癌细胞DNA的损伤修复,导致DNA断片增加 [18]。褪黑素还可通过下调DNA损伤修复蛋白的表达而提高乳腺癌对辐射的敏感性[19]。同样,本研究结果发现,照射前给予褪黑素的HCT 116细胞DNA损伤加重。该结果提示,褪黑素降低了结肠癌细胞DNA的损伤修复能力,因而结肠癌表现出对辐射较为敏感。
同时,本研究观察褪黑素对裸鼠结肠癌移植瘤放射治疗的效果时发现,照射前给予褪黑素的裸鼠移植瘤生长明显缓慢,抑瘤率远远高于褪黑素组和照射组。该结果说明照射前给予褪黑素提高了裸鼠结肠癌对辐射的敏感性,有较好的放射治疗效果。本研究的体外实验和体内实验结果一致说明,照射前给予褪黑素能提高结肠癌细胞对辐射的敏感性。
大量的体外和体内研究发现,褪黑素的毒性很低,对动物无致死剂量;随机临床实验中口服褪黑素(20 mg /d)可减少肿瘤患者放疗的不良反应,从而改善患者的生活质量[12]。该研究结果提示,褪黑素有可能作为一种安全有效的辐射增敏剂,应用于结肠癌的临床放射治疗中。下一步研究将揭示褪黑素提高结肠癌细胞辐射敏感性的分子机制,为临床上解决结肠癌辐射抵抗性的难题奠定理论基础。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 王芹负责研究命题的设计、论文的撰写和最终版本的修订;徐畅负责方法的建立和数据的分析;杜利清负责现场的实验;王彦负责论文的起草和数据的获取;柳杨负责论文的起草和论文的审阅;刘强负责研究命题的设计和论文的审阅。
褪黑素对人结肠癌细胞辐射敏感性的影响
Effects of melatonin on the radiosensitivity of human colon cancer cells
-
摘要:
目的 分析褪黑素联合γ射线照射对体外和体内人结肠癌HCT 116细胞生长的影响,探讨褪黑素在人结肠癌HCT 116细胞辐射敏感性中的作用。 方法 将人结肠癌HCT 116细胞分为4组,即:空白对照组(不给予任何处理)、褪黑素组(给予褪黑素,给药浓度为1 mmol/L,给药时间为2 h)、照射组(接受6 Gy γ射线照射)及褪黑素+照射组(在照射前2 h给予褪黑素,给药浓度为1 mmol/L,然后接受6 Gy γ射线照射)。体外实验:人结肠癌HCT 116细胞分别进行2、4、6、8 Gy照射,采用克隆形成实验检测细胞的增殖能力;人结肠癌HCT 116细胞进行6 Gy照射,采用流式细胞术检测24 h后细胞周期以及24 h和48 h后细胞的凋亡;采用彗星实验检测2 h后细胞DNA的损伤。体内实验:将人结肠癌HCT 116细胞接种于裸鼠体内建立肿瘤模型,检测结肠癌瘤体体积和瘤体质量的变化并计算抑瘤率。两组间比较采用t检验。 结果 ①体外实验:照射前给予褪黑素处理的人结肠癌HCT 116 细胞的克隆形成数目明显少于对照组,差异有统计学意义(t=3.83,P=0.005);褪黑素+照射组停留在G2期的人结肠癌HCT 116细胞比例显著增加(53.04%±4.67%),与照射组(42.83%±7.10%)和褪黑素组(12.95%±0.96%)相比,差异均有统计学意义(t=2.94、20.66,P=0.017、P<0.01);褪黑素+照射组在处理后24 h和 48 h大量人结肠癌HCT 116细胞发生细胞凋亡,凋亡率分别达到(12.15±0.41)%和(30.57±1.91)%,与照射组(9.00%±0.70%、8.69%±0.71%)和褪黑素组(3.03%±0.42%、12.56%±0.89%)相比,差异均有统计学意义(t=7.46、17.75、29.12、14.80,均P<0.01);褪黑素+照射组HCT 116细胞的尾部DNA含量、尾长、尾矩和Olive尾矩均明显高于照射组(t=4.72、4.16、4.74、4.50,均P<0.01)和褪黑素组(t=20.27、22.80、13.81、18.85,均P<0.01),差异均有统计学意义。②体内实验:褪黑素+照射组结肠癌生长速度减慢,到处理后的第15天肿瘤体积明显小于照射组和褪黑素组,差异有统计学意义(t=3.51、2.72, P=0.006、P=0.021);褪黑素+照射组抑瘤率最高(54.7%±8.0%),远远高于照射组和褪黑素组(t=7.50、4.12,均P<0.01)。 结论 褪黑素联合辐射对人结肠癌细胞生长有显著的抑制效应,提高了细胞对γ射线辐射的敏感性。 Abstract:Objective To detect the effects of melatonin combined with γ-ray ionizing radiation on the proliferation of human colon cancer HCT 116 cells in vitro and in vivo and to explore the role of melatonin in regulating the radiosensitivity of HCT 116 cells. Methods The cohorts were divided into blank control group (HCT 116 cells were not given any treatment), melatonin group (HCT 116 cells were treated with 1 mmol/L melatonin for 2 h), radiation group (HCT 116 cells were exposed to 6 Gy γ-ray radiation), and melatonin+radiation group (HCT 116 cells were treated with 1 mmol/L melatonin for 2 h and then exposed to 6 Gy γ-ray radiation). In in vitro experiments, colony formation assay was used to detect cell proliferation after exposure to 0, 2, 4, 6, or 8 Gy radiation. Flow cytometry was applied to detect the cell cycle distribution at 24 h and cell apoptosis at 24 and 48 h after exposure to 6 Gy radiation. Comet assay was performed to detect DNA damage to cells 2 h after exposure to 6 Gy radiation. In in vivo experiments, the tumor-bearing nude mouse model was built by inoculating HCT 116 cells. The volume and inhibition ratio of tumor xenografts were examined. T-test was used for comparison between groups. Results ① In in vitro experiments, the colony number of HCT 116 cells treated with melatonin prior to radiation was significantly less than that of control cells (t=3.83, P=0.005). HCT 116 cells that were arrested at the G2 phase in the melatonin+radiation group (53.04%±4.67%) were increased, and significant differences were noted between the melatonin+radiation group and the radiation or melatonin group (t=2.940 and 20.660, P=0.017 and P<0.01, respectively). The cell apoptosis rate of HCT 116 cells in the melatonin+radiation group at 24 and 48 h after treatment was increased and reached (12.15±0.41)% and (30.57±1.91)%, respectively, which were markedly higher than those of the radiation (9.00%±0.70%, 8.69%±0.71%) or melatonin group (3.03%±0.42%, 12.56%±0.89%) (t=7.46, 17.75, 29.12, and 14.80, all P<0.01). The value of tail DNA, tail length, tail moment, and Olive tail moment in HCT 116 cells in the melatonin+radiation group were significantly higher than that in the radiation (t=4.72, 4.16, 4.74, 4.50, all P<0.01) or melatonin group (t=20.27, 22.80, 13.81, and 18.85, all P<0.01). ② In in vivo experiments, tumor xenografts in nude mice of the melatonin+radiation group grew slowly. The volume of tumor xenografts in the melatonin+radiation group at day 15 was significantly decreased compared with that in the radiation or melatonin group (t=3.51 and 2.72, P=0.006 and P=0.021, respectively). The inhibition ratio of xenografts in the melatonin+radiation group (54.7%±8.0%) was significantly higher than that in the radiation or melatonin group (t=7.50, 4.12, all P<0.01). Conclusion Melatonin combined with γ-ray radiation had obvious inhibition effect on colon cancer cells and increased the radiosensitivity of colon cancer cells. -
Key words:
- Colonic neoplasms /
- Radiation tolerance /
- Tumor-bearing nude mice /
- Melatonin
-
-
[1] Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries[J]. CA Cancer J Clin, 2018, 68(6): 394−424. DOI: 10.3322/caac.21492. [2] Alberti C. Melatonin: the first hormone isolated from the pineal body[J]. Farmaco Sci, 1958, 13(8): 604−605. [3] Su SC, Hsieh MJ, Yang WE, et al. Cancer metastasis: Mechanisms of inhibition by melatonin[J/OL]. J Pineal Res, 2017, 62(1): e12370 [2019-01-17]. https://onlinelibrary.wiley.com/doi/full/10.1111/jpi.12370. DOI: 10.1111/jpi.12370. [4] Singh M, Jadhav HR. Melatonin: functions and ligands[J]. Drug Discov Today, 2014, 19(9): 1410−1418. DOI: 10.1016/j.drudis.2014.04.014. [5] Alonso-González C, González A, Martínez-Campa C, et al. Melatonin enhancement of the radiosensitivity of human breast cancer cells is associated with the modulation of proteins involved in estrogen biosynthesis[J]. Cancer Lett, 2016, 370(1): 145−152. DOI: 10.1016/j.canlet.2015.10.015. [6] Zhou QY, Gui SY, Zhou Q, et al. Melatonin Inhibits the Migration of Human Lung Adenocarcinoma A549 Cell Lines Involving JNK/MAPK Pathway[J/OL]. PLoS One, 2014, 9(7): e101132 [2019-01-17]. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0101132. DOI: 10.1371/journal.pone.0101132. [7] Lu YX, Chen DL, Wang DS, et al. Melatonin enhances sensitivity to fluorouracil in oesophageal squamous cell carcinoma through inhibition of Erk and Akt pathway[J/OL]. Cell Death Dis, 2016, 7(10): e2432 [2019-01-17]. https://www.nature.com/articles/cddis2016330. DOI: 10.1038/cddis.2016.330. [8] Ju HQ, Li H, Tian T, et al. Melatonin overcomes gemcitabine resistance in pancreatic ductal adenocarcinoma by abrogating nuclear factor-κB activation[J]. J Pineal Res, 2016, 60(1): 27−38. DOI: 10.1111/jpi.12285. [9] Tai SY, Huang SP, Bao BY, et al. Urinary melatonin-sulfate/cortisol ratio and the presence of prostate cancer: A case-control study[J/OL]. Sci Rep, 2016, 6: 29606 [2019-01-17]. https://www.nature.com/articles/srep29606. DOI: 10.1038/srep29606. [10] Fic M, Gomulkiewicz A, Grzegrzolka J, et al. The Impact of Melatonin on Colon Cancer Cells' Resistance to Doxorubicin in An In Vitro Study[J/OL]. Int J Mol Sci, 2017, 18(7): 1396[2019-01-17]. https://www.mdpi.com/1422-0067/18/7/1396. DOI: 10.3390/ijms18071396. [11] Bułdak RJ, Pilc-Gumuła K, Bułdak Ł, et al. Effects of ghrelin, leptin and melatonin on the levels of reactive oxygen species, antioxidant enzyme activity and viability of the HCT 116 human colorectal carcinoma cell line[J]. Mol Med Rep, 2015, 12(2): 2275−2282. DOI: 10.3892/mmr.2015.3599. [12] Onseng K, Johns NP, Khuayjarernpanishk T, et al. Beneficial Effects of Adjuvant Melatonin in Minimizing Oral Mucositis Complications in Head and Neck Cancer Patients Receiving Concurrent Chemoradiation[J]. J Altern Complement Med, 2017, 23(12): 957−963. DOI: 10.1089/acm.2017.0081. [13] Zou ZW, Liu T, Li Y, et al. Melatonin suppresses thyroid cancer growth and overcomes radioresistance via inhibition of p65 phosphorylation and induction of ROS[J]. Redox Biol, 2018, 16: 226−236. DOI: 10.1016/j.redox.2018.02.025. [14] Hirota Y, Masunaga S, Kondo N, et al. High linear-energy-transfer radiation can overcome radioresistance of glioma stem-like cells to low linear-energy-transfer radiation[J]. J Radiat Res, 2014, 55(1): 75−83. DOI: 10.1093/jrr/rrt095. [15] Pawlik TM, Keyomarsi K. Role of cell cycle in mediating sensitivity to radiotherapy[J]. Int J Radiat Oncol Biol Phys, 2004, 59(4): 928−942. DOI: 10.1016/j.ijrobp.2004.03.005. [16] Li W, Fan M, Chen Y, et al. Melatonin Induces Cell Apoptosis in AGS Cells Through the Activation of JNK and P38 MAPK and the Suppression of Nuclear Factor-Kappa B: A Novel Therapeutic Implication for Gastric Cancer[J]. Cell Physiol Biochem, 2015, 37(6): 2323−2338. DOI: 10.1159/000438587. [17] Wenzel U, Nickel A, Daniel H. Melatonin potentiates flavone-induced apoptosis in human colon cancer cells by increasing the level of glycolytic end products[J]. Int J Cancer, 2005, 116(2): 236−242. DOI: 10.1002/ijc.20837. [18] Kontek R, Nowicka H. The modulatory effect of melatonin on genotoxicity of irinotecan in healthy human lymphocytes and cancer cells[J]. Drug Chem Toxicol, 2013, 36(3): 335−342. DOI: 10.3109/01480545.2012.737805. [19] Alonso-González C, González A, Martínez-Campa C, et al. Melatonin sensitizes human breast cancer cells to ionizing radiation by downregulating proteins involved in double-strand DNA break repair[J]. J Pineal Res, 2015, 58(2): 189−197. DOI: 10.1111/jpi.12205. -





 下载:
下载: