-
聚酰胺-胺(polyamidamines,PAMAM)是应用最广泛的树枝状聚合物,其以正电荷形式存在,表面具有大量氨基基团,便于修饰和被细胞摄取,内部空膨广阔并且含有大量的疏水空腔,可以通过非共价作用包裹疏水的药物分子,目前已应用于包括药物输送、DNA及小干扰RNA递送、癌细胞凋亡检测、靶向用药和肿瘤成像等领域[1-3]。甲状腺髓样癌(medullary thyroid carcinoma,MTC)来源于不表达钠碘共同转运体的甲状腺滤泡旁细胞,不能进行放射性碘治疗,其治疗方法主要以及时甲状腺根治性外科手术为首选[4-5]。因此,术前早期诊断、准确定位原发灶及转移灶至关重要。然而,常用的肿瘤标志物降钙素及癌胚抗原与肿瘤负荷密度密切相关,应用其对MTC诊断易出现假阴性结果[6]。超声对MTC的诊断缺乏特异性,MTC可见甲状腺乳头状癌的超声特征,甚至多达1/3的MTC具有“非恶性”超声表现[7]。2015年美国甲状腺协会指南[8]及Trimboli等[9]Meta分析结果显示,细针穿刺细胞学检查对MTC诊断的准确率不到50%。为此,本研究旨在利用经修饰后的第五代聚酰胺-胺[PAMAM(G5.0)]纳米平台装载131I及不同靶向肽[靶向MTC细胞的丝氨酸-精氨酸-谷氨酸-丝氨酸-脯氨酸-组氨酸-脯氨酸(Ser-Arg-Glu-Ser-Pro-His-Pro, SRESPHP)(简称SR)、靶向肿瘤新生血管的甘氨酸-脯氨酸-亮氨酸-脯氨酸-亮氨酸-精氨酸(Gly-Pro-Leu-Pro-Leu-Arg, GPLPLR)(简称GP)及双靶向肽SR/GP],分别合成不同的分子靶向探针,探讨其在MTC模型中的靶向性,现报道如下。
-
胎牛血清、F12培养基以及胰酶购自美国Gibco公司。PBS购自上海碧云天生物技术有限公司。Na131I购自原子高科股份有限公司。靶向肽SR、GP与树枝状聚合物PAMAM(G5.0)共价合成的PAMAM(G5.0)-SR、PAMAM(G5.0)-GP和PAMAM(G5.0)-SR/GP由项目合作方美国堪萨斯州立大学化学部提供。C18固相萃取柱购自美国安捷伦公司。氯胺T购自南京化学试剂股份有限公司。偏重亚硫酸钠购自无锡市晶科化工有限公司。甲醇购自浙江三鹰化学试剂有限公司。新华1号层析纸购自杭州新华纸业公司。
1470全自动γ计数仪购自美国PerkinElmer公司。MINI-SCAN TLC薄层放射性扫描仪购自美国BIOSCAN公司。Symbia T16 SPECT/CT仪购自德国Siemens公司。CRC-25R放射性核素活度计购自北京华瑞森科技发展有限公司。Agilent1100/1200系列高效液相色谱仪购自美国安捷伦公司。Nicomp 380 动态光散射粒度分析仪购自美国PSS公司。
-
人MTC细胞(TT细胞株)购自广州吉妮欧生物科技有限公司。雌性BALB/c-nu鼠75只购自湖南斯莱克景达实验动物有限公司,合格证号:SCXK(湘)2016-0002,4~5周龄,体重为18~22 g。
-
TT细胞采用F12培养基(含10%胎牛血清)于37℃、5%CO2细胞培养箱中培养,培养基每周例行更换3次,选择处于对数生长期的TT细胞用于实验。常规消毒后于BALB/c-nu鼠左腋窝皮下注射1×107个/0.2 mL的肿瘤细胞悬液,荷人MTC TT细胞鼠在无特定病原体级标准环境中饲养,食物充足。每周测量一次肿瘤大小,待肿瘤长至1.5 cm3左右时进行实验。取眼球血检测降钙素和癌胚抗原水平,取肿瘤组织进行病理及免疫组化检测,对建立的肿瘤模型进行验证。
-
采用固相肽链合成法分别合成靶向肽SR、GP及SR/GP并对其进行高效液相色谱纯化及分析,选用反相C18柱(2.1 mm×50 mm,3.5 μm)分离,柱温40℃,流速0.3 mL/min,进样量10 μL,流动相A为体积分数比为1000∶1的纯水与三氟乙酸,B液为体积分数比为1000∶1的乙腈与三氟乙酸,紫外检测波长为254 nm。分别将PAMAM(G5.0)与靶向肽SR、GP及SR/GP通过共价键连接合成前体药物PAMAM(G5.0)-SR、PAMAM(G5.0)-GP和PAMAM(G5.0)-SR/GP,将这3种前体药物分别溶于双蒸水,稀释至质量浓度为1 mg/mL,再用Millipore 0.22 μm针头过滤器过滤,随后应用动态光散射粒度分析仪测定其纳米粒径及Zeta电位。
-
分别于含有100 μL(质量浓度为30 mg/mL)的PAMAM(G5.0)、PAMAM(G5.0)-SR、PAMAM(G5.0)-GP和PAMAM(G5.0)-SR/GP的离心管中加入约555 MBq(15 mCi)的Na131I(比放射性浓度为3.7 GBq/mL)和60 μL氯胺T(质量浓度为1 mg/mL),充分混匀,室温下反应13 min,随后加入60 μL偏重亚硫酸钠(质量浓度为30 mg/mL)终止反应。将上述混合物分别用C18萃取柱过滤杂质,最后用300 μL(0.1 mol/L)盐酸乙醇洗脱后获得放射性标记探针131I-PAMAM(G5.0)、131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP。
-
采用薄层色谱法测定131I-PAMAM(G5.0)、131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP及131I-PAMAM(G5.0)-SR/GP的放射化学纯度及稳定性。用毛细管取原液点样于新华1号层析纸上,于甲醇∶生理盐水体积比为8.5∶1.5的展开剂中上行展开,在空气中自然晾干,用薄层放射性扫描仪扫描后计算探针的放射化学纯度。取约74 MBq(2 mCi)131I-PAMAM(G5.0)、131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP分别置于200 μL PBS中,混匀,室温下孵育1、2、4、8、24、48 h后采用薄层色谱法检测并观察其体外稳定性。
-
荷瘤裸鼠75只(为保护模型鼠的甲状腺,实验前24 h给予5%碘化钾溶液封闭甲状腺直至实验结束)分为5组,每组15只。于腹腔分别注射新鲜制备的Na131I(阴性对照组)、131I-PAMAM(G5.0)(阳性对照组)和131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP(实验组),每只注射37 MBq/200 μL(1 mCi/200 μL),注射后4、8、12、24、48、72 h用体积分数为2%异氟烷气体麻醉并行SPECT/CT显像,同时勾画肿瘤与对侧相应部位的ROI(大小约2.0 cm×2.0 cm),并计算T/NT。
-
4、8、12、24 h SPECT/CT显像结束后,各组分别处死3只荷瘤裸鼠,并对肿瘤、甲状腺、全血、肝、胰、脾、肾、胃、肠等器官或组织进行称重,用γ计数器测定其放射性,以每克组织百分注射剂量率(percentage activity of injection dose per gram of tissue,%ID/g)表示。%ID/g=组织放射性计数/(注射药物放射性计数×组织质量)×100%。
-
应用IBM SPSS 20.0软件进行数据分析。所得数据符合正态分布以均数±标准差(
${\bar {x}} \pm {s}$ )表示,符合偏态分布以四分位数[M(P25,P75)]表示。同一时间点不同探针的T/NT比较,同一探针不同时间点的T/NT比较,以及24 h不同探针的肿瘤放射性摄取值比较均采用单因素方差分析。在方差齐的条件下,两两比较采用LSD-t检验。P<0.05表示差异有统计学意义。 -
纯化后靶向肽SR、GP及SR/GP的纯度达99%。PAMAM(G5.0)、PAMAM(G5.0)-SR、PAMAM(G5.0)-GP、PAMAM(G5.0)-SR/GP的纳米粒径分别为4.47、5.70、4.71、5.95 nm,Zeta电位分别为+37.95、+20.02、+28.34、+24.37 mV。
-
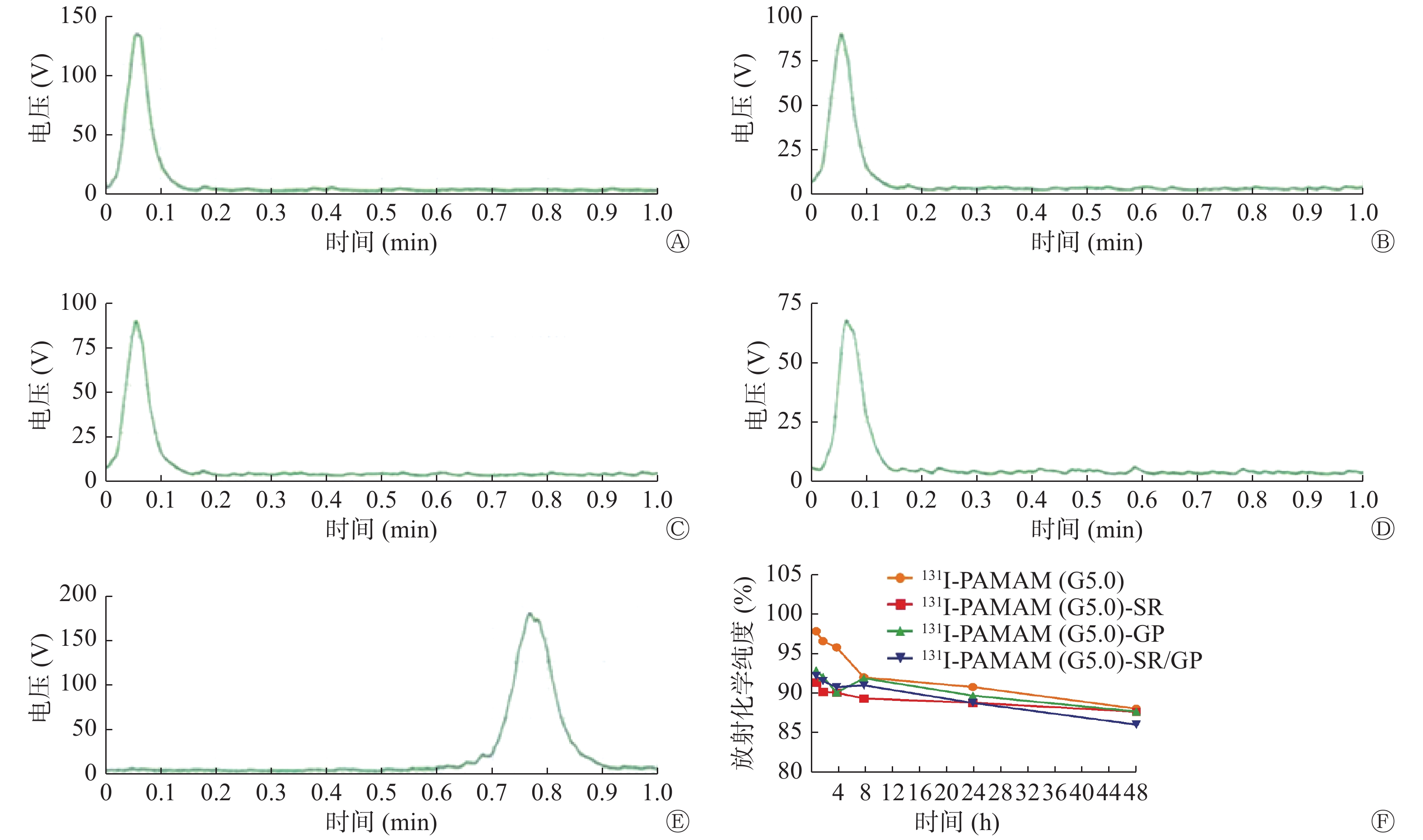
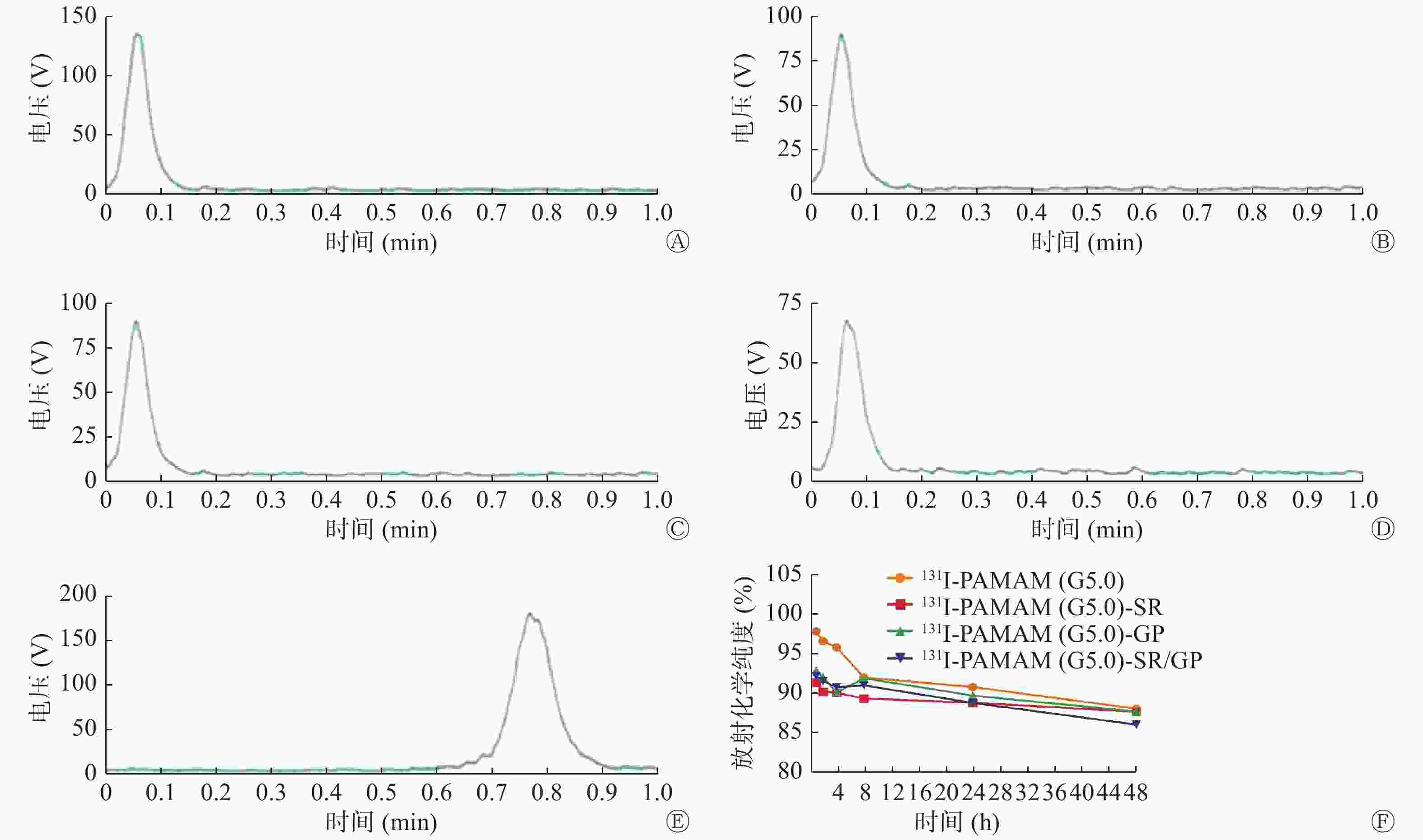
131I-PAMAM(G5.0)、131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP的标记率分别为77.48%、76.33%、75.21%和75.26%。放射化学纯度检测结果显示,标记物集中在原点(图1中A~D),而游离的Na131I随溶剂迁移到层析纸前沿(图1中E),131I-PAMAM(G5.0)、131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP的放射化学纯度分别为99.91%、93.44%、93.98%和90.28%。131I-PAMAM(G5.0)、131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP在PBS中静置48 h后,放射化学纯度均在85%以上,并且4种探针的放射化学纯度无明显变化(图1中F)。
-
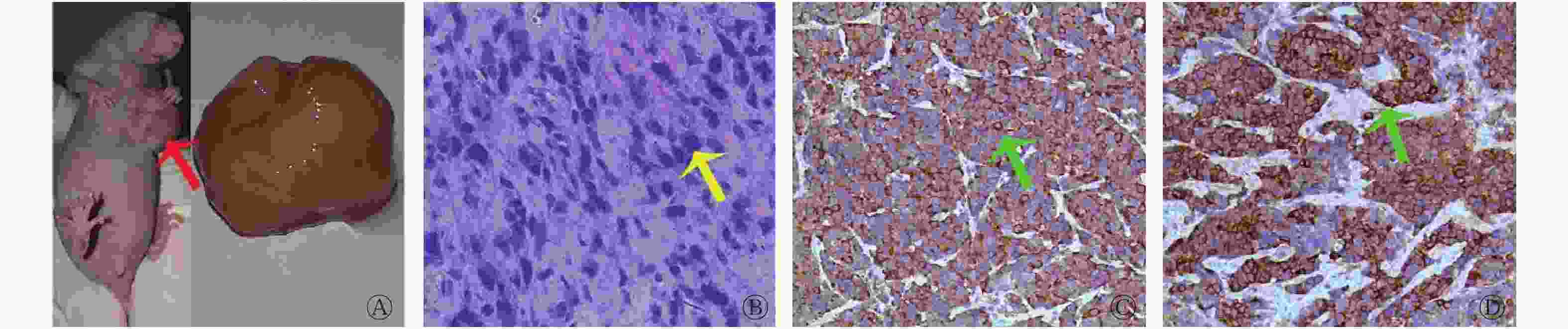
血清体外分析结果显示,降钙素水平为(347.76 ± 84.4 )pg/mL,癌胚抗原水平为(7.84 ± 3.18)ng/mL。荷瘤模型鼠及肿瘤标本如图2中A所示。肿瘤组织病理结果显示细胞量丰富,细胞呈浆细胞样,部分呈多角型,细胞核呈偏心性(图2中B);免疫组化结果显示,降钙素染色细胞质染色阳性(图2中C),癌胚抗原染色细胞质染色阳性(图2中D)。以上结果提示本研究中荷瘤裸鼠模型建立成功。
-
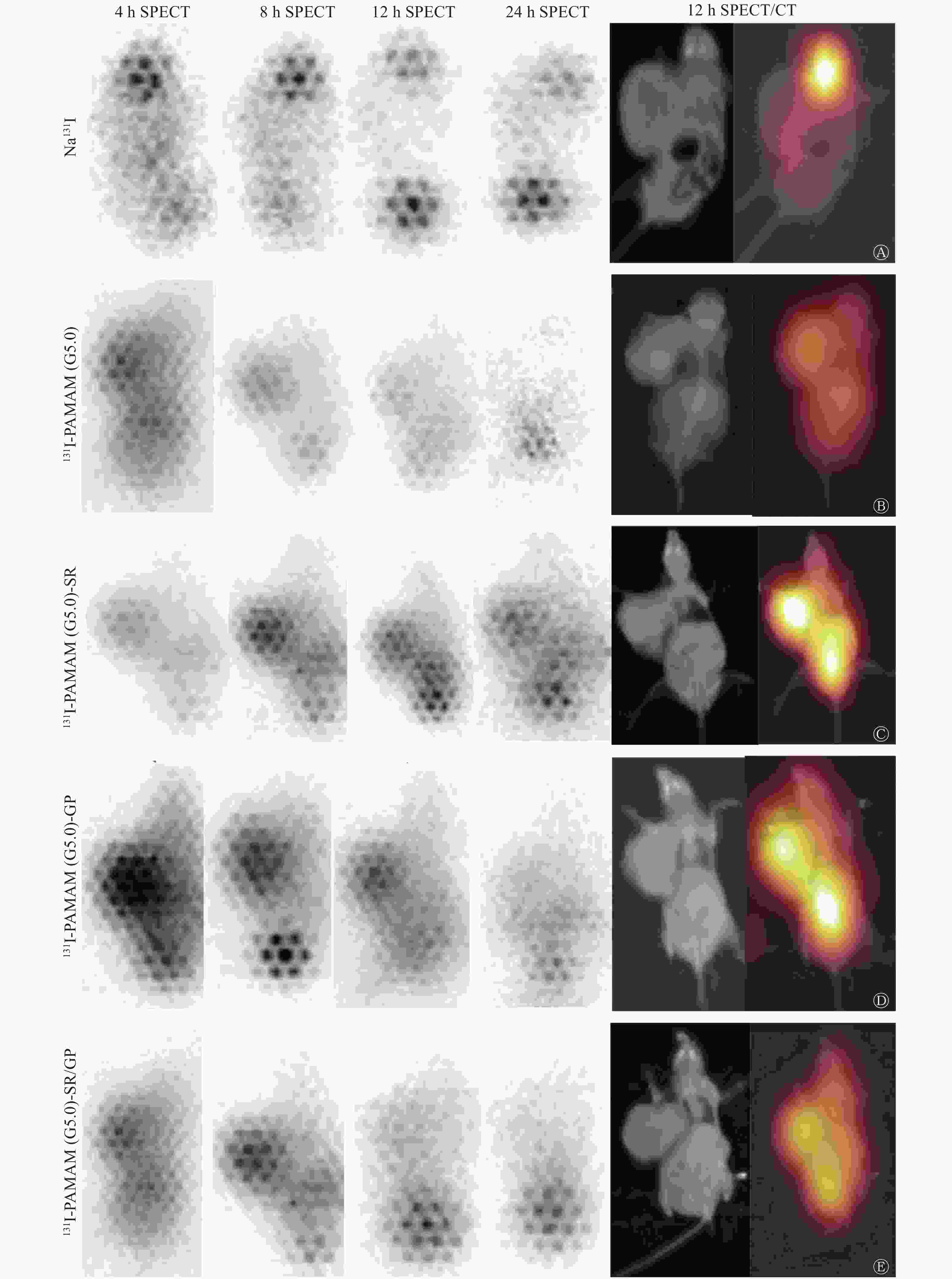
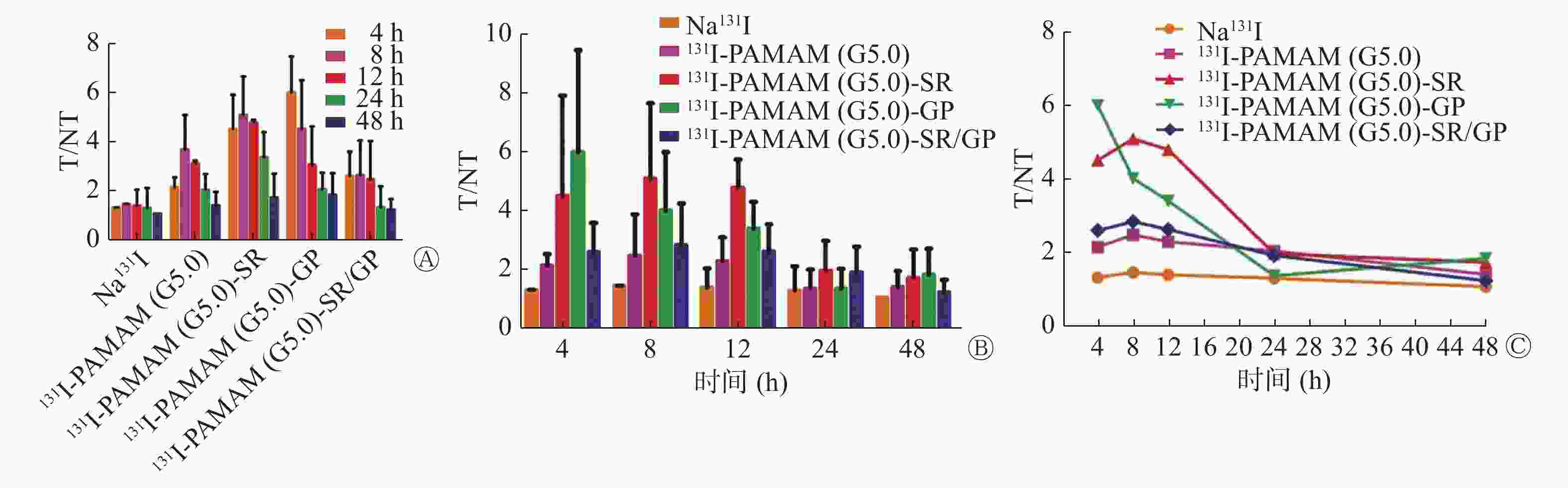
SPECT/CT显像结果显示,注射Na131I后荷瘤裸鼠甲状腺区放射性摄取明显(图3中A),注射131I-PAMAM(G5.0)后4 h和8 h肿瘤部位放射性摄取浅淡,但洗脱较快(图3中B),注射131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP及131I-PAMAM(G5.0)-SR/GP后,肿瘤部位有不同程度的放射性摄取,但随着时间的推移有不同程度的洗脱(图3中C~E)。与对照组比较,实验组不同时间SPECT/CT显像的T/NT均较高,4、8、12和24 h时不同探针的T/NT比较差异有统计学意义(F=3.532、3.151、6.972、3.864,P=0.042、0.048、0.003、0.038),48 h时差异无统计学意义(F=0.818,P=0.535);4 h时131I-PAMAM(G5.0)-GP的T/NT(6.03±1.45)较阳性(2.18±0.39)和阴性对照组(1.36±0.00)均明显升高,且差异均有统计学意义(t=3.235,P=0.033;t=3.834,P=0.019);8、12、24 h时131I-PAMAM(G5.0)-SR的T/NT(5.12±1.65、4.82±0.09、3.41±1.01)较阴性对照组(1.50±0.00、1.43±0.65、1.34±0.81)明显升高,且差异均有统计学意义(t=4.004,P=0.017;t=3.388,P=0.027;t=4.180,P=0.009)。实验组组内比较,24 h时131I-PAMAM(G5.0)-SR的T/NT(3.41±1.01)较131I-PAMAM(G5.0)-GP(2.10±0.67)明显升高,且差异有统计学意义(t=3.996,P=0.016)(图4中A)。注射131I标记的4种探针后T/NT的达峰时间:131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP均为4 h,131I-PAMAM(G5.0)和131I-PAMAM(G5.0)-SR均为8 h。达峰后随着时间的推移缓慢下降,131I-PAMAM(G5.0)-GP的放射性洗脱较快,12 h与4 h比较下降约57%。131I-PAMAM(G5.0)、131I-PAMAM(G5.0)-GP不同时间点的T/NT比较,差异有统计学意义(F=4.670,P=0.012;F=3.801,P=0.025),131I-PAMAM(G5.0)-GP 24 h和48 h的T/NT较4 h明显降低(t=−6.648,P=0.004;t=−5.200,P=0.007),131I-PAMAM(G5.0)48 h的T/NT较8 h明显降低(t=−7.173,P=0.002);131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-SR/GP不同时间点的T/NT比较,差异无统计学意义(F=2.520,P=0.085;F=1.559,P=0.236)。而Na131I在各个时间点的T/NT均处于较低水平,且随时间推移无明显变化(图4中B~C)。
-
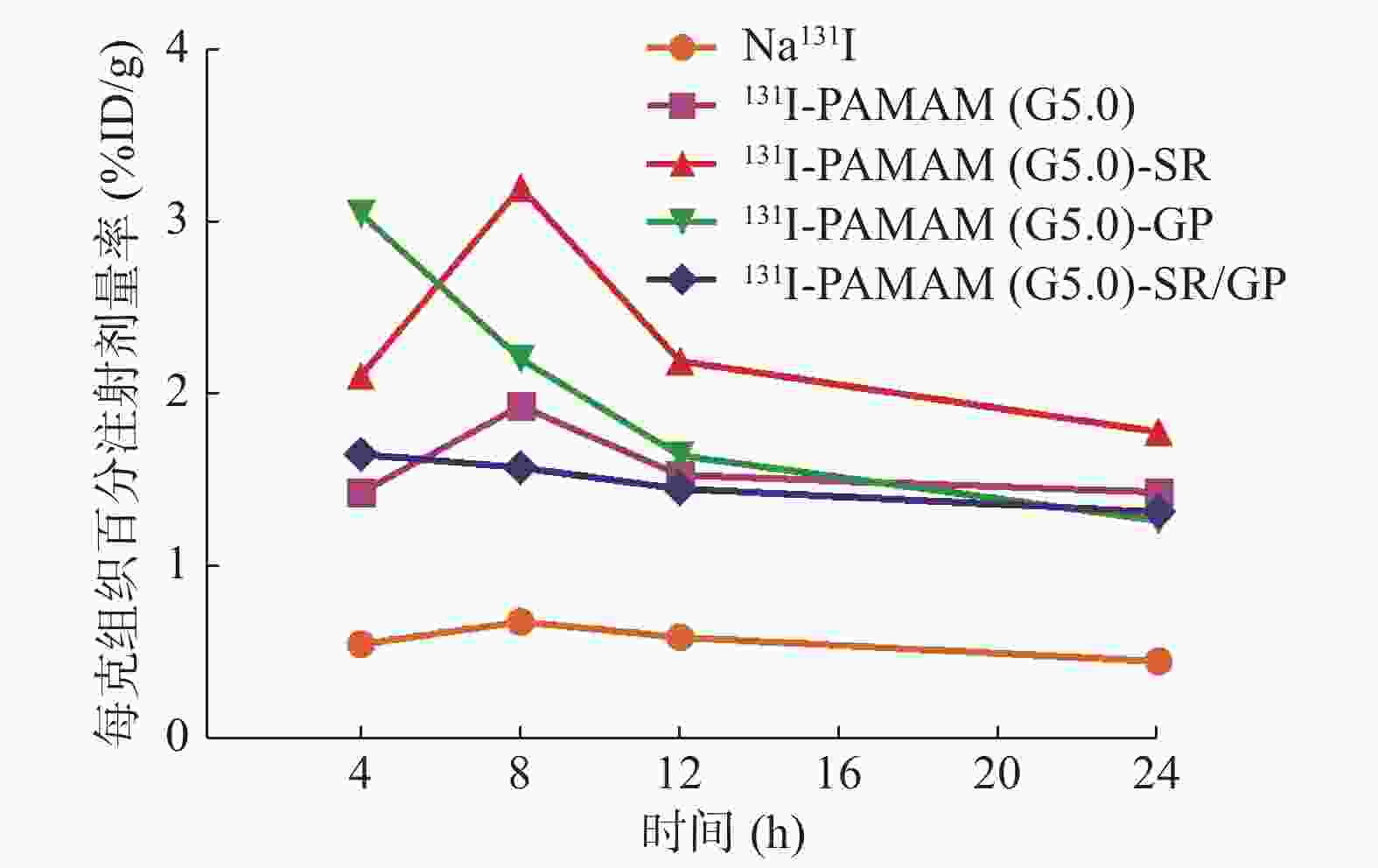
注射131I标记的4种探针后肿瘤放射性的达峰时间及清除过程与SPECT/CT显像T/NT结果一致。4 h时肿瘤放射性131I-PAMAM(G5.0)-GP最高,8、12和24 h时131I-PAMAM(G5.0)-SR最高(图5)。131I标记分子探针及Na131I注射后24 h在MTC模型中的生物学分布见表1,各组间肿瘤组织的放射性差异无统计学意义(F=3.366,P=0.059)。131I-PAMAM(G5.0)主要分布于甲状腺、肾脏及脾脏;131I-PAMAM(G5.0)-SR主要分布于肾脏、胃、全血、脾脏及肿瘤;131I-PAMAM(G5.0)-GP主要分布于肾脏、肿瘤及肝脏;131I-PAMAM(G5.0)-SR/GP主要分布于甲状腺、肾脏、全血及肿瘤;而Na131I主要分布于甲状腺、全血及胃。
器官或组织 131I- PAMAM(G5.0) 131I- PAMAM(G5.0)-SR 131I- PAMAM(G5.0)-GP 131I- PAMAM(G5.0)-SR/GP Na131I 肿瘤 1.45±0.28 1.80±0.18 1.58±0.14 1.13±0.33 0.57±0.25 肝 0.50±0.41 0.89±0.75 1.11±1.62 0.39±0.52 0.28±0.15 脾 2.02±1.83 2.28±2.98 0.21±0.17 0.61±0.54 0.28±0.03 胰 0.70±0.65 0.76±0.62 0.30±0.26 0.19±0.21 0.07±0.02 肾 3.02±1.50 8.08±4.63 3.41±3.18 2.46±1.82 0.33±0.02 肠 0.59±0.49 2.35±2.21 0.47±0.70 0.29±0.35 0.23±0.00 全血 0.51±0.49 2.64(0.01,4.40) 0.53±0.40 1.51±1.67 5.93±2.10 甲状腺 4.02±5.31 1.41±1.88 0.47±0.31 0.69(0.58,6.67) 832.00±159.73 胃 0.92±0.64 5.11±4.48 0.52±0.45 0.32±0.25 1.39±0.26 注:表中,MTC:甲状腺髓样癌;PAMAM(G5.0):第五代聚酰胺-胺;SR:丝氨酸-精氨酸-谷氨酸-丝氨酸-脯氨酸-组氨酸-脯氨酸(SRESPHP);GP:甘氨酸-脯氨酸-亮氨酸-脯氨酸-亮氨酸-精氨酸(GPLPLR);%ID/g:每克组织百分注射剂量率。 表 1 131I标记探针注射后24 h在荷MTC裸鼠体内的生物学分布[(M(P25,P75),
$\bar{x}\pm s$ Table 1. %ID/g of the tumor and important organs at 24 h post-injection of probes labeled with radioisotope 131I [(M(P25, P75),
$\bar{x}\pm s$ -
PAMAM分子表面具有大量氨基基团,极易被修饰,其具有热力学和动力学的稳定性好、比表面积大,生物相容性好等优点[10-12]。此外,PAMAM高度枝化,内部空膨广阔[13]。本研究对PAMAM(G5.0)进行修饰,使之与纯化后的靶向肽通过共价键连接形成前体药物,随着与靶向肽SR、GP及SR/GP的键合,PAMAM(5.0)的纳米粒径增大,Zeta电位缩小,提示靶向肽连接成功并且带负电荷。再应用氯胺T的氧化作用将碘阴离子氧化成碘分子,从而取代前体药物羟基位的1个或2个氢离子,使修饰后的前体药物成为含有131I、放射化学纯度较高、体外稳定性好的分子探针。
Kondo等[14]发现用脂质体载入肽链GP后行PET/CT显像者,其在人脐带静脉内皮细胞和肿瘤细胞的浓聚程度能比单用脂质体者高出4倍,并且同时应用DPP-CNDAC/GP(其中,DPP-CNDAC:二棕榈酰磷酯酰-2′-C-氰基-2′-脱氧-1-β-D-阿拉伯-戊呋喃糖基胞嘧啶)比单用DPP-CNDAC抗癌药物具有较强的抗肿瘤生长作用。因多肽GP与脂质体制剂结合能靶向易于新血管形成的肿瘤组织,多肽GP能与肿瘤细胞和肿瘤新生血管内皮细胞上过多表达的特异性肿瘤血管标志物Ⅰ型基质金属蛋白酶结合[15-16]。本研究中注射含有靶向肽GP的探针131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP后,肿瘤放射性达峰时间为4 h,早于不含靶向肽GP的131I-PAMAM(G5.0)和131I-PAMAM(G5.0)-SR,并且131I-PAMAM(G5.0)-GP 12 h的肿瘤放射性较4 h下降约57%,这提示靶向肽GP在MTC内摄取和排泄较快。131I-PAMAM(G5.0)-GP 4 h的T/NT较131I-PAMAM(G5.0)明显升高,差异具有统计学意义,这提示靶向肽GP提高了131I-PAMAMM(G5.0)对肿瘤的靶向性。MTC属于中度恶性肿瘤,新生血管丰富,我们前期的体外实验结果表明,与131I-PAMAM(G5.0)相比,131I-PAMAM(G5.0)-GP对TT细胞的靶向性差异并无统计学意义[17],而体内实验结果显示二者对MTC的靶向性差异有统计学意义,因此131I-PAMAM(G5.0)-GP有望应用于活体MTC的靶向成像。
MTC表面能表达SR特异性受体,SR作为靶向肽连接在腺病毒上可以特定靶向和杀伤MTC细胞,并且体内外实验结果显示SR能选择性地结合到人MTC细胞上,从而使结合到其他组织的量减少达90%以上[18-19]。我们的前期研究结果也表明,与131I-PAMAM(G5.0)相比,131I-PAMAM(G5.0)-SR能够更好地靶向TT细胞并抑制其生长[20]。本研究结果显示,131I-PAMAM(G5.0)-SR 于8、12及24 h的T/NT及肿瘤放射性均较实验组的其余探针、阴性对照组及阳性对照组高。与阴性对照组比较,131I-PAMAM(G5.0)-SR 于8、12及24 h的T/NT均明显升高,差异有统计学意义。实验组内比较,131I-PAMAM(G5.0)-SR 24 h的T/NT较131I-PAMAM(G5.0)-GP明显升高,差异有统计学意义。以上结果提示131I-PAMAM(G5.0)-SR对MTC模型具有很好的靶向性及滞留性。
我们还发现,荷瘤鼠注射131I-PAMAM(G5.0)、131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP后,探针在体内主要分布在肿瘤、肾脏、脾脏、肝脏、胃、甲状腺及全血等器官或组织。这是因为PAMAM(G5.0)-SR、PAMAM(G5.0)-GP和PAMAM(G5.0)-SR/GP的纳米粒径较小,能有效过滤排泄到尿液中,主要通过肾脏排泄[21],其次肝、脾组织含有大量巨噬细胞,纳米粒子可经网状内皮系统吞噬清除[22-23],同时少部分游离的Na131I可被甲状腺、全血及胃吸收。而Na131I注射后主要聚集于甲状腺、全血及胃。以上结果进一步揭示了本研究以PAMAM(5.0)为载体合成分子探针的可行性,以及靶向肽SR、GP及SR/GP对MTC模型的靶向性。另外,含有双靶向肽的131I-PAMAM(G5.0)-SR/GP在MTC模型中的摄取高于对照组而低于含单靶向肽的131I-PAMAM(G5.0)-SR及131I-PAMAM(G5.0)-GP,这可能与探针中GP和SR的含量有关,药物装载的深入研究是下一步研究的方向。
综上所述,靶向肽SR和GP提高了131I-PAMAMM(G5.0)对MTC的靶向性,131I-PAMAM(G5.0)-GP在MTC中的摄取及洗脱较快,有望应用于MTC的显像诊断,而131I-PAMAM(G5.0)-SR在MTC中具有更好的细胞靶向性和滞留性,有望应用于MTC的靶向诊治及预后评估。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 冯成涛负责实验的实施、数据的收集及整理、论文的撰写;张海波、郑浩、王梓延、邓赟负责实验的实施;赵继华负责指导探针的合成及理化性质的鉴定;朱高红负责实验的设计、方法的建立、项目的支持及论文的审阅。
131I-PAMAM(G5.0)介导靶向肽在甲状腺髓样癌模型中的实验研究
Efficiacy of 131I-generation 5.0 polyamidamine-mediated targeting peptide in the mice with medullary thyroid carcinoma
-
摘要:
目的 评价新型分子靶向探针131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP及131I-PAMAM(G5.0)-SR/GP[其中,PAMAM(G5.0):第五代聚酰胺-胺;SR:丝氨酸-精氨酸-谷氨酸-丝氨酸-脯氨酸-组氨酸-脯氨酸(SRESPHP,简称SR);GP:甘氨酸-脯氨酸-亮氨酸-脯氨酸-亮氨酸-精氨酸(GPLPLR,简称GP)]在荷瘤甲状腺髓样癌(MTC)模型中的靶向性。 方法 采用高效液相色谱法纯化和分析靶向肽SR、GP及SR/GP,分别将其与修饰后的PAMAM(G5.0)共价连接,合成前体药物PAMAM(G5.0)-SR、PAMAM(G5.0)-GP及PAMAM(G5.0)-SR/GP。采用动态光散射粒度分析法检测PAMAM(G5.0)键合靶向肽前后的纳米粒径及Zeta电位。采用氯胺T法对修饰后的PAMAM(G5.0)、PAMAM(G5.0)-SR、PAMAM(G5.0)-GP、PAMAM(G5.0)-SR/GP进行131I标记,合成阳性对照组[131I-PAMAM(G5.0)]及实验组[131I-PAMAM(G5.0)-SR、131I-PAMAM(G5.0)-GP、131I-PAMAM(G5.0)-SR/GP]的4种探针。通过薄层色谱法分别测定探针的标记率、放射化学纯度及稳定性。经模型鼠腹腔注射阴性对照组(Na131I)、阳性对照组及实验组探针后,分别于4、8、12、24及48 h进行SPECT/CT显像,并计算靶/非靶值(T/NT),同时处死裸鼠取肿瘤及重要脏器测定放射性,以每克组织百分注射剂量率(%ID/g)表示。组间同一时间点T/NT、组内不同时间点T/NT及组间24 h肿瘤放射性摄取值的比较采用单因素方差分析,两两比较采用LSD-t检验。 结果 纯化后的靶向肽SR、GP和SR/GP的纯度均达99%。PAMAM(G5.0)键合SR、GP、SR/GP前后的纳米粒径分别为4.47、5.70、4.71、5.95 nm,Zeta电位分别为+37.95、+20.02、+28.34、+24.37 mV。131I标记4种探针的标记率均>75%,放射化学纯度均>90%,48 h的体外稳定性显示,放射化学纯度均在85%以上。SPECT/CT图像显示,实验组探针在不同时间的T/NT均较对照组有增高趋势:131I-PAMAM(G5.0)-GP 在4 h的T/NT(6.03±1.45)与阳性对照组(2.18±0.39)、阴性对照组(1.36±0.00)比较,且差异均有统计学意义(t=3.235,P=0.033;t=3.843,P=0.019);而131I-PAMAM(G5.0)-SR在8、12、24 h的T/NT(5.12±1.65、4.82±0.09、3.41±1.01)均较阴性对照组(1.50±0.00、1.43±0.65、1.34±0.81)高,且差异均有统计学意义(t=4.004,P=0.017;t=3.388,P=0.027;t=4.180,P=0.009)。实验组组内比较,131I-PAMAM(G5.0)-SR在24 h的T/NT(3.41±1.01)较131I-PAMAM(G5.0)-GP(2.10±0.67)高,差异有统计学意义(t=3.990,P=0.016)。注射131I-PAMAM(G5.0)-SR的肿瘤放射性摄取在24 h[(1.80±0.18)%ID/g]均较实验组其他探针、阴性对照组及阳性对照组高,但差异无统计学意义(F=3.366,P=0.059)。T/NT及肿瘤放射性摄取的达峰时间:131I-PAMAM(G5.0)-GP和131I-PAMAM(G5.0)-SR/GP 为4 h,131I-PAMAM(G5.0)和131I-PAMAM(G5.0)-SR为8 h。131I-PAMAM(G5.0)-GP的放射性洗脱较快,12 h的T/NT较4 h下降约57%。 结论 SR及GP提高了131I-PAMAMM(G5.0)对MTC的靶向性,131I-PAMAM(G5.0)-GP靶向MTC肿瘤新生血管使其在瘤体的摄取及代谢较快,有望应用于MTC SPECT显像,而131I-PAMAM(G5.0)-SR对MTC细胞具有很好的靶向性和滞留性,有望应用于MTC的靶向诊治及预后评估。 -
关键词:
- 甲状腺肿瘤 /
- 癌,髓样 /
- 分子探针 /
- 第五代聚酰胺-胺 /
- 丝氨酸-精氨酸-谷氨酸-丝氨酸-脯氨酸-组氨酸-脯氨酸 /
- 甘氨酸-脯氨酸-亮氨酸-脯氨酸-亮氨酸-精氨酸 /
- 碘放射性同位素
Abstract:Objective To synthesize the novel targeting probes 131I-generation 5.0 polyamidamine (PAMAM(G5.0))-SR (where SR: Ser-Arg-Glu-Ser-Pro-His-Pro), 131I-PAMAM(G5.0)-GP (where GP: Gly-Pro-Leu-Pro-Leu-Arg), and 131I-PAMAM(G5.0)-SR/GP (double targeting peptides) as experimental groups and evaluate their target capability toward medullary thyroid carcinoma (MTC). Methods The target peptides SR, GP, and SR/GP were purified and analyzed by using high-performance liquid chromatography and then covalently linked with the modified PAMAM(G5.0) to synthesize PAMAM(G5.0) peptides [PAMAM(G5.0)-SR, PAMAM(G5.0)-GP, and PAMAM(G5.0)-SR/GP]. The diameter and zeta potential of PAMAM(G5.0) alone and PAMAM(G5.0) peptides were detected by using dynamic light scattering. The modified PAMAM(G5.0) and PAMAM(G5.0) peptides were labeled with radioisotope 131I using the chloramine T method. The radiolabeling rate, radiochemistry purity, and stability were determined by using thin-layer chromatography. SPECT/CT imaging was performed in all groups at 4, 8, 12, 24, and 48 h after the probes were injected into the peritoneal cavity of the model mice. The ratio of target to non-target (T/NT) was then detected. The percentage injection dose per gram (%ID/g) with the tumor and important organs of the model mice were calculated. One-way analysis was used to compare the T/NT in different groups at the same time, T/NT in same group at the different time, %ID/g at 24 h in different groups. LSD-t test was used to compare date between two groups. Results The purity of purified PAMAM(G5.0)-SR, -GP, and -SR/GP were at 99%. The diameter of PAMAM(G5.0), PAMAM(G5.0)-SR, PAMAM(G5.0)-GP, PAMAM(G5.0)-SR/GP were 4.47, 5.70, 4.71, 5.95 nm, and zeta potentials were +37.95, +20.02, +28.34, +24.37 mV respectively. The radiolabeling rates of the four types of 131I probes were above 75%. The radiochemistry purities of the purified probes were more than 90% and remained over 85% after 48 h incubation at room temperature. All the T/NT of the experimental groups were higher than those of the control groups. The T/NT significantly increased in 131I-PAMAM(G5.0)-GP (6.03±1.45) at 4 h (t=3.235, P=0.033; t=3.843, P=0.019) compared with those in the positive and negative control groups (2.18±0.39 and 1.36±0.00, respectively). The T/NT significantly increased in 131I-PAMAM(G5.0)-SR (5.12±1.65, 4.82±0.09, and 3.41±1.01) at 8, 12, and 24 h (t=4.004, P=0.017; t=3.388, P=0.027; t=4.180, P=0.009, respectively) compared with that in the negative control group (1.50±0.00, 1.43±0.65, and 1.34±0.81). The T/NT of 131I-PAMAM(G5.0)-SR (3.41±1.01) was significantly higher than that of 131I-PAMAM(G5.0)-GP (2.10±0.67) at 24 h post-injection (t=3.990, P=0.016). The tumor %ID/g in 131I-PAMAM(G5.0)-SR (1.80±0.18) was higher than that in other groups at 24 h, but no significant differences were observed (F=3.366, P=0.059). The peak values of the T/NT and ID%/g of the tumor were observed at 4 h in the 131I-PAMAM(G5.0)-GP and 131I-PAMAM(G5.0)-SR/GP groups and at 8 h in the 131I-PAMAM(G5.0) and 131I-PAMAM(G5.0)-SR groups. The T/NT value decreased to 57% from 4 h to 12 h in 131I-PAMAM(G5.0)-GP group. Conclusions The SR and GP peptides enhanced the targetability of 131I-PAMAMM (G5.0) on MTC cells and neovascularization. The 131I-PAMAM(G5.0)-GP probe may be suitable for diagnosis because of its rapid ingestion and excretion than other probes in the model mice. The 131I-PAMAM(G5.0)-SR probe may provide a new precision method for MTC treatment and follow-up because of its better targetability and longer residence time than other probes in the model mice. -
表 1 131I标记探针注射后24 h在荷MTC裸鼠体内的生物学分布[(M(P25,P75),
$\bar{x}\pm s$ Table 1. %ID/g of the tumor and important organs at 24 h post-injection of probes labeled with radioisotope 131I [(M(P25, P75),
$\bar{x}\pm s$ 器官或组织 131I- PAMAM(G5.0) 131I- PAMAM(G5.0)-SR 131I- PAMAM(G5.0)-GP 131I- PAMAM(G5.0)-SR/GP Na131I 肿瘤 1.45±0.28 1.80±0.18 1.58±0.14 1.13±0.33 0.57±0.25 肝 0.50±0.41 0.89±0.75 1.11±1.62 0.39±0.52 0.28±0.15 脾 2.02±1.83 2.28±2.98 0.21±0.17 0.61±0.54 0.28±0.03 胰 0.70±0.65 0.76±0.62 0.30±0.26 0.19±0.21 0.07±0.02 肾 3.02±1.50 8.08±4.63 3.41±3.18 2.46±1.82 0.33±0.02 肠 0.59±0.49 2.35±2.21 0.47±0.70 0.29±0.35 0.23±0.00 全血 0.51±0.49 2.64(0.01,4.40) 0.53±0.40 1.51±1.67 5.93±2.10 甲状腺 4.02±5.31 1.41±1.88 0.47±0.31 0.69(0.58,6.67) 832.00±159.73 胃 0.92±0.64 5.11±4.48 0.52±0.45 0.32±0.25 1.39±0.26 注:表中,MTC:甲状腺髓样癌;PAMAM(G5.0):第五代聚酰胺-胺;SR:丝氨酸-精氨酸-谷氨酸-丝氨酸-脯氨酸-组氨酸-脯氨酸(SRESPHP);GP:甘氨酸-脯氨酸-亮氨酸-脯氨酸-亮氨酸-精氨酸(GPLPLR);%ID/g:每克组织百分注射剂量率。 -
[1] Yang H, Lopina ST, DiPersio LP, et al. Stealth dendrimers for drug delivery: correlation between PEGylation, cytocompatibility, and drug payload[J]. J Mater Sci Mater Med, 2008, 19(5): 1991−1997. DOI: 10.1007/s10856−007−3278−0. [2] He XD, Alves CS, Oliveira N, et al. RGD peptide-modified multifunctional dendrimer platform for drug encapsulation and targeted inhibition of cancer cells[J]. Colloids Surf B Biointerfaces, 2015, 125: 82−89. DOI: 10.1016/j.colsurfb.2014.11.004. [3] Zeng Y, Kurokawa Y, Win-Shwe TT, et al. Effects of PAMAM dendrimers with various surface functional groups and multiple generations on cytotoxicity and neuronal differentiation using human neural progenitor cells[J]. J Toxicol Sci, 2016, 41(3): 351−370. DOI: 10.2131/jts.41.351. [4] Asimakopoulos P, Nixon IJ, Shaha AR. Differentiated and Medullary Thyroid Cancer: Surgical Management of Cervical Lymph Nodes[J]. Clin Oncol (R Coll Radiol), 2017, 29(5): 283−289. DOI: 10.1016/j.clon.2017.01.001. [5] Trimboli P, Castellana M, Virili C, et al. Efficacy of Vandetanib in Treating Locally Advanced or Metastatic Medullary Thyroid Carcinoma According to RECIST Criteria: A Systematic Review and Meta-Analysis[J/OL]. Front Endocrinol (Lausanne), 2018, 9: 224 [2018-11-30]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5943569. DOI: 10.3389/fendo.2018.00224. [6] Cappagli V, Potes CS, Ferreira LB, et al. Calcitonin receptor expression in medullary thyroid carcinoma[J/OL]. PeerJ, 2017, 5: e3778[2018-11-30]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5600720. DOI: 10.7717/peerj.3778. [7] Valderrabano P, Klippenstein DL, Tourtelot JB, et al. New American Thyroid Association Sonographic Patterns for Thyroid Nodules Perform Well in Medullary Thyroid Carcinoma: Institutional Experience, Systematic Review, and Meta-Analysis[J]. Thyroid, 2016, 26(8): 1093−1100. DOI: 10.1089/thy.2016.0196. [8] Wells SA Jr, Asa SL, Dralle H, et al. Revised American Thyroid Association Guidelines for the Management of Medullary Thyroid Carcinoma[J]. Thyroid, 2015, 25(6): 567−610. DOI: 10.1089/thy.2014.0335. [9] Trimboli P, Treglia G, Guidobaldi L, et al. Detection rate of FNA cytology in medullary thyroid carcinoma: a meta-analysis[J]. Clin Endocrinol (Oxf), 2015, 82(2): 280−285. DOI: 10.1111/cen.12563. [10] Chauhan AS, Jain NK, Diwan PV. Pre-clinical and behavioural toxicity profile of PAMAM dendrimers in mice[J]. Proc Roy Soc A, 2009, 466(2117): 1535−1550. DOI: 10.1098/rspa.2009.0448. [11] Nabavizadeh F, Fanaei H, Imani A, et al. Evaluation of Nanocarrier Targeted Drug Delivery of Capecitabine-PAMAM Dendrimer Complex in a Mice Colorectal Cancer Model[J]. Acta Med Iran, 2016, 54(8): 485−493. [12] Vidal F, Vásquez P, Cayumán FR, et al. Prevention of Synaptic Alterations and Neurotoxic Effects of PAMAM Dendrimers by Surface Functionalization[J/OL]. Nanomaterials (Basel), 2017, 8(1): 7[2018-11-30]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5791094. DOI: 10.3390/nano8010007. [13] Tomalia DA, Baker H, Dewald J, et al. A New Class of Polymers: Starburst-Dendritic Macromolecules[J]. Polym J, 1985, 17(1): 117−132. DOI: 10.1295/polymj.17.117. [14] Kondo M, Asai T, Katanasaka Y, et al. Anti-neovascular therapy by liposomal drug targeted to membrane type-1 matrix metalloproteinase[J]. Int J Cancer, 2004, 108(2): 301−306. DOI: 10.1002/ijc.11526. [15] Genís L, Gálvez BG, Gonzalo P, et al. MT1-MMP: Universal or particular player in angiogenesis?[J]. Cancer Metastasis Rev, 2006, 25(1): 77−86. DOI: 10.1007/s10555−006−7891−z. [16] Davaa E, Lee J, Jenjob R, et al. MT1-MMP Responsive Doxorubicin Conjugated Poly (lactic-co-glycolic Acid) /Poly (styrene-alt-maleic Anhydride) Core/Shell Microparticles for Intrahepatic Arterial Chemotherapy of Hepatic Cancer[J]. ACS Appl Mater Interfaces, 2017, 9(1): 71−79. DOI: 10.1021/acsami.6b08994. [17] He R, Wang H, Su Y, et al. Incorporating 131I into a PAMAM (G5.0) dendrimer-conjugate: design of a theranostic nanosensor for medullary thyroid carcinoma[J]. RSC Adv, 2017, 7(26): 16181−16188. DOI: 10.1039/C7RA00604G. [18] Böckmann M, Hilken G, Schmidt A, et al. Novel Sresphp Peptide Mediates Specific Binding to Primary Medullary Thyroid Carcinoma After Systemic Injection[J]. Hum Gene Ther, 2005, 16(11): 1267−1275. DOI: 10.1089/hum.2005.16.1267. [19] Schmidt A, Eipel C, Fürst K, et al. Evaluation of systemic targeting of RET oncogene-based MTC with tumor-selective peptide-tagged Ad vectors in clinical mouse models[J]. Gene Ther, 2011, 18(4): 418−423. DOI: 10.1038/gt.2010.165. [20] 陈礼林, 谢丽君, 张海波, 等. 靶向肽结合131I-PAMAM (G5.0)抑制甲状腺髓样癌细胞增殖的研究[J]. 国际放射医学核医学杂志, 2017, 41(5): 307−313. DOI: 10.3760/cma.j.issn.1673−4114.2017.05.001.
Chen LL, Xie LJ, Zhang HB, et al. Effects of targeted peptide-conjugated 131I-PAMAM (G5.0) on the inhibition of medullary thyroid carcinoma cells proliferation[J]. Int J Radiat Med Nucl Med, 2017, 41(5): 307−313. DOI: 10.3760/cma.j.issn.1673−4114.2017.05.001.[21] Matsuura S, Katsumi H, Suzuki H, et al. L-Serine-modified polyamidoamine dendrimer as a highly potent renal targeting drug carrier[J]. Proc Natl Acad Sci USA, 2018, 115(41): 10511−10516. DOI: 10.1073/pnas.1808168115. [22] Cui W, Zhang YQ, Xu XP, et al. Synthesis and 188Re Radiolabelling of Dendrimer Polyamide Amine (PAMAM) Folic Acid Conjugate[J]. Med Chem, 2012, 8(4): 727−731. DOI: 10.2174/157340612801216256. [23] 王立, 薛冰, 张文君, 等. 聚酰胺-胺型树枝状大分子的应用研究[J]. 哈尔滨商业大学学报: 自然科学版, 2018, 34(1): 24−26, 48. DOI: 10.3969/j.issn.1672−0946.2018.01.007.
Wang L, Xue B, Zhang WJ, et al. Advances in application of polyamide-amine dendrimers[J]. J Harbin Univ Commer: Nat Sci Ed, 2018, 34(1): 24−26, 48. DOI: 10.3969/j.issn.1672−0946.2018.01.007. -





 下载:
下载: