-
在我国乃至世界范围,乳腺癌都是女性发病率及病死率最高的恶性肿瘤之一。近年来我国乳腺癌发病率迅速上升,2000年我国女性的乳腺癌年龄标化发病率为19.9/10万,已经跃居我国女性恶性肿瘤的第一位[1]。对于复发转移性乳腺癌,目前认为是不可治愈性疾病,中位生存期仅2~3年,5年生存率仅15%~25%[2]。目前,乳腺癌治疗后复发和转移的概率高达20%~35%[3]。因此,尽早及准确地诊断出乳腺癌复发及转移,提供个体化的治疗方案以延长患者的生存期及提高生活质量,是非常有意义的。随着分子生物学技术的应用和医疗水平的不断提高,以肿瘤形态学结合基因表达特征的分子分型概念已被大家认同,对乳腺癌进行分型,有助于正确诊断乳腺癌并对治疗方法的选择和疾病的预后起指导作用。我们通过回顾性分析59例行18F-FDG PET/CT检查的女性乳腺癌术后患者,探讨18F-FDG PET/CT在乳腺癌术后复发转移以及治疗决策改变和预后中的价值。
-
回顾性分析2007年7月至2016年9月在本:行18F-FDG PET/CT检查的59例女性乳腺癌术后患者临床资料,年龄30~80岁(中位年龄50岁),平均年龄(50.17±9.91)岁。患者均行乳腺癌手术治疗(其中,行根治术55例、保乳术4例)。59例患者均为浸润性导管癌。术后至PET/CT检查时间间隔为9 d~128个月,中位时间为13个月。因部分复发转移部位取样活检有较大难度,PET/CT显像真阳性患者中仅4例经病理学检查确诊复发转移,其余经B超、CT、MRI、SPECT/CT、PET/CT等影像学检查,以及肿瘤标志物检查和临床随访确诊。PET/CT检查后随访时间为6~97个月,平均随访时间为(28.89±21.06)个月。本研究获得了我:伦理委员会批准。所有患者及其家属均于检查前签署了知情同意书。
-
采用美国GE公司Discovery STE16 PET/CT仪,18F-FDG由南京安迪科公司供应,放化纯度>95%。患者检查前空腹超过6 h,检查前血糖水平<8.0 mmol/L。静脉注射3.70~4.44 MBq/kg 18F-FDG,平静休息45~50 min。全身扫描范围从颅顶至大腿中部。CT扫描参数为120 kV、150 mA,层厚3.75 mm;PET扫描采用三维采集,每床位3 min,层厚3.27 mm。采集完成后应用CT数据对PET图像进行衰减校正,迭代法重建获得横断面、冠状面、矢状面影像及PET与CT的融合图像。
-
采用目测法和半定量分析法相结合的方式。目测法由2位具有PET诊断经验的核医学科、影像科副主任及以上医师共同阅片,诊断意见有分歧时,另请一位主任医师阅片,两位或以上医师意见一致时方可作出诊断。PET图像、CT图像及PET/CT的融合图像,根据病变局部18F-FDG代谢特征和病变数目、部位、形态、大小及密度等作出诊断,观察有无异常的18F-FDG浓聚病灶,病灶放射性摄取高于纵隔摄取背景为摄取增高,与纵隔相近或低于纵隔为无摄取增高。半定量分析法是在视觉分析发现的代谢异常区Ⅱ的直径最大层面勾画ROI,并由工作站自动计算出病灶部位SUVmax作为定量评价标准。
-
根据St.Gallen共识[4]对乳腺癌进行分子分型:(1)Luminal A型。雌激素受体(estrogen receptor,ER)和(或)孕激素受体(progesterone receptor, PR)阳性,且Ki67(一种增殖细胞相关的核抗原)<14%,人表皮生长因子受体2(human epidermal growth factor receptor-2, HER2)阴性。(2)Luminal B型。ER和(或)PR阳性,且Ki67≥14%或HER2阳性。(3)HER2过表达型。ER和PR均为阴性,同时HER2表达为阳性。(4)三阴性。ER、PR及HER2表达均为阴性。
-
复发及转移灶的诊断标准为:(1)术后再次手术或活检后病理证实。(2)转移灶的影像学随访确诊依据2次及2次以上的影像学结果确定:① 随访中发现原有病灶增大或出现新发病灶,且B超、CT、MRI或SPECT/CT明确诊断为转移灶;② 经过治疗影像学检查显示原转移病灶明显缩小或消失,并与患者的病情好转相一致也考虑为转移灶。随访时间均大于6个月。总生存时间(overall survival,OS)从手术确认乳腺癌到患者死亡或随访截止为止。无进展生存时间(progression free survival, PFS)从手术后到疾病进展或患者死亡为止。
-
采用SPSS 19.0软件进行统计学分析。根据诊断标准,计算18F-FDG PET/CT的灵敏度、特异度、准确率、阳性预测值及阴性预测值。按照患者年龄、ER、PR、HER2及乳腺癌亚型分组,计量数据均以均数±标准差(x±s)表示,符合正态分布,组间SUVmax的比较采用t检验。采用寿命表法计算年生存率及中位生存时间,Kaplan-Meier分析法进行生存分析,Log-rank检验进行PET/CT检查真阳性组和真阴性组的组间分析。P<0.05表示差异有统计学意义。
-
59例乳腺癌术后患者,其中35例经组织病理学或临床随访确诊存在复发转移,24例未发现复发转移。59例患者中,PET/CT检查真阳性35例、真阴性21例、假阳性3例(图 1)、假阴性0例。18F-FDG PET/CT诊断乳腺癌术后复发转移的灵敏度、特异度、准确率、阳性预测值和阴性预测值分别为100%、87.5%、94.9%、92.1%、100%。PET/CT共发现复发转移病灶72个,SUVmax为1.63~16.22(6.03±3.58),复发转移病灶分布情况见表 1。

图 1 乳腺癌术后PET/CT检查假阳性患者的PET/CT显像图。患者女性, 44岁, 行右乳肿块切除术, 病理示右乳浸润性导管癌Ⅲ级。CT随访42个月未见明显异常病灶, 考虑术后改变。图中, A:CT示右侧腋窝稍低密度灶, PET/CT见18F-FDG葡萄糖高代谢灶SUVmax为3.2;B:右乳局部, PET/CT见18F-FDG葡萄糖高代谢灶SUVmax为2.5。
Figure 1. PET/CT images of a false-positive patient
复发转移部位 病灶数 SUVmax(x±s) 乳腺 06 8.93±6.15 胸壁 11 5.15±3.95 淋巴结 30 5.62±2.75 骨 11 6.57±3.46 肺 08 4.10±2.89 肝 06 8.24±2.76 表 1 72个乳腺癌术后复发转移病灶分布情况及SUVmax
Table 1. Distribution and SUVmax of 72 recurrence and metastasis lesions in patients with postoperative breast cancer
-
乳腺癌术后复发转移灶的SUVmax在年龄、ER、PR、HER2及乳腺癌亚型分组间的差异无统计学意义(t=0.808、0.225、0.355、-0.426和-1.076,均P>0.05)(表 2)。
参数 分组 病灶数(%) SUVmax t值 P值 年龄 ≤50岁 26(36.11) 7.58±5.03 -0.808 0.815 >50岁 46(63.88) 6.50±3.14 ER 阴性 47(65.27) 7.20±3.89 -0.225 0.677 阳性 25(34.72) 6.76±4.29 PR 阴性 49(68.06) 6.47±3.43 -0.355 0.851 阳性 23(31.94) 8.19±3.30 HER2 阴性 33(45.83) 6.24±3.61 -0.426 0.737 阳性 39(54.17) 7.27±3.61 乳腺癌亚型 三阴型 28(38.89) 6.14±4.07 -1.076 0.998 非三阴型 44(61.11) 6.93±3.59 注:表中,ER:雌激素受体;PR:孕激素受体;HER2:人表皮生长因子受体2。 表 2 72个乳腺癌术后复发转移病灶按不同临床病理学参数分组后的SUVmax比较
Table 2. Comparison of SUVmax of 72 recurrence and metastasis lesions according to different clinical pathologic parameters
-
35例PET/CT检查真阳性的患者中,25例分期上调,10例分期未变(表 3)。
PET/CT检查前 PET/CT检查后 分期 例数 分期 例数 Ⅰ 1 Ⅰ 0 Ⅱ 9 Ⅱ 0 Ⅲ 22 Ⅲ 12 Ⅳ 3 Ⅳ 23 表 3 35例18F-FDG PET/CT检查真阳性患者临床再分期改变情况
Table 3. Clinical restaging of 35 cases of true-positve patients under 18F-FDG PET/CT examination
-
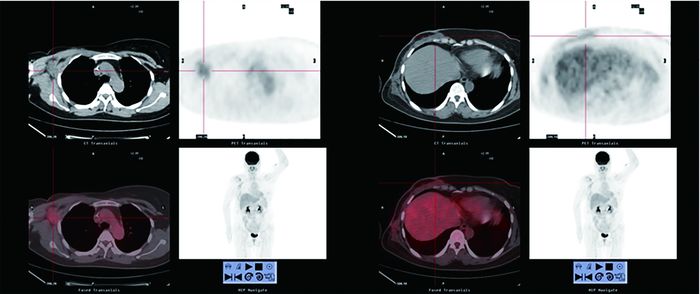
在行PET/CT检查前,有7例患者(11.86%)未接受过任何辅助治疗;40例患者(67.80%)仅接受过化疗;12例患者(20.34%)接受过化疗及内分泌治疗。35例PET/CT检查真阳性患者中,30例PET/CT检查后治疗策略改变(21例PET/CT检查后诊断多发转移,治疗策略由随访改为化疗和(或)放疗(图 2);5例PET/CT检查后诊断淋巴结转移3例、对侧乳腺阳性病灶2例,治疗策略由随访改为手术及化疗;2例为术后化疗前评估;1例发现淋巴结转移,治疗策略改变了放疗的部位;1例发现疾病进展,改变了化疗方案)。另外5例PET/CT检查后治疗策略未改变,3例按原方案继续化疗,2例检查后继续随访或对症治疗(图 3)。21例PET/CT检查真阴性患者中,4例行PET/CT术后评估全身情况后制定治疗方案,9例维持原化疗方案,8例继续随访。

图 2 PET/CT检查后治疗策略改变的乳腺癌患者的PET/CT显像图。患者女性, 59岁, 行左乳改良根治术, 病理示左乳浸润性导管癌Ⅲ级。术后20个月行PET/CT检查示病情进展多处转移, 左胸壁结节SUVmax为2.11, 淋巴结多处SUVmax为3.58, 骨盆骨及脊柱多处SUVmax为5.3。临床分期由Ⅱ级上调至Ⅳ级, 改变化疗方案。图中, A:左侧胸壁结节, PET/CT见18F-FDG葡萄糖高代谢灶SUVmax为2.11;B:纵隔淋巴结, PET/CT见18F-FDG葡萄糖高代谢灶SUVmax为3.58。
Figure 2. PET/CT images of a patient with a change in therapeutic strategy

图 3 PET/CT检查后治疗决策未改变的乳腺癌患者的PET/CT显像图。患者女性, 52岁, 行左乳改良根治术, 术后病理示左乳浸润性导管癌Ⅱ级。术后14个月行PET/CT检查示病情进展多处转移, 左乳手术区尧右乳尧甲状腺尧肝脏尧左侧膈肌局部尧全身淋巴结多处葡萄糖代谢增高, 最高处SUVmax为8.28。临床分期由Ⅲ级上调至Ⅳ级, 患者放弃治疗。图中, A:肝脏多处18F-FDG葡萄糖高代谢灶;B:右乳局部见18F-FDG葡萄糖高代谢灶。
Figure 3. PET/CT images of a patient without a change in therapeutic strategy
-
18F-FDG PET/CT真阳性组和真阴性组患者的中位数生存时间分别为3.58年和4.16年。PET/CT真阳性组患者的1年、2年、3年及4年生存率分别是100%、97.14%、77.14%、57.14%;PET/CT真阴性组患者的1年、2年、3年及4年生存率分别是100%、100%、100%、95%。两组患者OS及PFS的差异有统计学意义(χ2=10.274和17.187,均P<0.01),从生存曲线可以看出,PET/CT真阳性患者的OS及PFS短于真阴性患者(图 4)。
-
本研究是一项关于18F-FDG PET/CT显像诊断乳腺癌术后复发转移的价值,以及PET/CT显像在治疗决策与预后评估中价值的回顾性研究。乳腺癌是组异质性疾病,其癌组织中ER、PR和HER2是重要的分子生物学标志物,也是重要的药物靶点,它们是决定是否行内分泌治疗、靶向治疗甚至化疗的依据,这些治疗会影响转移性乳腺癌的预后[5-6]。Mavi等[7]发现早期乳腺癌原发灶的SUV能够提供有价值的ER、PR及HER2状态信息。但本研究结果显示,乳腺癌复发转移灶的SUVmax在ER、PR、HER2及亚型分组间的差异无统计学意义。据相关文献报道,乳腺癌转移灶的ER、PR、HER2表达并不完全与原发灶一致[8],约有9%的患者ER表达发生变化,22%的患者PR发生变化[9],24%的患者HER2发生变化[10]。因此,对于乳腺癌术后复发转移性病灶,其分子分型有可能发生变化。转移部位的ER、PR、HER2表达有待病理学的证实,其表达与FDG摄取的关系也还需要进一步的研究证实。
对乳腺癌术后患者进行准确地再分期对于患者的意义重大,临床Ⅱ、Ⅲ期患者依然可以通过综合治疗手段争取到相对好的预后,然而一旦被确诊为Ⅳ期乳腺癌,其预后就不尽相同了。乳腺癌术后局部复发率高达30%,复发的部位多累及乳腺或者胸壁[11]。乳腺癌患者术后出现淋巴结转移也较为常见。Cermik等[12]比较了PET/CT在对N1、N2及N3期乳腺癌患者检查时的表现,发现对于N1期患者PET/CT显示腋窝淋巴结转移灵敏度为41%,而对于N2和N3期患者则分别上升为67%和100%。此外,乳腺癌较常见的远处转移部位为骨骼、肺及肝脏。陈建伟等[13]对70例乳腺癌术后患者同日行18F-FDG PET/CT检查及胸部诊断CT扫描,结果显示18F-FDG PET/CT在淋巴结转移、骨骼及胸壁软组织转移中的灵敏度均高于胸部诊断CT,但在肺转移灶检测中的灵敏度(52.3%)低于胸部诊断CT(98.9%)。在Murakami等[14]对47例怀疑乳腺癌复发的患者的研究中,PET/CT诊断25例(53%)肿瘤复发,灵敏度、特异度、准确率分别为96%、91%、94%,灵敏度和准确率均较高。近年来国内外学者的研究均证明,18F-FDG PET/CT显像在诊断乳腺癌原发病灶和转移灶方面都有着重要的临床价值[14-15]。本研究35例PET/CT真阳性患者中有25例分期上调,30例显像后治疗策略改变,对于有效延缓疾病的进展有着重要的意义。与传统的影像学手段相比,18F-FDG PET/CT能更准确、更全面地评价乳腺癌患者的复发和(或)转移,能为临床制定正确的治疗方案提供更重要的信息。
18F-FDG摄取值是肿瘤患者一项重要的预后因素。据文献报道18F-FDG PET异常摄取的患者总体存活率降低[16],这与本研究的结果一致,本研究中PET/CT真阳性组患者的中位生存时间、3年及4年生存率均低于真阴性组患者,表明PET/CT真阳性患者的预后相对较差。
然而,本研究的不足之处在于病例数较少,并且有部分患者术后至PET/CT检查时间较短,易造成PET/CT显像假阳性,仍有待于进一步大样本的长期随访研究。
综上所述,18F-FDG PET/CT是一种先进的功能影像学方法,对诊断乳腺癌术后复发转移有较高的灵敏度和准确率,能及时修正乳腺癌术后患者的临床再分期,以及指导复发转移患者临床治疗策略的改变,这对改善患者的预后有重要意义。
18F-FDG PET/CT在乳腺癌术后随访中的价值
Clinical value of 18F-FDG PET/CT in follow-up of postoperative breast cancer
-
摘要:
目的研究18F-FDG PET/CT在乳腺癌术后复发转移中的诊断价值以及在治疗决策改变和预后中的价值。 方法回顾性分析2007年7月至2016年9月行18F-FDG PET/CT检查的59例女性乳腺癌术后患者,分析PET/CT对真阳性患者临床治疗策略的影响,并对真阳性组和真阴性组患者进行Kaplan-Meier生存分析。在PET/CT显像真阳性组中,按照患者年龄、雌激素受体(ER)、孕激素受体(PR)、人类表皮生长因子受体2(HER2)及乳腺癌亚型进行分组,组间SUVmax的比较采用t检验。 结果在59例乳腺癌术后患者中,18F-FDG PET/CT诊断乳腺癌术后复发转移的灵敏度、特异度、准确率、阳性预测值和阴性预测值分别为100%、87.5%、94.9%、92.1%、100%。PET/CT真阳性组与真阴性组患者平均随访时间分别为(58.33±37.69)个月和(47.40±25.99)个月,中位数生存时间分别为3.58年和4.16年,PET/CT真阴性患者的总生存时间(OS)和无进展生存时间(PFS)长于真阳性患者,且差异有统计学意义(χ2=10.274和17.187,均P < 0.01)。35例PET/CT真阳性患者中,25例分期上调,30例检查后治疗策略改变。乳腺癌复发转移灶的SUVmax在年龄、ER、PR、HER2及亚型分组间的差异无统计学意义(t=0.808、0.225、0.355、-0.426和-1.076,均P>0.05)。 结论 18F-FDG PET/CT显像在诊断乳腺癌术后复发转移中有较高的灵敏度和准确率,能及时修正乳腺癌术后复发转移患者的临床再分期,以及指导临床治疗策略的改变,在乳腺癌的术后随访中具有较高的临床应用价值。 -
关键词:
- 乳腺肿瘤 /
- 正电子发射断层显像术 /
- 体层摄影术,X线计算机 /
- 预后 /
- 肿瘤转移 /
- 肿瘤复发
Abstract:Objective To evaluate the clinical value of 18F-FDG PET/CT imaging in detecting the recurrence and metastasis, therapeutic strategy, and prognosis of postoperative breast cancer patients. Methods From July 2007 to September 2016, 59 patients with postoperative breast cancer were retrospectively studied. The influence of PET/CT imaging results on the choice of therapeutic strategy was analyzed. Kaplan-Meier survival analysis was conducted based on the true-positive and true-negative groups of patients. Comparison of SUVmax according to the age, estrogen receptor(ER), progesterone receptor (PR), human epidermal growth factor receptor-2(HER2), and subtypes of the patients in the PET/CT imaging true-positive group was performed using t-test. Results The sensitivity, specificity, accuracy, positive predictive value, and negative predictive value of 18F-FDG PET/CT were 100%, 87.5%, 94.9%, 92.1%, and 100%, respectively. No statistically significant difference in the SUVmax for breast cancer recurrence and metastasis was observed between the two groups of patients according to age, ER, PR, HER2, and subtypes of breast cancer(t=0.808, 0.225, 0.355, -0.426 and -1.076, all P>0.05). The true-positive and true-negative groups were followed for a median period of 58.33±37.69 and 47.40±25.99 months, and their median survival times were 3.58 and 4.16 years, respectively. The two groups significantly differed in the overall survival and progression-free survival times (Kaplan-Meier survival analysis, χ2=10.274, P < 0.01; χ2=17.187, P < 0.01). Clinical restaging was raised in 25 true-positive patients after PET/CT imaging, and the therapeutic strategy was changed in 30 true-positive patients. Conclusions PET/CT imaging is highly sensitive and accurate in diagnosing the recurrence and metastasis of postoperative breast cancer and allows clinical restaging in a timely manner. It helps in choosing the appropriate clinical therapeutic strategy, thereby improving the prognosis of patients. Compared with the true-positive patients, the true-negative patients who underwent PET/CT imaging had a better prognosis. Therefore, PET/CT imaging has a high clinical value in the follow-up of postoperative breast cancer patients. -
图 2 PET/CT检查后治疗策略改变的乳腺癌患者的PET/CT显像图。患者女性, 59岁, 行左乳改良根治术, 病理示左乳浸润性导管癌Ⅲ级。术后20个月行PET/CT检查示病情进展多处转移, 左胸壁结节SUVmax为2.11, 淋巴结多处SUVmax为3.58, 骨盆骨及脊柱多处SUVmax为5.3。临床分期由Ⅱ级上调至Ⅳ级, 改变化疗方案。图中, A:左侧胸壁结节, PET/CT见18F-FDG葡萄糖高代谢灶SUVmax为2.11;B:纵隔淋巴结, PET/CT见18F-FDG葡萄糖高代谢灶SUVmax为3.58。
Figure 2. PET/CT images of a patient with a change in therapeutic strategy
图 3 PET/CT检查后治疗决策未改变的乳腺癌患者的PET/CT显像图。患者女性, 52岁, 行左乳改良根治术, 术后病理示左乳浸润性导管癌Ⅱ级。术后14个月行PET/CT检查示病情进展多处转移, 左乳手术区尧右乳尧甲状腺尧肝脏尧左侧膈肌局部尧全身淋巴结多处葡萄糖代谢增高, 最高处SUVmax为8.28。临床分期由Ⅲ级上调至Ⅳ级, 患者放弃治疗。图中, A:肝脏多处18F-FDG葡萄糖高代谢灶;B:右乳局部见18F-FDG葡萄糖高代谢灶。
Figure 3. PET/CT images of a patient without a change in therapeutic strategy
表 1 72个乳腺癌术后复发转移病灶分布情况及SUVmax
Table 1. Distribution and SUVmax of 72 recurrence and metastasis lesions in patients with postoperative breast cancer
复发转移部位 病灶数 SUVmax(x±s) 乳腺 06 8.93±6.15 胸壁 11 5.15±3.95 淋巴结 30 5.62±2.75 骨 11 6.57±3.46 肺 08 4.10±2.89 肝 06 8.24±2.76 表 2 72个乳腺癌术后复发转移病灶按不同临床病理学参数分组后的SUVmax比较
Table 2. Comparison of SUVmax of 72 recurrence and metastasis lesions according to different clinical pathologic parameters
参数 分组 病灶数(%) SUVmax t值 P值 年龄 ≤50岁 26(36.11) 7.58±5.03 -0.808 0.815 >50岁 46(63.88) 6.50±3.14 ER 阴性 47(65.27) 7.20±3.89 -0.225 0.677 阳性 25(34.72) 6.76±4.29 PR 阴性 49(68.06) 6.47±3.43 -0.355 0.851 阳性 23(31.94) 8.19±3.30 HER2 阴性 33(45.83) 6.24±3.61 -0.426 0.737 阳性 39(54.17) 7.27±3.61 乳腺癌亚型 三阴型 28(38.89) 6.14±4.07 -1.076 0.998 非三阴型 44(61.11) 6.93±3.59 注:表中,ER:雌激素受体;PR:孕激素受体;HER2:人表皮生长因子受体2。 表 3 35例18F-FDG PET/CT检查真阳性患者临床再分期改变情况
Table 3. Clinical restaging of 35 cases of true-positve patients under 18F-FDG PET/CT examination
PET/CT检查前 PET/CT检查后 分期 例数 分期 例数 Ⅰ 1 Ⅰ 0 Ⅱ 9 Ⅱ 0 Ⅲ 22 Ⅲ 12 Ⅳ 3 Ⅳ 23 -
[1] Yang L, Parkin DM, Ferlay J, et al. Estimates of cancer incidence in China for 2000 and projections for 2005[J]. Cancer Epidemiol Biomarkers Prev, 2005, 14(1):243-250. [2] Mayer EL, Burstein HJ. Chemotherapy for metastatic breast cancer[J]. Hematol Oncol Clin North Am, 2007, 21(2):257-272. DOI:10. 1016/j.hoc.2007.03.001. [3] Berman AT, Thukral AD, Hwang WT, et al. Incidence and patterns of distant metastases for patients with early-stage breast cancer after breast conservation treatment[J]. Clin Breast Cancer, 2013, 13(2):88-94. DOI:10.1016/j.clbc.2012.11.001. [4] Goldhirsch A, Winer EP, Coates AS, et al. Personalizing the treatment of women with early breast cancer:hightlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2013[J]. Ann Oncol, 2013, 24(9):2206-2223. DOI:10.1093/annonc/mdt303. [5] Slamon DJ, Leyland-Jones B, Shak S, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2[J]. N Engl J Med, 2001, 344(11):783-792. DOI:10.1056/NEJM200103153441101. [6] Blackwell KL, Burstein HJ, Storniolo AM, et al. Randomized study of Lapatinib alone or in combination with trastuzumab in women with ErbB2-positive, trastuzumab-refractory metastatic breast cancer[J]. J Clin Oncol, 2010, 28(7):1124-1130. DOI:10.1200/JCO. 2008.21.4437. [7] Mavi A, Cermik TF, Urhan M, et al. The effects of estrogen, progesterone, and C-erbB-2 receptor states on 18F-FDG uptake of primary breast cancer lesions[J]. J Nucl Med, 2007, 48(8):1266-1272. DOI:10.2967/jnumed.106.037440. [8] 陈道宝, 杨红健. 乳腺癌ER与FES-PET[J]. 中国肿瘤, 2008, 17(7):588-592. DOI:10.3969/j.issn.1004-0242.2008.07.013.
Chen DB, Yang HJ. Estrogen receptor and fluoroestradiol-positron emission tomography in breast cancer[J]. Chin Cancer, 2008, 17(7):588-592. doi: 10.3969/j.issn.1004-0242.2008.07.013[9] Montagna E, Bagnardi V, Rotmensz N, et al. Breast cancer subtypes and outcome after local and regional relapse[J]. Ann Oncol, 2012, 23(2):324-331. DOI:10.1093/annonc/mdr129. [10] Niikura N, Liu J, Hayashi N, et al. Loss of human epidermal growth factor receptor 2(HER2) expression in metastatic sites of HER2-overexpressing primary breast tumors[J]. J Chin Oncol, 2012, 30(6):593-599. DOI:10.1200/JCO.2010.33.8889. [11] Zangheri B, Messa C, Picchio M, et al. PET/CT and breast cancer[J]. Eur J Nucl Med Mol Imaging, 2004, 31 supple1:S135-142. DOI:10.1007/s00259-004-1536-7. [12] Cermik TF, Mavi A, Basu S, et al. Impact of FDG PET on the pre-operative staging of newly diagnosed breast cancer[J]. Eur J Nucl Med Mol Imaging, 2008, 35(3):475-483. DOI:10.1007/s00259-007-0580-5. [13] 陈建伟, 王杰, 刘标, 等. PET/CT与胸部诊断CT在乳腺癌术后随访中的比较[J]. 南京医科大学学报:自然科学版, 2010, 30(2):195-198, 213.
Chen JW, Wang J, Liu B, et al. Impact of 18F-FDG PET/CT compared with diagnostic CT on following-up breast cancer after surgery[J]. Acta Univ Med Nanjing(Natural Science), 2010, 30(2):195-198, 213.[14] Murakami R, Kumita S, Yoshida T, et al. FDG-PET/CT in the diag-nosis of recurrent breast cancer[J]. Acta Radiol, 2012, 53(1):12-16. DOI:10.1258/ar.2011.110245. [15] 刘晓强, 周海中, 杜鹃. 乳腺癌患者术前18F-FDG PET/CT显像肿瘤代谢体积测定的预后价值[J]. 实用临床医药杂志, 2014, 18(21):55-59. DOI:10.7619/jcmp.201421016.
Liu XQ, Zhou HZ, Du J. Prognositc value of metabolic tumor volume measured by 18F-FDG PET/CT imaging in preoperative breast cancer patients[J]. J Clin Med Prac, 2014, 18(21):55-59. doi: 10.7619/jcmp.201421016[16] Maffione AM, Lopci E, Bluemel C, et al. Diagnostic accuracy and impact on management of 18F-FDG PET and PET/CT in colorectal liver metastasis:a meta-analysis and systematic review[J]. Eur J Nucl Med Mol Imaging, 2015, 42(1):152-163. DOI:10.1007/s00259-014-2930-4. -





 下载:
下载: