-
SUV是PET/CT中最为常用的半定量指标,已经被广泛用于肿瘤良恶性的鉴别诊断、疗效的评价及预后评估,但其受到的影响因素有很多,包括采集时间间隔、血糖浓度和胰岛素水平以及药物剂量[1]。其中18F-FDG药物的注射活度就关系到临床的半定量指标分析,用于计算SUV。本研究旨在研究残留活度的影响因素,从而实现药物分装的规范化操作。
-
选取2017年10月至12月在首都医科大学附属北京世纪坛医院核医学科行PET/CT检查的患者100例。注射18F-FDG剂量按5.55 MBq/kg计算,按患者所需注射的放射性活度分为296、333、370、407、444 MBq 5组,误差不超过7.4 MBq;再按注射药物的体积分为0.6、0.9、1.2、1.5 mL的4组,共20组,每组患者不少于3例。
-
18F-FDG由中国原子高科公司生产,符合质量控制要求。2 mL型和5 mL型(山东威高集团有限公司)一次性使用无菌注射器。编号409511CN的医用三通(德国B.Braun Melsungen AG公司)。H-201型一次性使用静脉输液针(天津哈娜好医材有限公司)。CRC-25R放射性活度计(美国,CAPINTEC.INC公司),放射性活度计使用前经中国计量科学研究院鉴定合格。
-
采用头皮静脉针连接医用三通,医用三通再分别连接2 mL和5 mL注射器给患者注射药物,由同一名操作人员进行注射。2 mL注射器用于分装正电子药物,5 mL注射器装有5 mL灭菌注射用水,用于冲洗2 mL注射器及注射管道。使用无菌注射用水配准药物体积并记录,用放射性活度计记录注射前的药物活度,注射后将注射器针筒、注射器针头、三通及头皮静脉针分别放入一次性手套中,测量其残留的放射性活度以及空气中的放射性本底,测量过程中避免放射性污染,并统计总残留放射性活度。
-
采用SPSS 17.0软件对数据进行统计学分析。对注射药物的体积、放射性活度与相对应的总残留放射性活度进行正态分布检验及Spearman相关性分析,若数据为非正态分布,则使用中位数描述。r为相关系数,P<0.05表示差异有统计学意义。对注射药物浓度对总残留活度的影响进行曲线拟合。
-
注射后注射器针筒、注射器针头、三通及头皮静脉针残留的放射性活度减去空气中的放射性本底后的中位数(四分位距)分别为1.59(0.93)、3.35(3.43)、1.70(0.92)MBq,总残留放射性活度为7.09(4.31)MBq。总残留活度最小值为1.59 MBq,最大值为20.68 MBq。注射18F-FDG的100例患者的注射材料不同部位的放射性残留活度见表 1。
统计结果 残留活度/MBq 总的 针筒 针头针帽 三通及头皮静脉针 均值 7.54 1.71 4.04 1.80 中位数 7.09 1.59 3.35 1.70 方差 12.37 0.62 8.21 0.57 标准差 3.52 0.79 2.87 0.75 最小值 1.59 0.44 0.33 0.48 最大值 20.68 3.74 13.51 4.00 截距 19.09 3.30 13.18 3.52 四分位距 4.31 0.93 3.43 0.92 表 1 注射18F-FDG的100例患者的注射材料不同部位的残留放射性活度的统计值
Table 1. Radioactive residual activity statistics of 100 patients with 18F-FDG PET/CT examination
-
注射药物的活度与体积的比值为注射药物的放射性浓度。由表 2可见,18F-FDG注射药物的放射性活度与总残留放射性活度无相关性(r=0.099,P>0.05);18F-FDG注射药物的体积与总残留放射性活度呈负相关(r=-0.605,P<0.01),这表明18F-FDG注射药物的体积越大,总残留放射性活度越小;18F-FDG注射药物的放射性浓度与总残留放射性活度呈正相关(r=0.594,P<0.01),这表明总残留放射性活度随18F-FDG注射药物放射性浓度的增大而增大。
残留活度 相关性 注射药物活度 注射药物体积 注射药物浓度 总的 r=0.099,
P=0.328r=-0.605,
P=0.000r=0.594,
P=0.000针筒 r=0.027,
P=0.788r=-0.661,
P=0.000r=0.633,
P=0.000针头针帽 r=0.061,
P=0.550r=-0.312,
P=0.002r=0.298,
P=0.003三通及头皮经脉 r=0.279,
P=0.005r=-0.682,
P=0.000r=0.747,
P=0.000表 2 100例行18F-FDG PET/CT检查的患者注射18F-FDG材料的不同部位的残留放射性活度与相关参数的Spearman相关性分析
Table 2. Spearman correlation analysis of radioactivity residue activity and related parameters in 100 patients with 18F-FDG PET/CT examination
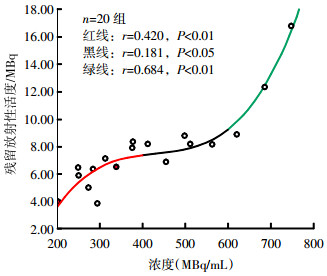
对20组数据取平均值,以注射药物放射性浓度为自变量,总残留放射性活度为因变量做曲线拟合(图 1)。根据图 1显示结果,把注射药物放射性浓度分为3组,分段进行相关性分析。当18F-FDG注射药物的放射性浓度>600 MBq/mL时,总残留放射性活度随18F-FDG注射药物放射性浓度的增大而增大(r=0.684,P < 0.01);当浓度 < 400 MBq/mL时,注射药物体积的增加和注射时间增长,增加了操作人员的受照时间(r=0.420,P < 0.01);当浓度介于400~600 MBq/mL之间时,总残留放射性活度与18F-FDG注射药物放射性浓度无相关性(r=0.181,P>0.05)。
-
SUV是PET在肿瘤诊断中常用的半定量指标,是指局部组织摄取显像剂的放射性活度与全身平均注射活度的比值。临床操作中,注射剂量是由注射前药物活度减去药物残留活度计算而得,但是药物注射后测量残留放射性活度会增加操作人员的受照剂量[2]。临床中采用头皮针连接注射器的方法进行静脉注射[3],药物残留的活度是由注射器的死腔以及注射通路中内壁的沾染而造成[4]。在注射过程中,注射器针筒、医用三通和头皮静脉针都由无菌注射用水冲洗,可使内壁沾染的放射性活度减少,但是从本研究中的标准差分析上也可以看出,注射器针筒、医用三通及头皮静脉针的残留放射性活度变化不大,主要影响总残留放射性活度的为注射器针头及针帽中的残留活度。
在本研究中,通过统计学分析发现,残留的放射性活度与注射药物的体积和放射性浓度相关。当药物的放射性浓度减少时,残留在注射器死腔中的药物浓度也减少,因为试验中采用同一规格的注射器,所以其死腔的体积相对固定[5-6],则残留的活度也相应减少。在临床操作中,增加注射药物体积和减少放射性浓度的方法主要是用无菌注射用水稀释。事先将分装用的2 mL注射器抽取无菌注射用水,再进行正电子药物的分装,可以增大注射药物的体积,减少注射药物的放射性浓度,从而有效减少残留的放射性活度。
18F-FDG为放射性显像剂,在临床操作中减少对操作人员的放射性照射十分重要。增加注射药物的体积使得注射药物时间增加,同样会增大工作人员受到的放射性照射[7],不符合放射性防护原则[8],因此应适当稀释注射药物的放射性浓度。从本研究结果来看,当药物放射性浓度在400~600 MBq/mL时,药物放射性浓度与总放射残留活度无相关性。在临床操作中,当放射性活度为400~600 MBq/mL时,注射活度更接近注射前所测量的药物活度,半定量分析测量的SUV才更加可靠。同时,注射后空针放射性活度降低也会减少对操作人员的受照剂量。
注射器规格对残留放射性活度有很大的影响,各单位医疗机构应根据本单位所用注射器规格测量适宜的注射药物稀释浓度,以指导临床正电子药物的分装。
18F-FDG注射残留放射性活度影响因素的分析
Analysis of factors affecting the residual radioactive activity of injection of 18F-FDG
-
摘要:
目的分析18F-FDG药物注射过程中影响其注射后残留放射性活度的因素,以指导临床放射性药物的分装。 方法收集2017年10月至12月在首都医科大学附属北京世纪坛医院核医学科行PET/CT检查的患者100例,按其注射药物的体积和放射性活度分组,用放射性活度计记录注射后注射器针筒、注射器针头、三通及头皮针残留的放射性活度以及空气中的放射性本底,并对总残留放射性活度和注射药物的放射性浓度进行曲线拟合。采用SPSS 17.0统计学软件进行正态分布检验及Spearman相关性分析。 结果注射后注射器针筒、注射器针头、三通及头皮静脉针残留的放射性活度的中位数(四分位距)分别为1.59(0.93)、3.35(3.43)、1.70(0.92)MBq,总残留放射性活度为7.09(4.31)MBq;总残留放射性活度与18F-FDG注射药物的放射性浓度呈正相关(r=0.594,P < 0.01),对其进行曲线拟合,放射性浓度 < 600 MBq/mL为宜。 结论对18F-FDG浓度进行稀释,降低其放射性浓度能减少总残留放射性活度,精确注射药物活度,同时能够降低对工作人员的受照剂量。 Abstract:ObjectiveThe factors influencing residual radioactivity in 18F-FDG injection were analyzed to guide the packing of radiopharmaceuticals. MethodsA total of 100 patients subjected to 18F-FDG PET/CT in Beijing Shijitan Hospital from October 2017 to December 2017 were analyzed. Patients were divided into different groups based on the volume and radioactivity of the radiopharmaceuticals they received. The residual radioactivity of the syringe tube, syringe needle, three-way valve, scalp needle, and background was then measured separately using a radioactivity meter. The curve of the total residual radioactivity as a function of 18F-FDG concentration was then fitted. SPSS 17.0 statistical software was used to test the normality of the data distribution, and results were analyzed by Spearman correlation. ResultsThe residual radioactivity values of the syringe needle tube, syringe needle, three-way valve, and scalp intravenous needle were 1.59(0.93), 3.35(3.43), and 1.70(0.92) MBq, respectively. The total residual radioactivity was 7.09(4.31 MBq) and significantly related to the concentration of 18F-FDG applied(r=0.594, P < 0.01). According to the fitting curve obtained, the appropriate radioactivity concentration of 18F-FDG is less than 600 MBq/mL. ConclusionsIn clinical work, appropriate dilution of the 18F-FDG injection volume and decreasing the radioactivity of pharmaceuticals can effectively reduce the total residual radioactivity of the procedure. Medical institutions should measure the appropriate dilution concentration of radioactive drugs according to the specifications of the syringes to increase the precision of injection and reduce the irradiation dose of the staff. -
Key words:
- Fluorodeoxyglucose F18 /
- Radioactivity /
- Drug residues
-
表 1 注射18F-FDG的100例患者的注射材料不同部位的残留放射性活度的统计值
Table 1. Radioactive residual activity statistics of 100 patients with 18F-FDG PET/CT examination
统计结果 残留活度/MBq 总的 针筒 针头针帽 三通及头皮静脉针 均值 7.54 1.71 4.04 1.80 中位数 7.09 1.59 3.35 1.70 方差 12.37 0.62 8.21 0.57 标准差 3.52 0.79 2.87 0.75 最小值 1.59 0.44 0.33 0.48 最大值 20.68 3.74 13.51 4.00 截距 19.09 3.30 13.18 3.52 四分位距 4.31 0.93 3.43 0.92 表 2 100例行18F-FDG PET/CT检查的患者注射18F-FDG材料的不同部位的残留放射性活度与相关参数的Spearman相关性分析
Table 2. Spearman correlation analysis of radioactivity residue activity and related parameters in 100 patients with 18F-FDG PET/CT examination
残留活度 相关性 注射药物活度 注射药物体积 注射药物浓度 总的 r=0.099,
P=0.328r=-0.605,
P=0.000r=0.594,
P=0.000针筒 r=0.027,
P=0.788r=-0.661,
P=0.000r=0.633,
P=0.000针头针帽 r=0.061,
P=0.550r=-0.312,
P=0.002r=0.298,
P=0.003三通及头皮经脉 r=0.279,
P=0.005r=-0.682,
P=0.000r=0.747,
P=0.000 -
[1] Adams MC, Turkington TG, Wilson JM, et al. A systematic review of the factors affecting accuracy of SUV measurements[J]. AJR Am J Roentgenol, 2010, 195(2):310-320. DOI:10.2214/AJR.10.4923. [2] 郑均正.电离辐射医学应用的防护与安全[M].北京:原子能出版社, 2009:284-285.
Zheng JZ. Protection and safety of medical applications of ionizing radiation[M]. Beijing:Atomic Energy Press, 2009:284-285.[3] 曹晓征.正电子发射断层显像检查中显像剂的注射方法比较[J].中国实用医药, 2013, 8(10):110-111. DOI:10.3969/j.issn.1673-7555.2013.10.078.
Cao XZ. Comparison of injection methods of imaging agents in positron emission tomography[J]. China Prac Med, 2013, 8(10):110-111. DOI:10.3969/j.issn.1673-7555.2013.10.078.[4] 李洁明, 刘晓华, 饶国辉, 等.不同容积注射器放射性残留量对患者核医学检查效果的影响及护理[J].现代临床护理, 2008, 7(10):16-17, 37. DOI:10.3969/j.issn.1671-8283.2008.10.006.
Li JM, Liu XH, Rao GH, et al. Effect of Radioactive Residues of Different Volume Syringes on Patients' Nuclear Medical Examination and Nursing Care[J]. Mod Clin Nurs, 2008, 7(10):16-17, 37. DOI:10.3969/j.issn.1671-8283.2008.10.006.[5] 刘慧萍, 武惠萍, 郭会萍.一次性小剂量注射器无效腔容量的探讨[J].全科护理, 2006, 4(15):28-29. DOI:10.3969/j.is]sn.1674-4748.2006.15.017.
Liu HP, Wu HP, Guo HP. Invalid cavity capacity of disposable small dose syringe[J]. Fam Nurs, 2006, 4(15):28-29. DOI:10.3969/j.issn.1674-4748.2006.15.017.[6] 谢欢, 林惠明, 陈英.从注射剂装量检测探讨注射器死腔残留量[J].护理学杂志, 2017, 32(19):69-70. DOI:10.3870/j.issn.1001-4152. 2017.19.069.
Xie H, Lin HM, Chen Y. Dead space volume of syringe:a perspective from medical agent injection volume[J]. J Nurs Sci, 2017, 32(19):69-70. DOI:10.3870/j.issn.1001-4152.2017.19.069.[7] 张巍, 李士雪, 李连波, 等.医院PET-CT中心18F正电子放射性药物生产及使用中工作人员的辐射剂量监测[J].山东大学学报(医学版), 2012, 50(12):126-129. DOI:10.6040/j.issn.1671-7554. 2012.12.026.
Zhang W, Li SX, Li LB, et al. Measurement and evaluation of personal radiation dose in cyclotron center and PET-CT department[J]. J Shandong Univ Health Sci, 2012, 50(12):126-129. DOI:10.6040/j.issn.1671-7554.2012.12.026.[8] 彭立华, 张锦艳.核医学医务人员的职业风险分析与放射防护措施[J].中国伤残医学, 2013, 21(6):384. DOI:10.3969/j.issn.1673-6567.2013.06.339.
Peng LH, Zhang JY. Occupational Risk Analysis and Radiation Protection Measures of Medical Staff in Nuclear Medicine[J]. Chinese Journal Trauma Disability Medicine, 2013, 21(6):384. DOI:10.3969/j.issn.1673-6567.2013.06.339. -






 下载:
下载: