-
胃癌是最常见的消化道恶性肿瘤,病死率位居肿瘤相关致死疾病的第2位[1]。目前胃癌的主要治疗手段是根治性胃切除联合淋巴结清扫,但术后5年生存率低于30%[2],术后生存率较低的主要原因之一是术后复发[3]。因此,对于胃癌术后复发的监测尤为重要,早期发现并合理调整治疗方案能够有效改善预后。18F-FDG PET/CT能够检测组织生理代谢功能,同时应用CT技术对核素分布情况进行解剖定位,被广泛应用于胃癌术后复发的监测。笔者回顾性分析18F-FDG PET/CT在胃癌术后复发监测中的临床价值。
-
回顾性分析2013年1月至2017年8月于本院核医学科及放射科行18F-FDG PET/CT和增强CT检查的52例胃癌根治术后患者,其中男性40例、女性12例,年龄30~86(61.73±11.44)岁。纳入标准:①有上腹部疼痛不适、进食后饱胀,食欲下降,乏力等临床症状;②消化道相关肿瘤标志物有(癌胚抗原、CA199、CA724)一项及一项以上水平升高;③行胃癌根治术;④行PET/CT及增强CT检查且检查间隔≤2周。排除标准:①胃癌根治术后至行PET/CT、增强CT检查间隔时间<3个月;②PET/CT与增强CT检查间隔>2周;③除胃癌外患有其他恶性肿瘤的患者;④未签署或不愿签署知情同意书的患者。52例患者中,中-高分化腺癌3例、中分化腺癌11例、中-低分化腺癌12例、低分化腺癌20例、黏液腺癌4例、神经内分泌癌2例。术后至PET/CT、增强CT检查间隔为3~48个月,中位时间为12个月,复发患者中20例经病理证实,其余经临床随访病情进展时间≥6个月,确认复发。所有患者均于检查前签署了知情同意书。
-
18F-FDG由南京安迪科正电子研究发展有限公司提供,放射化学纯度>95%。影像设备采用美国GE公司Discovery TM STE16 PET/CT显像仪。显像前患者空腹6 h以上(空腹血糖≤6.9 mmol/L),于静息状态下以3.7~4.4 MBq/kg(0.10~0.12 mCi/kg)静脉注射18F-FDG,休息45 min后显像,扫描范围为颅顶至股骨上端。CT采集电压为140 kV,电流为120 mA,层厚3.75 mm。最后行PET全身显像,每个床位采集2 min,共采集7~8个床位。采集数据完成后PET与CT均采用有序子集迭代法进行图像重建,重建图像层厚为3.27 mm,通过CT数据对PET图像进行衰减校正,最后进行图像融合。
-
检查前患者禁食6 h。采用德国Siemens公司Somatom Sensation 64层CT扫描仪行横断面扫描,扫描范围自膈顶至盆腔。管电压120 kV,管电流250 mA,扫描层厚5 mm。CT增强扫描采用高压注射器团注对比剂碘海醇(含碘300 mg/mL),流速为3.0 mL/s,注射剂量为1.5 mL/kg,注射总量为70~80 mL。动脉期和门静脉期扫描延迟时间分别为27 s和50 s。
-
定性分析:吻合口胃壁代谢增高,放射性分布高于正常肝脏和(或)吻合口胃壁增厚为18F-FDG PET/CT阳性。半定量分析:勾画吻合口部位的最大层面ROI,工作站计算吻合口SUV,取其最大值(SUVmax)。使用受试者工作特征(receiver operating characteristic, ROC)曲线分析吻合口SUVmax临界值,SUVmax≥临界值为阳性,SUVmax<临界值为阴性。增强CT:吻合口胃壁增厚伴明显强化(CT值>20 HU)为CT阳性。
-
18F-FDG PET/CT阳性:淋巴结代谢高于正常肝脏和(或)淋巴结短径≥1.0 cm。增强CT阳性:淋巴结短径≥1.0 cm,静脉期明显强化(CT值>20 HU)或淋巴结有低密度坏死表现。
-
18F-FDG PET/CT阳性:全身其他部位高代谢灶(放射性分布高于其生理性分布)和(或)全身其他部位见肿块、密度改变。增强CT阳性:全身其他部位肿块、密度改变伴异常强化。
-
由2名副高级职称的核医学科医师独立阅片,通过分析影像学资料获得吻合口、淋巴结及远处转移灶的SUVmax、CT增强特点等,分析结果不一致时通过讨论得出一致意见。同一患者全身多发转移时分别入组,即1例患者同时存在吻合口复发、淋巴结及远处转移时,记为吻合口复发1例、淋巴结转移1例、远处转移1例。
-
采用 SPSS 22.0软件对数据进行统计学分析。符合正态分布的计量资料以均数±标准差(
$\overline x $ ±s)表示,组间比较采用独立样本t检验;采用χ2检验比较诊断效能。P<0.05表示差异有统计学意义。 -
52例胃癌术后疑似复发患者中,39例证实为复发或转移。其中,21例吻合口复发,25例腹盆腔淋巴结转移,32例腹盆腔脏器转移。
定性分析:18F-FDG PET/CT显像中,吻合口阳性25例、阴性27例,其中真阳性20例、假阴性1例、真阴性26例、假阳性5例,定性分析诊断吻合口复发的灵敏度为95.24%(20/21)、特异度为83.87%(26/31)。
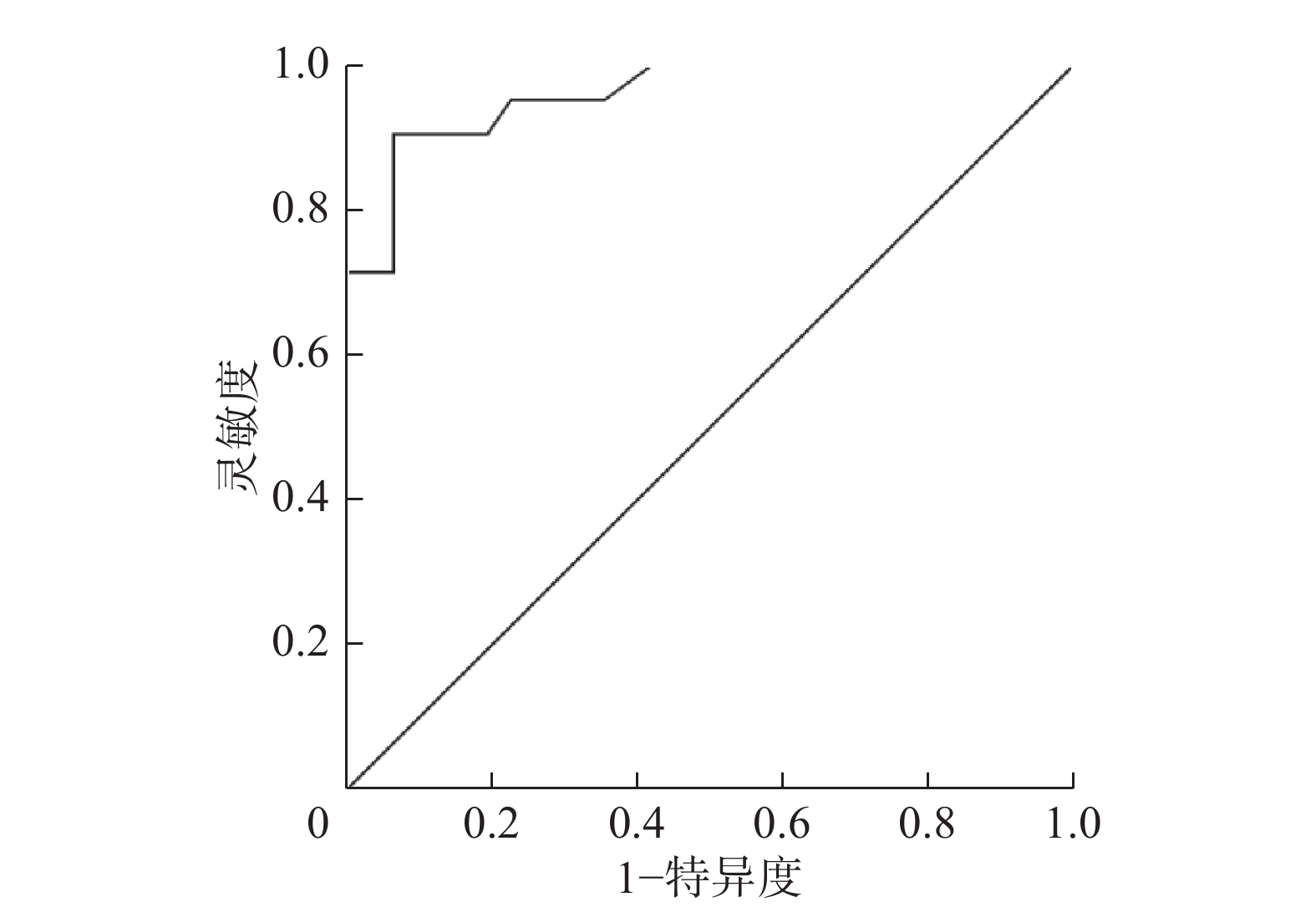
半定量分析:通过ROC曲线分析发现SUVmax=3.14时,18F-FDG PET/CT对于吻合口复发的诊断效能最高(曲线下面积=0.942,标准误=0.039,P<0.05)(图1)。因此本研究中18F-FDG PET/CT诊断吻合口复发的半定量分析标准确定为SUVmax≥3.14为阳性、SUVmax<3.14为阴性,在18F-FDG PET/CT显像中,吻合口阳性19例、阴性33例,其中真阳性19例、假阴性2例、真阴性31例、假阳性0例,半定量分析诊断吻合口复发的灵敏度为90.48%(19/21)、特异度为100%(31/31)。
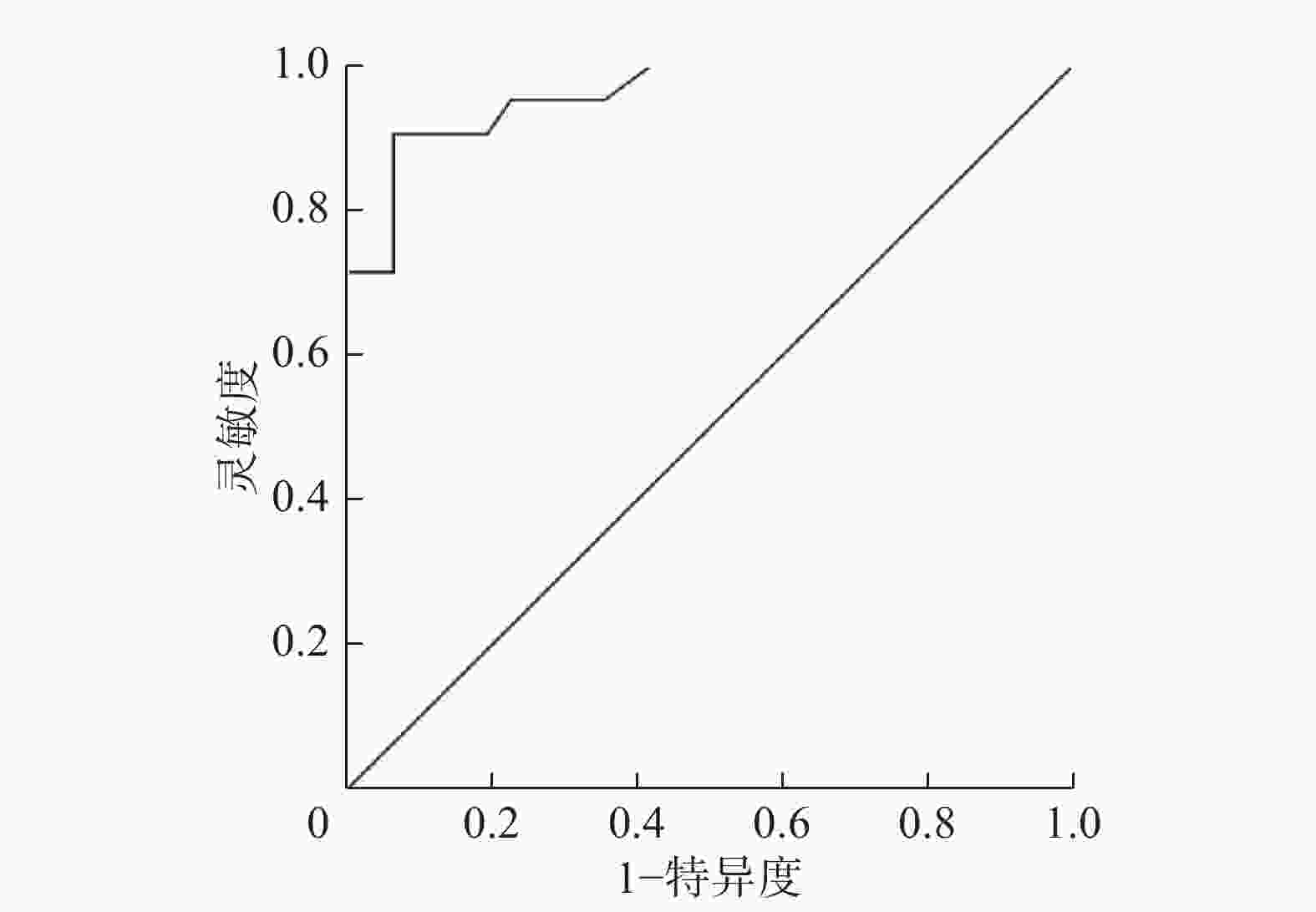
图 1 行胃癌根治术患者的吻合口复发SUVmax的ROC曲线 图中,SUVmax:最大标准化摄取值;ROC:受试者工作特征。
Figure 1. Receiver operating characteristic curve of SUVmax in anastomotic recurrence of patients undergoing radical gastrectomy
18F-FDG PET/CT半定量分析诊断吻合口复发的特异度高于定性分析,差异有统计学意义(χ2=5.439,P<0.05),两者灵敏度的差异无统计学意义(χ2=0.365,P>0.05)。因此,在与增强CT的对比分析中,吻合口复发的阳性标准采用半定量分析(SUVmax临界值为3.14)。
-
52例患者中吻合口复发21例、吻合口未复发31例(其中吻合口炎12例,其余19例经胃镜或随访证实未复发且无炎性改变)。吻合口复发的SUVmax(7.03±3.67)高于吻合口炎(1.74±1.63),差异有统计学意义(t=4.701,P<0.05)。
-
增强CT对吻合口复发诊断的灵敏度为23.81%(5/21)、特异度为100%(31/31),18F-FDG PET/CT诊断吻合口复发的灵敏度高于增强CT,且差异有统计学意义(χ2=19.369,P<0.05),两者特异度均为100%,差异无统计学意义(χ2=0.000,P>0.05)。
增强CT诊断吻合口复发的假阴性16例,均诊断为术后改变,其中,14例18F-FDG PET/CT见吻合口异常摄取,经病理证实为复发(典型病例图像见图2);增强CT诊断真阳性5例、真阴性31例、假阳性0例。18F-FDG PET/CT诊断假阴性2例,增强CT诊断为阴性,经病理证实为吻合口复发。5例患者增强CT和18F-FDG PET/CT诊断均为阳性,经病理或随访证实复发。
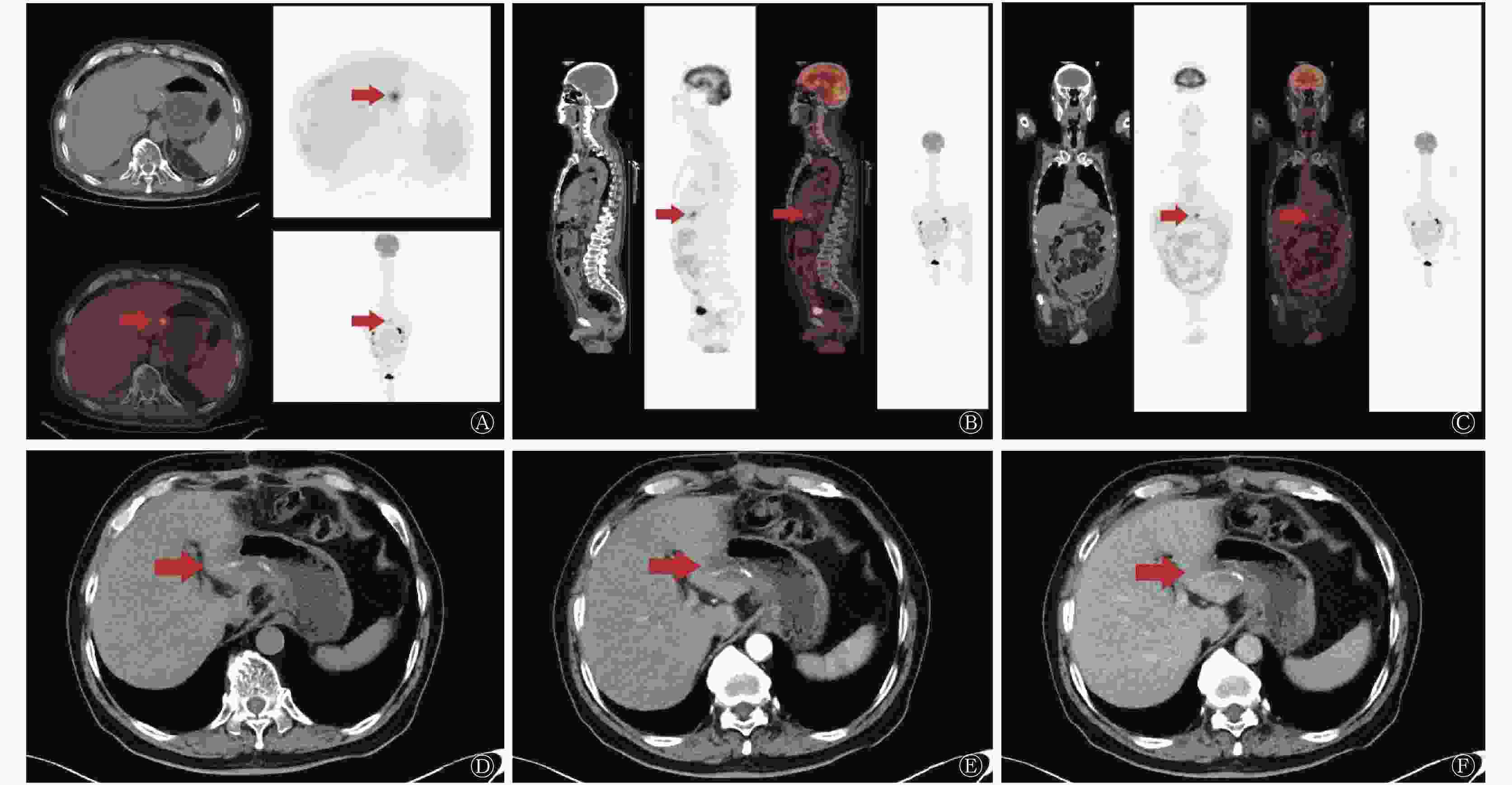
图 2 行胃癌根治术患者(男性,71岁,中分化腺癌)的18F-FDG PET/CT与增强CT的影像图 患者术后行常规辅助化疗,术后7个月行18F-FDG PET/CT和增强CT检查。图中,A、B、C:PET/CT横断位、矢状位、冠状位可见吻合口胃壁放射性摄取增高,SUVmax=6.75;D、E、F:CT平扫吻合口未见明显异常,增强CT动脉期、静脉期未见明显异常强化。患者最终经组织病理学证实为吻合口复发。箭头示复发部位。FDG:氟脱氧葡萄糖;PET:正电子发射断层显像术;CT:计算机体层摄影术;SUVmax;最大标准化摄取值。
Figure 2. 18F-FDG PET/CT and contrast-enhanced CT images of patient undergoing radical gastrectomy (male, 71 years old, moderately differentiated adenocarcinoma)
-
增强CT诊断假阳性2例,18F-FDG PET/CT未见异常摄取,经随访证实为淋巴结炎;增强CT诊断假阴性10例,其中8例18F-FDG PET/CT见淋巴结异常摄取(3例经术后病理证实为淋巴结转移,5例经随访证实为转移);增强CT诊断真阳性15例、真阴性25例。18F-FDG PET/CT诊断假阴性2例,增强CT未见明显强化,经随访证实为淋巴结转移(典型病例图像见图3),18F-FDG PET/CT诊断真阳性23例、真阴性27例、假阳性0例。
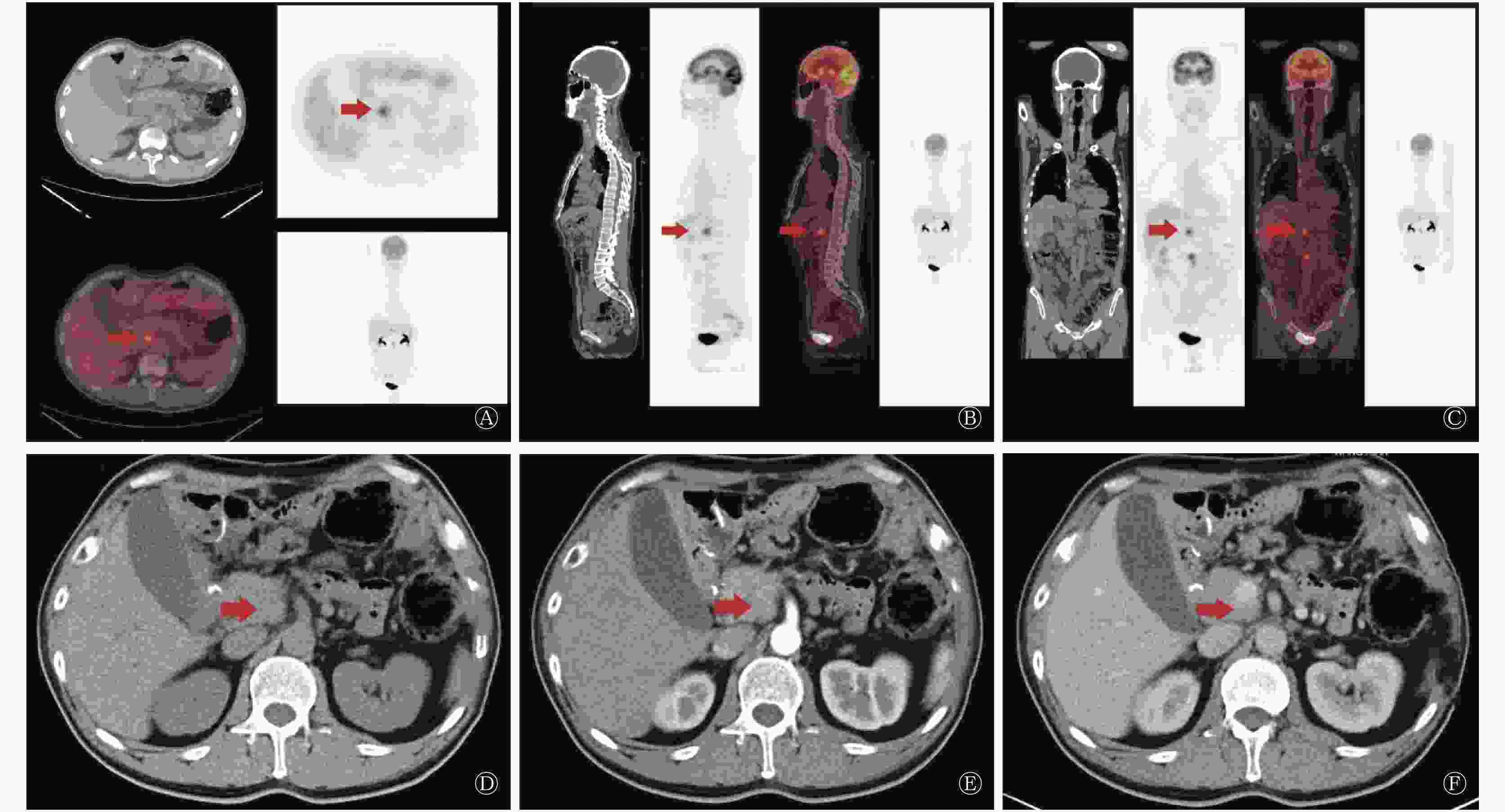
图 3 行胃癌根治术患者(男性,56岁,中-低分化腺癌)的18F-FDG PET/CT与增强CT的影像图 患者术后行常规辅助化疗,术后7个月行PET/CT和增强CT检查。图中,A、B、C:PET/CT横断位、矢状位、冠状位上均可见后腹膜淋巴结放射性摄取增高,SUVmax=6.7;D、E、F:CT平扫后腹膜淋巴结直径<1 cm,增强CT动脉期、静脉期未见明显异常强化。患者最终经组织病理学证实为淋巴结转移。箭头示转移部位。FDG:氟脱氧葡萄糖;PET:正电子发射断层显像术;CT:计算机体层摄影术;SUVmax;最大标准化摄取值。
Figure 3. 18F-FDG PET/CT and contrast-enhanced CT images of patient undergoing radical gastrectomy (male, 56 years old, moderately-poorly differentiated adenocarcinoma)
18F-FDG PET/CT诊断腹盆腔淋巴结转移的灵敏度为92.00%(23/25)、特异度为100%(27/27),增强CT诊断腹盆腔淋巴结转移的灵敏度为60.00%(15/25)、特异度为92.59%(25/27),18F-FDG PET/CT对腹盆腔淋巴结转移诊断的灵敏度高于CT,且差异有统计学意义(χ2=5.373,P<0.05),两者特异度的差异无统计学意义(χ2=2.850,P>0.05)。
-
增强CT诊断远处转移假阳性1例,为肠道局部肠壁增厚,18F-FDG PET/CT未见异常摄取,后行肠镜未见明显异常;增强CT诊断假阴性21例,其中17例18F-FDG PET/CT见腹盆腔脏器(肝、胰腺、脾脏、肾脏、膀胱、子宫及附件等)异常摄取,经病理和随访证实为转移(典型病例图像见图4);增强CT诊断真阳性11例、真阴性19例。18F-FDG PET/CT假阴性4例(3例腹水可见癌细胞、1例术后病理证实为肝转移),18F-FDG PET/CT诊断真阳性28例、真阴性20例、假阳性0例。

图 4 行胃癌根治术患者(男性,61岁,中分化腺癌)的18F-FDG PET/CT与增强CT的影像图 患者术后行常规辅助化疗,术后7个月行PET/CT和增强CT检查。A、B、C:PET/CT横断位、矢状位、冠状位上均可见直肠前方结节放射性摄取增高,SUVmax=5.03;D、E、F:CT平扫见直肠前方组织结节状稍增厚,增强CT动脉期、静脉期未见明显异常强化。患者最终经组织病理学证实为种植转移。箭头示转移部位。FDG:氟脱氧葡萄糖;PET:正电子发射断层显像术;CT:计算机体层摄影术;SUVmax;最大标准化摄取值。
Figure 4. 18F-FDG PET/CT and contrast-enhanced CT images of patient undergoing radical gastrectomy (male, 61 years old, moderately differentiated adenocarcinoma)
18F-FDG PET/CT诊断腹盆腔脏器转移的灵敏度为87.50%(28/32)、特异度为100%(20/20),增强CT诊断腹盆腔脏器转移的灵敏度为34.38%(11/32)、特异度为95.00%(19/20),18F-FDG PET/CT对远处复发转移诊断的灵敏度高于CT,且差异有统计学意义(χ2=16.804,P<0.05),两者特异度的差异无统计学意义(χ 2=1.412,P>0.05)。
-
胃癌根治术后,28%~47%的患者可能复发,术后的辅助化疗在一定程度上降低了复发率,但一旦复发往往预示着不良的生存和预后[4-5]。因此对胃癌术后复发的监测是患者管理的重要组成部分,及早发现复发并给予适当的治疗手段进行干预对于提高患者的生活质量具有重要意义。Meta分析报道18F-FDG PET/CT诊断胃癌复发的整体灵敏度和特异度分别为78%~86%、82%~88%,虽然总体的诊断效能较高,但仍存在漏诊和误诊的可能[6-7]。18F-FDG PET/CT诊断胃癌复发假阳性的主要原因可能是:术后的吻合口炎症反应使吻合口的18F-FDG摄取偏高,难以与吻合口复发鉴别。本研究发现吻合口复发的SUVmax明显高于吻合口炎,且差异有统计学意义,与张敬勉等[8]研究结果相似,通过ROC曲线分析得出SUVmax临界值取3.14时,18F-FDG PET/CT对吻合口复发的诊断效能更高,本研究界定18F-FDG PET/CT SUVmax良恶性的临界值为3.14,与常规以SUVmax=2.5作为临界值不同,主要优势在于减少了因吻合口炎症产生的假阳性。本研究中,采用定性分析作为吻合口阳性标准时,假阳性5例(胃镜证实均为吻合口炎),半定量分析假阳性0例。综上,吻合口复发的18F-FDG摄取明显高于吻合口炎,有助于PET/CT对吻合口复发的监测,且吻合口SUVmax临界值取3.14时,减少了假阳性,诊断效能更高,避免了不必要的检查和治疗。
检测胃癌术后复发的常用方法主要有肿瘤标志物、内镜、CT等,血清肿瘤标志物容易受多种因素的影响,灵敏度和特异度不高,且不能准确定位病灶,只能作为参考[9]。内镜下组织病理学活检对吻合口或残胃复发诊断的价值较高,但由于其为侵入性腔内检查,其在淋巴结及远处转移的作用受到局限。经济、简便的CT最为常用,但术后腹腔内解剖结构的改变使CT对胃癌术后复发的诊断价值受限。Wu和Zhu[10]研究发现,胃癌术后的形态学改变与胃癌复发转移难以鉴别。在本研究中,增强CT诊断假阴性16例,均误诊为胃部术后改变,后经胃镜证实为吻合口复发。
传统的影像学检查手段已不能满足临床医师的需求,因此集CT解剖显像与PET代谢显像于一体的非侵入性检查——18F-FDG PET/CT逐渐被广泛应用,其能更全面地评估全身情况,包括寻找吻合口复发、淋巴结及远处转移灶,在评估治疗后反应以及术后复发的检测方面也具有重要的临床价值。Ozkan等[11]报道18F-FDG PET/CT较CT能够检出更多的复发病灶,18F-FDG PET/CT的诊断灵敏度、特异度、准确率、阳性预测值、阴性预测值均优于CT[12],与本研究结果相似。其他研究结果也表明18F-FDG PET/CT诊断胃癌复发的灵敏度及特异度较高,分别达到84.2%~95.9%、79.5%~100%,表明18F-FDG PET/CT在胃癌术后复发的监测中具有重要的临床价值[13-14]。此外,有研究报道18F-FDG PET/CT诊断胃癌腹膜转移的灵敏度较低,原因可能为:影像学上腹膜转移性病变多数表现为腹膜增厚或小结节病变,使18F-FDG PET/CT对腹膜转移病灶的检测有一定的局限性,有待进一步研究完善[15]。
另一方面,本研究仍然存在些许不足,如样本量较少引起的抽样误差;部分胃癌患者手术至术后18F-FDG PET/CT检查间隔较短,产生了假阳性显像,这些均有待长期随访或病理学活检进一步证实。
综上,吻合口复发的18F-FDG摄取明显高于吻合口炎,有助于18F-FDG PET/CT对吻合口复发的监测;18F-FDG PET/CT显像对于胃癌术后复发转移的诊断效能优于增强CT,有较高的灵敏度和特异度,对胃癌术后复发检测有重要的临床价值。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 徐鑫负责研究命题的提出、方法的建立以及论文的撰写;邓胜明负责论文的审阅;李继会、黄莹负责数据的获取、分析;张玮负责最终版本的修订。
18F-FDG PET/CT在胃癌复发及转移监测中的临床价值
The value of 18F-FDG PET/CT in the detection of gastric cancer recurrence and metastasis
-
摘要:
目的 探讨18F-FDG PET/CT在胃癌术后复发及转移监测中的临床价值。 方法 回顾性分析2013年1月至2017年8月行18F-FDG PET/CT及增强CT检查的52例胃癌术后疑似复发转移患者[男性40例,女性12例,年龄30~86(61.73±11.44)岁]。通过定量分析与半定量分析的比较,确定吻合口复发的最大标准化摄取值(SUVmax)的临界值。采用ROC曲线分析吻合口的SUVmax临界值,t检验比较组间SUVmax,χ2检验比较定性分析与半定量分析及18F-FDG PET/CT与增强CT对于胃癌术后吻合口、淋巴结、远处转移的诊断效能。 结果 吻合口SUVmax临界值取3.14时,18F-FDG PET/CT诊断吻合口复发的特异度高于定性分析法,差异有统计学意义(χ2=5.439,P<0.05)。吻合口复发的SUVmax明显高于吻合口炎,差异有统计学意义(t=4.701,P<0.05)。18F-FDG PET/CT与增强CT诊断吻合口复发、淋巴结、远处转移的灵敏度的比较,差异有统计学意义(90.48% vs. 23.81%、92.00% vs. 60.00%、87.50% vs. 34.38%,χ2=19.363、5.373、16.804,均 P<0.05);特异度的比较均无统计学意义(100% vs. 100% 、 100% vs. 92.5% 、 100% vs. 95.00% , χ2 = 0.000、2.850、1.412 , 均 P>0.05)。 结论 18F-FDG PET/CT诊断胃癌术后复发转移优于CT,对胃癌术后复发转移的监测有重要临床价值。 Abstract:Objective To investigate the value of 18F-FDG PET/CT in the detection of gastric cancer recurrence and metastasis. Methods A retrospective analysis involving 52 patients with suspected recurrence and metastasis after gastric cancer surgery and who underwent 18F-FDG PET/CT and contrast-enhanced CT examinations over the period of January 2013 to August 2017 was performed, including 40 males and 12 females, aged between 30 and 86 years old(61.73±11.44). Receiver operating characteristic analysis was conducted on the basis of imaging data for the detection of a cutoff SUVmax value that is indicative of the presence of recurrent lesions. Differences among groups were determined through the t-test. The results of different methods were compared through the χ2 test. Results The specificity of 18F-FDG PET/CT was favorable and was higher than that of the qualitative analytical method(χ2=5.439, P<0.05) when a cutoff of 3.14 was used to evaluate for anastomosis. The SUVmax of anastomotic recurrence was higher than that of anastomotic inflammation(t=4.701, P<0.05). The sensitivity of 18F-FDG PET/CT in diagnosing recurrence, lymph node metastasis and distant metastasis was higher than that of contrast-enhanced CT, the difference was statistically significant(90.48% vs.23.81%, 92.00% vs.60.00%, 87.50% vs.34.38%, χ2=19.369, 5.373, 16.804, all P<0.05). The specificity of 18F-FDG PET/CT and contrast-enhanced CT in diagnosing recurrence, lymph node metastasis and distant metastasis was not statistically significant(100% vs.100%, 100% vs.92.5%, 100% vs.95.00%, χ2=0.000, 2.850, 1.412, all P>0.05). Conclusions The performance of PET/CT in the diagnosis of gastric cancer recurrence and metastasis is better than that of contrast-enhanced CT. Therefore, 18F-FDG PET/CT imaging has a high clinical value in the postoperative detection of gastric cancer. -
图 2 行胃癌根治术患者(男性,71岁,中分化腺癌)的18F-FDG PET/CT与增强CT的影像图 患者术后行常规辅助化疗,术后7个月行18F-FDG PET/CT和增强CT检查。图中,A、B、C:PET/CT横断位、矢状位、冠状位可见吻合口胃壁放射性摄取增高,SUVmax=6.75;D、E、F:CT平扫吻合口未见明显异常,增强CT动脉期、静脉期未见明显异常强化。患者最终经组织病理学证实为吻合口复发。箭头示复发部位。FDG:氟脱氧葡萄糖;PET:正电子发射断层显像术;CT:计算机体层摄影术;SUVmax;最大标准化摄取值。
Figure 2. 18F-FDG PET/CT and contrast-enhanced CT images of patient undergoing radical gastrectomy (male, 71 years old, moderately differentiated adenocarcinoma)
图 3 行胃癌根治术患者(男性,56岁,中-低分化腺癌)的18F-FDG PET/CT与增强CT的影像图 患者术后行常规辅助化疗,术后7个月行PET/CT和增强CT检查。图中,A、B、C:PET/CT横断位、矢状位、冠状位上均可见后腹膜淋巴结放射性摄取增高,SUVmax=6.7;D、E、F:CT平扫后腹膜淋巴结直径<1 cm,增强CT动脉期、静脉期未见明显异常强化。患者最终经组织病理学证实为淋巴结转移。箭头示转移部位。FDG:氟脱氧葡萄糖;PET:正电子发射断层显像术;CT:计算机体层摄影术;SUVmax;最大标准化摄取值。
Figure 3. 18F-FDG PET/CT and contrast-enhanced CT images of patient undergoing radical gastrectomy (male, 56 years old, moderately-poorly differentiated adenocarcinoma)
图 4 行胃癌根治术患者(男性,61岁,中分化腺癌)的18F-FDG PET/CT与增强CT的影像图 患者术后行常规辅助化疗,术后7个月行PET/CT和增强CT检查。A、B、C:PET/CT横断位、矢状位、冠状位上均可见直肠前方结节放射性摄取增高,SUVmax=5.03;D、E、F:CT平扫见直肠前方组织结节状稍增厚,增强CT动脉期、静脉期未见明显异常强化。患者最终经组织病理学证实为种植转移。箭头示转移部位。FDG:氟脱氧葡萄糖;PET:正电子发射断层显像术;CT:计算机体层摄影术;SUVmax;最大标准化摄取值。
Figure 4. 18F-FDG PET/CT and contrast-enhanced CT images of patient undergoing radical gastrectomy (male, 61 years old, moderately differentiated adenocarcinoma)
-
[1] Torre LA, Bray F, Siegel RL, et al. Global cancer statistics, 2012[J]. CA Cancer J Clin, 2015, 65(2): 87−108. DOI: 10.3322/caac.21262. [2] Lu J, Wang W, Zheng C, et al. Influence of total lymph node count on staging and survival after gastrectomy for gastric cancer: an analysis from a two-institution database in China[J]. Ann Surg Oncol, 2017, 24(2): 486−493. DOI: 10.1245/s10434−016−5494−7. [3] Chen K, Xu XW, Mou YP, et al. Systematic review and meta-analysis of laparoscopic and open gastrectomy for advanced gastric cancer[J/OL]. World J Surg Oncol, 2013, 11(1): 182[2018−07−03]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3750547/pdf/1477-7819-11-182.pdf. DOI: 10.1186/1477−7819−11−182. [4] Lee JH, Son SY, Lee CM, et al. Factors predicting peritoneal recurrence in advanced gastric cancer: implication for adjuvant intraperitoneal chemotherapy[J]. Gastric Cancer, 2014, 17(3): 529−536. DOI: 10.1007/s10120−013−0306−2. [5] Kang WM, Meng QB, Yu JC, et al. Factors associated with early recurrence after curative surgery for gastric cancer[J]. World J Gastroenterol, 2015, 21(19): 5934−5940. DOI: 10.3748/wjg.v21.i19.5934. [6] Wu LM, Hu JN, Hua J, et al. 18F-fluorodeoxyglucose positron emission tomography to evaluate recurrent gastric cancer: a systematic review and meta-analysis[J]. J Gastroenterol Hepatol, 2012, 27(3): 472−480. DOI: 10.1111/j.1440−1746.2011.06919.x. [7] Zou H, Zhao Y. 18FDG PET-CT for detecting gastric cancer recurrence after surgical resection: A meta-analysis[J]. Surg Oncol, 2013, 22(3): 162−166. DOI: 10.1016/j.suronc.2013.05.001. [8] 张敬勉, 赵新明, 王建方, 等. 18F-FDG PET/CT显像对胃癌治疗后临床再分期及治疗决策的影响[J].中国全科医学, 2013,16(15): 1360-1365. DOI: 10.3969/j.issn.1007-9572.2013.04.088.
Zhang JM, Zhao XM, Wang JF, et al. Impact of 18F-FDG PET/CT on the Clinical Restaging and Therapeutic Regimens in Post-Treatment Patients with Gastric Carcinoma[J]. Chin Gen Pract, 2013, 16(15): 1360-1365. DOI: 10.3969/j.issn.1007-9572.2013.04.088.[9] Ajanj JA, Barthel JS, Bekaii-Saab T, et al. Gastric cancer[J]. J Natl Compr Canc Netw, 2010, 8(4): 378−409. DOI: 10.6004/jnccn.2010.0030. [10] Wu CX, Zhu ZH. Diagnosis and evaluation of gastric cancer by positron emission tomography[J]. World J Gastroenterol, 2014, 20(16): 4574−4585. DOI: 10.3748/wjg.v20.i16.4574. [11] Ozkan E, Araz M, Soydal C, et al. The role of 18F-FDG-PET/CT in the preoperative staging and posttherapy follow up of gastriccancer: Comparison with spiral CT[J]. World J Surg Oncol, 2011, 9(1): 75. DOI: 10.1186/1477−7819−9−75. [12] Bilici A, Ustaalioglu BB, Seker M, et al. The role of 18F-FDG PET/CT in the assessment of suspected recurrent gastric cancer after initial surgical resection: can the results of FDG PET/CT influence patients' treatment decision making?[J]. Eur J Nucl Med Mol Imaging, 2011, 38(1): 64−73. DOI: 10.1007/s00259−010−1611−1. [13] Lee JW, Lee SM, Lee MS, et al. Role of 18F-FDG PET/CT in the prediction of gastric cancer recurrence after curative surgical resection[J]. Eur J Nucl Med Mol Imaging, 2012, 39(9): 1425−1434. DOI: 10.1007/s00259−012−2164−2. [14] Sharma P, Singh H, Suman SK, et al. 18F-FDG PET-CT for detecting recurrent gastric adenocarcinoma: results from a Non-Oriental Asian population[J]. Nucl Med Commun, 2012, 33(9): 960−966. DOI: 10.1097/MNM.0b013e328355b694. [15] Kawanaka Y, Kitajima K, Fukushima K, et al. Added value of pretreatment 18F-FDG PET/CT for staging of advanced gastric cancer: Comparison with contrast-enhanced MDCT[J]. Eur J Radiol, 2016, 85(5): 989−995. DOI: 10.1016/j.ejrad.2016.03.003. -





 下载:
下载: