-
在1920年最早由Krukenberg提出主动脉壁间血肿(aortic intramural hematoma,AIH),AIH定义为主动脉壁间发生的血肿,没有游离内膜片,无明显内膜撕裂或血流交通[1]。急性期主动脉壁间血肿(acute aortic intramural hematoma,AAIH)是指2周内发病并确诊的AIH。笔者回顾性分析AAIH的多层螺旋CT血管造影(multiple slice computed tomography angiography,MSCTA)影像学特征,探讨MSCTA的应用价值,为临床个性化治疗提供参考。
-
收集我院2009年5月至2016年10月收治的76例AAIH患者的MSCTA资料,其中男性53例、女性23例,年龄41~89岁,平均年龄(65.6±11.2)岁。76例患者中有高血压病史患者56例;临床表现为突发胸背部疼痛或上腹部疼痛患者69例,气促5例,意识障碍2例。所有患者均接受主动脉MSCTA检查。纳入标准:① 临床表现为急性胸痛并确诊为AAIH;② 发病时间为2周内;③ 均接受MSCTA检查。排除标准:① 主动脉夹层(aortic dissection,AD)和穿透性主动脉粥样硬化性溃疡;② 发病时间超过2周;③ MSCTA图像质量不能满足影像学诊断[2-3]。
-
所有患者均使用64排CT(Aquilion 64,日本东芝)行胸腹主动脉MSCTA检查。扫描参数:管电压120 kV,管电流300~400 mAs,扫描层厚0.5 mm,扫描野320 mm,重建层厚3 mm,层间距3 mm。先行CT平扫,扫描范围:胸廓入口水平至耻骨联合水平。MSCTA增强扫描保持与平扫相同的体位、扫描范围和方向。自动对比剂跟踪技术启动扫描,ROI置于降主动脉近段肺动脉分叉水平,ROI CT值阈值150 HU。采用双筒高压注射器经肘前静脉注射碘对比剂,剂量1.0~1.5 mL/kg,流速4.0~5.0 mL/s。注射对比剂后再以相同速度注射生理盐水40 mL。0.5 mm层厚的原始容积数据传至Vitrea工作站,通过多平面重建、曲面重建、最大密度投影及容积再现等技术进行图像后处理。本研究经我院伦理委员会批准。所有患者均签署了知情同意书。
-
图像由2名具备10年以上工作经验的胸部放射科医师共同阅读,并测量和记录相关参数、指标,最终意见由两人协商后决定。AAIH的MSCTA诊断标准:① 主动脉管壁环形或新月形增厚;② 厚度>7 mm或钙化内膜明显内移;③ 平扫增厚主动脉管壁内CT值>60 HU,增强扫描增厚的主动脉壁内没有对比剂显影[4]。
记录AAIH的分型:依据Stanford分型标准[4]分为A型和B型,A型累及升主动脉,B型未累及升主动脉。在MSCTA横断位图像上,测量AAIH累及主动脉最大管径及壁间血肿最大厚度。记录AAIH受累主动脉有无合并溃疡类病变及其深度、局灶性强化。测量指标均在横断位图像上完成。溃疡类病变指增强后AAIH内形成的龛影;局灶性强化定义为壁间血肿内局限性的对比剂充盈影,伴有与主动脉的小分支动脉连通。记录有无心包积液、胸腔积液等其他阳性发现。
-
按照病情发展将患者分为进展组、稳定和缓解组。进展组:壁间血肿厚度增加;进展为AD或主动脉瘤;主动脉破裂。稳定和缓解组:壁间血肿完全吸收或部分吸收;临床胸痛症状明显缓解;无其他并发症。随访方法采用查阅医院电子病案系统及图像存储和传输系统。随访时间为自AAIH发病后3个月。
-
采用SPSS 20.0软件进行统计学分析,符合正态分布的资料采用均数±标准差(x±s)表示,年龄、受累主动脉最大管径采用两独立样本t检验;不符合正态分布的定量资料(包括壁间血肿最大厚度、溃疡类病变深度)采用Mann-Whitney U检验;定性资料包括性别、溃疡类病变、局灶性强化、心包积液、胸腔积液,采用χ2检验。对比Stanford A型和B型在不同因素间的差异有无统计学意义。将疾病进展与否作为因变量,将性别、年龄、Stanford分型、主动脉管径、壁间血肿厚度、溃疡类病变、溃疡类病变深度、局灶性强化、心包积液、胸腔积液作为自变量,采用单因素检验分析有统计学意义的指标,再将有统计学意义的指标纳入多因素Logistic回归分析,建立回归模型,分析影响疾病进展的危险预测因素。以P < 0.05为差异有统计学意义。
-
76例AAIH患者中Stanford A型27例(35.5%)(图 1),其中男性18例、女性9例,平均年龄(64.0±12.6)岁;B型49例(64.5%)(图 2),男性35例、女性14例,平均年龄(66.5±10.3)岁。Stanford A型与B型AAIH患者在性别、年龄分布上的差异的均无统计学意义(χ2=0.187,P =0.665;t=-0.964,P =0.338)。

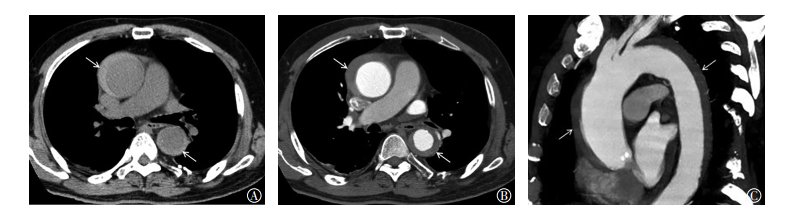
图 1 患者男性,72岁,急性期主动脉壁间血肿Stanford A型,MSCTA平扫横断位、增强横断位和矢状位最大密度投影图像图中,A:MSCTA平扫横断位显示升主动脉和降主动脉管径增粗,外围新月形稍高密度影(白色箭头所示);B:MSCTA增强横断位显示壁间血肿,内部无对比剂充盈(白色箭头所示);C:MSCTA增强矢状位最大密度投影重建直观显示壁间血肿累及范围(白色箭头所示)。MSCTA:多层螺旋CT血管造影。
Figure 1. Male, 72-year-old, Stanford A acute aortic intramural hematoma. Multiple slice computed tomography angiography plain axial image, enhancement axial image and maximum intensity projection image

图 2 患者男性,51岁,急性期主动脉壁间血肿Stanford B型,MSCTA平扫横断位和增强横断位图像图中,A:MSCTA平扫横断位显示降主动脉外围新月形稍高密度影(白色箭头所示);B:MSCTA增强横断位显示壁间血肿无对比剂充盈,升主动脉未受累(白色箭头所示)。MSCTA:多层螺旋CT血管造影。
Figure 2. Male, 51-year-old, Stanford B acute aortic intramural hematoma. Multiple slice computed tomography angiography plain axial image and enhancement axial image
-
76例患者的MSCTA均能清晰显示AAIH的累及范围和程度。CT平扫表现为主动脉管壁增厚,沿主动脉管壁纵向延伸,新月形增厚46例(60.5%),环形增厚30例(39.5%)。CT平扫壁间血肿CT值均>60 HU,内膜钙化影内移48例(63.2%)。MSCTA扫描示主动脉管腔均匀强化,壁内无假腔形成。AAIH溃疡类病变32例(42.1%),局限性强化11例(14.4%),合并心包积液15例(19.7%),胸腔积液39例(64.5%)。
Stanford A型与B型AAIH患者在壁间血肿厚度、合并溃疡类病变、局灶性强化、合并胸腔积液之间的差异均无统计学意义(P>0.05)。Stanford A型与B型AAIH患者在受累主动脉最大管径、溃疡类病变深度、合并心包积液之间的差异均有统计学意义(t=5.020、Z=-2.345、χ2=16.138,均P < 0.05)(表 1)。
Stanford分型
(例数)男/女
(例)平均年龄/岁 受累主动脉最大管径/mm 壁间血肿最大厚度/mm 溃疡类病变深度/mm 溃疡类病变/例 局灶性强化/例 心包积液/例 胸腔积液/例 A型(n=27) 18/9 64.0±12.6 45.1±9.1 10.6±3.8 2.8±2.9 10 3 12 12 B型(n=49) 35/14 66.5±10.3 35.7±4.7 10.4±3.2 2.5±3.3 22 8 3 27 检验值 χ2=0.187 t=-0.964 t=5.020 Z=-0.788 Z=-2.345 χ2=0.441 χ2=0.383 χ2=16.138 χ2=0.338 P值 0.665 0.338 < 0.001 0.431 0.019 0.507 0.536 < 0.001 0.561 注:表中,AAIH:急性期主动脉壁间血肿;MSCTA:多层螺旋CT血管造影。 表 1 Stanford A型与Stanford B型AAIH患者的临床特征和MSCTA影像学表现
Table 1. Clinical characteristics and multiple slice computed tomography angiography features between Stanford A and B acute aortic intramural hematoma
-
76例AAIH患者中,41例患者有短期随访。其中进展组20例,男性14例、女性6例,平均年龄(65.0±10.8)岁;稳定和缓解组21例,男性17例、女性4例,平均年龄(62.4±12.3)岁。进展组Stanford A型9例(进展为AD 4例;进展为主动脉瘤2例;死亡3例),B型11例[进展为AD 4例,进展为主动脉瘤3例,均接受主动脉腔内隔绝修补术(图 3);壁间血肿厚度增加4例]。稳定和缓解组Stanford A型5例、B型16例(图 4)。进展组受累主动脉最大管径和壁间血肿最大厚度分别为(39.6±9.7)mm和(10.7±3.0)mm;稳定组为(38.4±6.9)mm和(9.8±3.7)mm。进展组有12例合并溃疡类病变,平均溃疡深度为(3.9±4.0)mm;稳定和缓解组有3例合并溃疡类病变,平均溃疡深度为(1.3±3.5)mm。进展组及稳定和缓解组合并局灶性强化均为4例。进展组出现心包积液和胸腔积液分别为5例和13例,其中3例心包积液密度较高,提示为心包积血,短期随访死亡;稳定和缓解组分别为2例和7例。

图 3 患者男性,50岁,急性期主动脉壁间血肿Stanford B型,MSCTA增强横断位、矢状位和数字减影血管造影图像图中,A:MSCTA增强横断位显示降主动脉外围新月形稍高密度影,合并左侧胸腔积液和左肺下叶压迫性膨胀不全(白色箭头所示);B:MSCTA增强冠状位显示壁间血肿,内部多个溃疡类病变(白色箭头所示);C:两天后该患者接受主动脉腔内隔绝术。MSCTA:多层螺旋CT血管造影。
Figure 3. Male, 50-year-old, Stanford B acute aortic intramural hematoma. Multiple slice computed tomography angiography enhancement axial, saggital and digital subtraction angiography images

图 4 患者男性,45岁,急性期主动脉壁间血肿Stanford B型,首次检查和复查MSCTA增强横断位图像图中,A:MSCTA横断位显示降主动脉外围新月形稍高密度影,厚度约为8.7 mm(白色箭头所示);B:患者保守治疗3个月后复查,MSCTA横断位显示壁间血肿厚度明显缩小约为2.0 mm(白色箭头所示)。AAIH:急性主动脉壁间水肿;MSCTA:多层螺旋CT血管造影。
Figure 4. Male, 45-year-old, Stanford B acute aortic intramural hematoma. Multiple slice computed tomography angiography axial images in acute stage and follow up
-
采用单因素检验方法分析,结果显示进展组、稳定和缓解组在合并溃疡类病变、溃疡类病变深度、胸腔积液间差异均有统计学意义(χ2=9.227、Z=-2.689、χ2=4.111,均P < 0.05);进展组、稳定和缓解组在性别、年龄、Stanford分型、受累主动脉管径、壁间血肿厚度、局灶性强化、心包积液方面差异均无统计学意义(χ2=0.666、t=0.723、χ2=1.453、t= -0.439、Z=-0.874、χ2=0.006、χ2=1.733,均P > 0.05)(表 2)。
组别
(例数)性别
(男/女)平均年龄/岁 Stanford分型
(A型/B型)受累主动脉最大管径/mm 壁间血肿最大厚度/mm 溃疡类病变深度/mm 溃疡类病变/例 局灶性强化/例 心包积液/例 胸腔积液/例 进展组(n=20) 14/6 65.0±10.8 9/11 39.6±9.7 10.7±3.0 3.9±4.0 12 4 5 13 稳定和缓解组(n=21) 17/4 62.4±12.3 5/16 38.4±6.9 9.8±3.7 1.3±3.5 3 4 2 7 检验值 χ2=0.666 t=0.723 χ2=1.453 t=-0.439 Z=-0.874 Z=-2.689 χ2=9.227 χ2=0.006 χ2=1.733 χ2=4.111 P值 0.414 0.474 0.228 0.663 0.387 0.007 0.002 0.939 0.188 0.043 注:表中,AAIH:急性期主动脉壁间血肿;MSCTA:多层螺旋CT血管造影。 表 2 进展组与稳定和缓解组AAIH患者的临床特征和MSCTA影像表现
Table 2. Clinical characteristics and multiple slice computed tomography angiography features between progression and stable acute aortic intramural hematoma in 41patients
将单因素分析中具有统计学意义的指标(溃疡类病变、溃疡深度、胸腔积液)纳入多因素Logistic回归分析,以AAIH是否进展Y为因变量,以溃疡类病变、溃疡类病变深度、胸腔积液为自变量,分别以X1、X2、X3表示,采用逐步向前法进行逐步回归分析,对各因素进行进一步筛选,结果显示仅溃疡类病变纳入回归方程(95% CI:0.024~0.505,P =0.004),回归方程:
$ P{\rm{ = 1/}}\left\{ {{\rm{1 + exp}}\left[{-\left( {{\rm{0}}{\rm{.811-2}}{\rm{.197X1}}} \right)} \right]} \right\} $
式中,X1表示自变量溃疡类病变,说明溃疡类病变是AAIH疾病进展的独立危险预测因学。
-
急性主动脉综合征包括急性AD、AAIH和主动脉穿透性动脉粥样硬化性溃疡,其中AAIH占5%~20%。急性主动脉综合征临床表现和风险因素相似,包括急性胸痛和长期高血压,但是病理生理学机制不同。既往认为AIH为主动脉中层滋养血管的破裂,没有合并内膜撕裂,但是也有少数文献报道在影像上或术中发现小的内膜撕裂口[2-3]。AAIH在CT平扫上有特异性,表现为主动脉管壁新月形或环形高密度影,Knollmann等[5]将MSCTA平扫主动脉密度高于45 HU作为临界值鉴别急性主动脉综合征3种疾病,其诊断AIH的灵敏度和特异度分别为100%(19/19)和94%(16/17)。本研究中所有患者CT平扫壁间血肿区域CT值均>60 HU,其中新月形增厚46例(60.5%)、环形增厚30例(39.5%)。
AAIH按照Stanford分型方法分为A型和B型,与AD分型方法相同,但40% AIH表现为A型,而70%的AD表现为A型[6]。本研究中AAIH患者中Stanford A型占35.5%(27/76),与文献报道接近[6]。AAIH的Stanford A和B型平均发病年龄为(69.6±9.6)岁和(68.6±11.7)岁,高于AD的Stanford A型和B型平均发病年龄(61.4±14.6)岁和(62.9±14.3)岁,并且风险低于AD患者[7]。本研究AAIH男女发病率之间的差异无统计学意义,但男性发病率(53/76;69.7%)高于女性(23/76;30.3%),可能与雌性激素(特别是雌二醇)等女性内源性抗动脉粥样硬化的作用有关。
Park等[8]研究结果发现,Stanford A型AAIH受累主动脉管径≥48 mm预示不良进展,Stanford B型受累主动脉管径≥41 mm预示不良进展。本研究中,受累主动脉最大管径为26.6~63.5 mm,其中A型平均管径为(45.1±9.1)mm,Stanford B型平均管径为(35.7±4.7)mm。Song等[9]研究认为,AAIH受累主动脉的最大管径是疾病预后独立预测因子,主动脉管径小提示血肿可以完全吸收,主动脉管径大提示可能进展为AD或动脉瘤,所以常规测量受累主动脉管径非常重要。本研究中进展组受累主动脉最大管径(39.6 ±9.7)mm高于稳定和缓解组壁间血肿(38.4±6.9)mm。壁间血肿的厚度亦是影响AAIH预后的重要危险因素。壁间血肿径线越厚,并发症的风险越大。Choi等[10]的随访结果显示,壁间血肿厚度>11~16 mm,风险明显增加,而壁间血肿厚度<10~11mm预示并发症风险降低,血肿消失的可能性增加。本研究中AAIH厚度为7.0~18.5 mm,Stanford A型平均厚度为(10.6±3.8)mm,Stanford B型平均厚度为(10.4±3.2)mm。
AAIH如发生动脉内膜局限性破裂、真腔与壁间血肿狭颈状沟通, 并在血肿内形成点状或团状对比剂填充区,即为溃疡性病变,需要与穿透性主动脉粥样硬化性溃疡相鉴别[11]。穿透性主动脉粥样硬化性溃疡和主动脉粥样硬化密切相关,邻近主动脉内膜管壁粗糙,多伴有钙化或低密度脂质斑块。AAIH伴发溃疡性病变多提示预后不好,尤其是出现在升主动脉和主动脉弓部,会进展为主动脉夹层、动脉瘤或动脉破裂[12]。Kitai等[13]回顾分析38例AAIH,71%(27/38)患者有溃疡类病变,高于本研究的发生率42.1%,原因可能为其观察1.0 mm薄层CT图像,检测出更小的病变,而本研究观察的图像为3.0 mm层厚。在Kitai等[13]研究中,11例没有溃疡性病变的患者随访过程中壁间血肿均全部吸收,而溃疡类病变深度≥5 mm预测疾病进展的灵敏度、特异度、阳性预测值和阴性预测值分别为84%、95%、94%和86%。
我们对比进展组与稳定和缓解组多项影响因素发现,AAIH伴有溃疡性病变,以及溃疡性病变深度之间的差异具有统计学意义,Logistic回归分析进一步显示溃疡性病变是AAIH的独立危险预测因素,说明在AAIH的随访中溃疡类病变需要密切监测。Sueyoshi等[14]研究认为,溃疡类病变无论是初诊发现还是在随访中发现,都是预测并发症的危险因素,伴有复杂溃疡性病变的AAIH患者,在接受主动脉腔内隔绝手术后,壁间血肿的压力减轻,有利于受累主动脉管壁的重塑,预后较好。
AAIH发生时,主动脉小分支开口处内膜被壁间血肿掀起,血肿向外推挤主动脉外膜和肌层,使主动脉小分支开口处陷入血肿内,由于血流的压力而扩张形成局限性强化影。局限强化影与主动脉腔不直接相连,而与主动脉壁间的肋间动脉、支气管动脉、腰动脉相连,冠状位、矢状位重组图像可以更好地帮助显示其走行。Wu等[15]分析了65例AAIH患者,其中10例(15.4%)发生局灶性强化,主要发生在降主动脉和腹主动脉,随访过程中(平均36个月)大部分稳定或消失,结果得出局灶性强化与AAIH进展无明显相关。本研究中有11例(14.4%)患者发现局灶性强化,检出率与其接近,起源多来自肋间动脉开口区。在41例随访病例中,8例(19.5%)发现局灶性强化,其中进展组4例、稳定和缓解组4例,两者之间差异无统计学意义。Park等[8]研究认为,AAIH伴有局灶性强化没有增加风险,危险程度明显低于溃疡类病变,不需要急诊介入手术治疗。
AAIH发病年龄较大,高血压和动脉粥样硬化疾病患者发生率高,病变靠近外膜,容易导致急性液体外渗。本研究中AAIH患者中合并心包积液A型12例、B型3例,两者间差异有统计学意义,因为升主动脉起始部的壁间血肿更容易向下累及心包膜,引起心包的渗出、积血甚至心包填塞。Stanford A型进展组3例死亡病例,发现均伴有明显心包积液,其中2例密度较高提示为心包积血。Song等[16]对比分析60例Standord A型和127例B型AAIH,发现A型胸腔积液发生率(7%)明显多于B型(1%)。本研究结果发现,在AAIH进展组的胸腔积液发生率为65.0%(13/20),高于稳定和缓解组的33.3%(7/21),是影响AAIH是否进展的重要因素。
本研究尚具有一定的局限性:① 本研究为回顾性研究,难免存在选择性偏倚,AAIH患者的监测时间难以统一;② 病例样本量较小,有较完整随访记录的患者较少;③ 随访时间较短,我们只对AAIH的短期演变进行了对比分析。下一步研究我们会增加样本数量,并对患者进行长期的随访和监测。
综上所述,MSCTA检查可以对AAIH做出快速、准确诊断。溃疡性病变是影响AAIH进展的独立危险因素。AAIH早期需要密切随访,监测有无进展。
急性主动脉壁间血肿的多层螺旋CT血管造影影像学表现和诊断价值
Imaging findings and clinical value of multiple-slice computed tomography angiography in acute aortic intramural hematoma
-
摘要:
目的探讨急性主动脉壁间血肿(AAIH)的多层螺旋CT血管造影(MSCTA)影像学特征和临床应用价值。 方法回顾性分析临床确诊76例AAIH患者的主动脉MSCTA检查资料,依据Stanford分型分为A型和B型,比较不同Stanford分型AAIH的影像学表现有无差异。根据患者随访期间的不同变化分为进展组与稳定和缓解组,分析进展的相关因素。所有结果均行统计学分析,符合正态分布的定量资料采用两独立样本t检验;不符合正态分布的定量资料采用Mann-Whitney U检验。定性资料采用χ2检验。 结果Stanford A型27例(35.5%)、B型49例(64.5%)。A型、B型壁间血肿最大厚度、溃疡类病变、局灶性强化、胸腔积液上的差异均无统计学意义(Z=-0.788、χ2=0.441、χ2=0.383、χ2=0.338,均P > 0.05)。A型、B型溃疡类病变的深度分别为(2.8±2.9)mm、(2.5±3.3)mm,差异有统计学意义(Z=-2.345,P=0.019)。心包积液15例(19.7%),A型12例多于B型3例,差异有统计学意义(χ2=16.138,P < 0.001)。41例AAIH患者短期随访,进展组20例,稳定和缓解组21例。单因素分析显示,进展组与稳定和缓解组在溃疡类病变、溃疡深度、胸腔积液间的差异均具有统计学意义(χ2=9.227、Z=-2.689、χ2=4.111,均P < 0.05);在Stanford分型、壁间血肿厚度、局灶性强化、心包积液上的差异均无统计学意义(χ2=1.453、Z=-0.874、χ2=0.006、χ2=1.733,均P > 0.05)。将单因素分析中有统计学意义的指标纳入多因素Logistic回归分析,显示溃疡类病变是影响AAIH的独立危险预测因子。 结论AAIH在MSCTA上表现为平扫密度较高、增强无强化的环形或新月形增厚主动脉管壁。MSCTA检查可以对AAIH做出快速、准确诊断。溃疡性病变是影响AAIH进展的独立危险因素。AAIH早期需要密切随访,监测有无进展。 -
关键词:
- 主动脉 /
- 体层摄影术,X线计算机 /
- 血管造影术 /
- 壁间血肿
Abstract:ObjectiveTo explore the imaging findings in and clinical value of multiple-slice computed tomography angiography (MSCTA) in acute aortic intramural hematoma (AAIH). MethodsA total of 76 AAIH cases with complete clinical information were recruited and subjected to MSCTA examinations.The patients were divided into progressive and stable groups according to the follow-up changes.Then, the clinical data and MSCTA features were compared.All outcomes were analyzed statistically.The quantitative data of normal distribution and abnormal distribution were tested by two independent samples and Mann-Whitney U test, respectively.The qualitative data were tested by chi-square test. ResultsA total of 27(35.5%) type A cases and 49(64.5%) type B cases were classified according to Stanford criteria.No significant difference in the maximum thickness of aortic hematoma, number of ulcer-like projections, degree of focal enhancement, and severity of pleural effusion was noted between the Stanford A and B groups (Z=-0.788, χ2=0.441, χ2=0.383, χ2=0.338, all P > 0.05).The ulcer depths in the Stanford A and B groups were (2.8±2.9) and (2.5±3.3) mm, respectively (Z=-2.345, P=0.019).Meanwhile, 15(19.7%) cases (12 type A cases and 3 type B cases) were accompanied by pericardial effusion (χ2=16.138, P < 0.001).Twenty cases belonged to the progressive group, whereas 21 cases were included in the stable group.Univariate statistical analysis showed significant differences in ulcer-like projection, ulcer depth, and pleural effusion between the progressive and stable groups (χ2=9.227, Z=-2.689, χ2=4.111, all P < 0.05).By contrast, no significant difference in Stanford subtype, maximum aortic thickness, maximum aortic hematoma thickness, focal enhancement, and pleural effusion was noted between the Stanford A and B groups (χ2=1.453, Z=-0.874, χ2=0.006, χ2=1.733, all P > 0.05).Multivariate analysis revealed that the amount of ulcer-like projections was the independent risk factor for AAIH progression. ConclusionsAAIH manifested as annular and/or crescent thickening aortic wall with higher density in plain scan and non enhancement in enhanced scan using MSCTA technique.MSCTA plays an important role in the correct diagnosis of AAIH patients.The number of ulcer-like projections is the independent risk factor for disease progression.Close follow up is hence recommended during the early stage of AAIH using MSCTA. -
Key words:
- Aorta /
- Tomography, X-ray computed /
- Angiography /
- Intramural hematoma
-
图 1 患者男性,72岁,急性期主动脉壁间血肿Stanford A型,MSCTA平扫横断位、增强横断位和矢状位最大密度投影图像图中,A:MSCTA平扫横断位显示升主动脉和降主动脉管径增粗,外围新月形稍高密度影(白色箭头所示);B:MSCTA增强横断位显示壁间血肿,内部无对比剂充盈(白色箭头所示);C:MSCTA增强矢状位最大密度投影重建直观显示壁间血肿累及范围(白色箭头所示)。MSCTA:多层螺旋CT血管造影。
Figure 1. Male, 72-year-old, Stanford A acute aortic intramural hematoma. Multiple slice computed tomography angiography plain axial image, enhancement axial image and maximum intensity projection image
图 2 患者男性,51岁,急性期主动脉壁间血肿Stanford B型,MSCTA平扫横断位和增强横断位图像图中,A:MSCTA平扫横断位显示降主动脉外围新月形稍高密度影(白色箭头所示);B:MSCTA增强横断位显示壁间血肿无对比剂充盈,升主动脉未受累(白色箭头所示)。MSCTA:多层螺旋CT血管造影。
Figure 2. Male, 51-year-old, Stanford B acute aortic intramural hematoma. Multiple slice computed tomography angiography plain axial image and enhancement axial image
图 3 患者男性,50岁,急性期主动脉壁间血肿Stanford B型,MSCTA增强横断位、矢状位和数字减影血管造影图像图中,A:MSCTA增强横断位显示降主动脉外围新月形稍高密度影,合并左侧胸腔积液和左肺下叶压迫性膨胀不全(白色箭头所示);B:MSCTA增强冠状位显示壁间血肿,内部多个溃疡类病变(白色箭头所示);C:两天后该患者接受主动脉腔内隔绝术。MSCTA:多层螺旋CT血管造影。
Figure 3. Male, 50-year-old, Stanford B acute aortic intramural hematoma. Multiple slice computed tomography angiography enhancement axial, saggital and digital subtraction angiography images
图 4 患者男性,45岁,急性期主动脉壁间血肿Stanford B型,首次检查和复查MSCTA增强横断位图像图中,A:MSCTA横断位显示降主动脉外围新月形稍高密度影,厚度约为8.7 mm(白色箭头所示);B:患者保守治疗3个月后复查,MSCTA横断位显示壁间血肿厚度明显缩小约为2.0 mm(白色箭头所示)。AAIH:急性主动脉壁间水肿;MSCTA:多层螺旋CT血管造影。
Figure 4. Male, 45-year-old, Stanford B acute aortic intramural hematoma. Multiple slice computed tomography angiography axial images in acute stage and follow up
表 1 Stanford A型与Stanford B型AAIH患者的临床特征和MSCTA影像学表现
Table 1. Clinical characteristics and multiple slice computed tomography angiography features between Stanford A and B acute aortic intramural hematoma
Stanford分型
(例数)男/女
(例)平均年龄/岁 受累主动脉最大管径/mm 壁间血肿最大厚度/mm 溃疡类病变深度/mm 溃疡类病变/例 局灶性强化/例 心包积液/例 胸腔积液/例 A型(n=27) 18/9 64.0±12.6 45.1±9.1 10.6±3.8 2.8±2.9 10 3 12 12 B型(n=49) 35/14 66.5±10.3 35.7±4.7 10.4±3.2 2.5±3.3 22 8 3 27 检验值 χ2=0.187 t=-0.964 t=5.020 Z=-0.788 Z=-2.345 χ2=0.441 χ2=0.383 χ2=16.138 χ2=0.338 P值 0.665 0.338 < 0.001 0.431 0.019 0.507 0.536 < 0.001 0.561 注:表中,AAIH:急性期主动脉壁间血肿;MSCTA:多层螺旋CT血管造影。 表 2 进展组与稳定和缓解组AAIH患者的临床特征和MSCTA影像表现
Table 2. Clinical characteristics and multiple slice computed tomography angiography features between progression and stable acute aortic intramural hematoma in 41patients
组别
(例数)性别
(男/女)平均年龄/岁 Stanford分型
(A型/B型)受累主动脉最大管径/mm 壁间血肿最大厚度/mm 溃疡类病变深度/mm 溃疡类病变/例 局灶性强化/例 心包积液/例 胸腔积液/例 进展组(n=20) 14/6 65.0±10.8 9/11 39.6±9.7 10.7±3.0 3.9±4.0 12 4 5 13 稳定和缓解组(n=21) 17/4 62.4±12.3 5/16 38.4±6.9 9.8±3.7 1.3±3.5 3 4 2 7 检验值 χ2=0.666 t=0.723 χ2=1.453 t=-0.439 Z=-0.874 Z=-2.689 χ2=9.227 χ2=0.006 χ2=1.733 χ2=4.111 P值 0.414 0.474 0.228 0.663 0.387 0.007 0.002 0.939 0.188 0.043 注:表中,AAIH:急性期主动脉壁间血肿;MSCTA:多层螺旋CT血管造影。 -
[1] Nienaber CA, von Kodolitsch Y, Petersen B, et al. Intramural hemorrhage of the thoracic aorta. Diagnostic and therapeutic[J]. Circulation. 1995, 92(6):1465-1472. DOI:10.1161/01.CIR.92.6. 1465. [2] Erbel R, Aboyans V, Boileau C, et al. 2014 ESC guidelines on the diagnosis and treatment of aortic diseases[J]. Kardiol Pol, 2014, 72(12):1169-1252. DOI:10.5603/KP.2014.0225. [3] Gutschow SE, Walker CM, Mart-Nez-Jimenez S, et al. Emerging concepts in intramural hematoma imaging[J]. Radiographics, 2016, 36(3):660-674.DOI:10.1148/rg.2016150094. [4] Morris JH, Mix D, Cameron SJ. Acute aortic syndromes:update in current medical management[J]. Curr Treat Options Cardiovasc Med, 2017, 19(4):29. DOI:10.1007/s11936-017-0523-9. [5] Knollmann FD, Lacomis JM, Ocak I, et al. The role of aortic wall CT attenuation measurements for the diagnosis of acute aortic syndromes[J]. Eur J Radiol, 2013, 82(12):2392-2398. DOI:10.1016/j.ejrad.2013.09.007. [6] Mussa FF, Horton JD, Moridzadeh RA, et al. Acute aortic dissection and intramural hematoma a systematic review[J]. JAMA, 2016, 316(7):754-763. DOI:10.1001/jama.2016.10026. [7] Harris KM, Braverman AC, Eagle KA, et al. Acute aortic intramural hematoma:an analysis from the International Registry of Acute Aortic Dissection[J]. Circulation, 2012, 126(11 Suppl 1):S91-96.DOI:10.1161/CIRCULATIONAHA.111.084541. [8] Park GM, Ahn JM, Kim DH, et al. Distal aortic intramural hematoma:clinical importance of focal contrast enhancement on CT images[J].Radiology, 2011, 259(1):100-108. DOI:10.1148/radiol.11101557. [9] Song JK, Yim JH, Ahn JM, et al. Outcomes of patients with acute type a aortic intramural hematoma[J]. Circulation, 2009, 120(21):2046-2052. DOI:10.1161/CIRCULATIONAHA.109.879783. [10] Choi SH, Choi SJ, Kim JH, et al. Useful CT findings for predicting the progression of aortic intramural hematoma to overt aortic dissection[J]. J Comput Assist Tomogr, 2001, 25(2):295-299. doi: 10.1097/00004728-200103000-00025 [11] Lee YK, Seo JB, Jang YM, et al. Acute and chronic complications of aortic intramural hematoma on follow-up computed tomography:incidence and predictor analysis[J]. J Comput Assist Tomogr, 2007, 31(3):435-440. DOI:10.1097/01.rct.0000250112.87585.8e. [12] Schlatter T, Auriol J, Marcheix B, et al. Type B intramural hematoma of the aorta:evolution and prognostic value of intimal erosion[J]. J Vasc Interv Radiol, 2011, 22(4):533-541. DOI:10.1016/j.jvir.2010.10.028. [13] Kitai T, Kaji S, Yamamuro A, et al. Detection of intimal defect by 64-row multidetector computed tomography in patients with acute aortic intramural hematoma[J]. Circulation, 2011, 124(11 Suppl):S174-178. DOI:10.1161/CIRCULATIONAHA.111.037416. [14] Sueyoshi E, Nagayama H, Hashizume K, et al. Computed tomography evaluation of aortic remodeling after endovascular treatment for complicated ulcer-like projection in patients with type B aortic intramural hematoma[J]. J Vasc Surg, 2014, 59(3):693-699. DOI:10.1016/j.jvs.2013.08.100. [15] Wu MT, Wang YC, Huang YL, et al. Intramural blood pools accompanying aortic intramural hematoma:CT appearance and natural course[J]. Radiology, 2011, 258(3):705-713. DOI:10.1148/radiol.10101270. [16] Song JK, Kim HS, Song JM, et al. Outcomes of medically treated patients with aortic intramural hematoma[J]. Am J Med, 2002, 113(3):181-187. doi: 10.1016/S0002-9343(02)01171-3 -





 下载:
下载: