-
HMGB1 is ubiquitously expressed in most cells to maintain a large "pool" of pre-formed protein in the nucleus[1-2] owing to the existence of two nuclear-localization sequences(NLS) that facilitate its nuclear transportation[3]. Within the nucleus, HMGB1 binds chromosomal DNA, and fulfills its nuclear functions in maintaining nucleosomal structures and regulating gene expression[4]. The complete depletion of HMGB1 expression renders animals more susceptible to infectious[5] or injurious insults[6-7], suggesting an overall protective role of intracellular HMGB1 against stresses[8].
In response to microbial toxins(such as CpG-DNA and endotoxin)[9-10], cytokines[e.g., interferon (IFN)-γ and Cold-inducible RNA-binding protein (CIRP)][11-13] or oxidative free radicals(e.g., hydrogen peroxide)[14], macrophages/monocytes acetylate and/or phosphorylate the NLS of HMGB1[8, 15-17], enabling its sequestration into cytoplasmic vesicles destined for subsequent secretion[2, 11, 18]. Cytoplasmic HMGB1 can be secreted through several pathways, including the double-stranded RNA-activated protein kinase R (PKR)-and Caspase-1/Caspase-11-mediated inflammasome activation and pyroptosis. For instance, genetic disruption PKR expression or pharmacological inhibition of PKR phosphorylation similarly reduces NLRP3 or NLRP1 agonists-induced inflammasome activation[19-20], pyroptosis[19-20] and HMGB1 release[19].
In addition to active secretion, HMGB1 can be passively released from damaged cells[21] following ischemia/reperfusion[22-23], trauma[24-25], or toxemia[26-28], thereby serving as damage-associated molecular pattern(DAMP) molecule. Although radiation emits high energy photons that can ionize atoms and disrupt molecular bonds, it was previously unknown whether it similarly induces HMGB1 cytoplasmic translocation and release. Here we provided evidence that X-ray irradiation induces a time- and dose-dependent HMGB1 cytoplasmic translocation and release by tumor cells in vitro, and stimulates systemic HMGB1 accumulation in vivo.
-
Human skin fibroblast GM0639 cell line was obtained from the Radiobiological Laboratory of the National Research Center for Environment and Health (GSF), Germany. Human bronchial epithelial 16HBE cell line was obtained from the Shanghai Cell Collection Center of the Chinese Academy of Sciences. These cells were maintained as a monolayer in low glucose DMEM culture medium supplemented with 10% fetal calf serum(Hyclone, Logan, UT, USA), 2 mmol/L glutamine, penicillin(100 U/ml) and streptomycin(100 U/ml). Cells were kept at 37℃ in an atmosphere of 5% carbon dioxide and 95% air, and subcultured twice a week to remain in exponential growth. Cells were washed twice with, and subsequently cultured in, serum-free OPTI-MEM I medium (Gibco BRL, Grand Island, NY) before X-ray irradiation.
-
Male and female Sprague-Dawley rats(8-12 weeks old, 220-250 g) were allowed to acclimate for 7 days before X-ray irradiation. All animal procedures were approved by the Institutional Animal Care and Use Committee of the Soochow University. Rats were housed in the animal facility of the Key Laboratory of Jiangsu Radiation Medicine and Protection under standard temperature, and light and dark cycles.
-
As a form of radiation when charged electrons or ions of sufficient energy hit a material with high speed, X-ray was generated in high voltage electron tubes of X-ray generators(Primus High-Energy Sie-mens), which emitted high energy photons at a rate of 200 cGy/min. For cell irradiation, a locator with a source-cell distance of 100 mm, irradiation field of 40 cm×40 cm was employed to expose cell cultures to X-ray at doses ranging from 0 to 8 Gy. For animal experiment, rats were irradiated using X-ray generator emitting at a fixed dose rate of 200 cGy/min, with an irradiation field of 40 cm×40 cm, centered 100 mm above the animals. At various time points after X-ray radiation, 0.5 ml blood was collected from 6 rats(3 male and 3 female) by tail bleeding, and serum hmgb1 levels were determined using the Shino-Test Corporation ELISA kit.
-
At 24 h after X-ray radiation, cellular HMGB1 was immunostained with HMGB1-specific polyclonal antibodies, and images were acquired using a fluorescent microscope as previously described[29]. Alternatively, the subcellular localization of HMGB1 was examined by a cell fractionation/Western blotting technique as previously described[29]. Cell fractionation is based on differential lysis of plasma and nuclear membranes by nonionic detergent(NP-40). Briefly, after selective lysis of the plasma membrane in low salt buffer(10 mmol/L HEPES, pH 7.9; 10 mmol/L KCl; 0.1 mmol/L EDTA; 0.1 mmol/L EGTA; 1 mmol/L DTT; 0.5 mmol/L PMSF, 1% NP-40), the intact nuclei was collected by a quick centrifugation step(7000 g, 1 min, 4℃), leaving the cytoplasmic fraction in the supernatant. The nuclei pellet was resuspended in NP-40 high salt buffer(20 mmol/L HEPES, pH 7.9; 0.4 mol/L NaCl; 1 mmol/L EDTA; 1 mmol/L EGTA; 1 mmol/L DTT; 1 mmol/L PMSF, 1% NP-40), and briefly sonicated to generate the nuclear fraction. After fractionation, the protein content of different fractions was determined by a Bradford method, and each fraction was assayed for levels of various protein by Western blotting analysis using primary antibodies specific for HMGB1, a cytoplasmic protein(β-actin, Santa Cruz Biotechnology), and a nuclear protein (Lamin B1, BD Biosciences).
-
Immediately after irradiation, cells grown on covered slide chambers(Lab-Tek, Nunc, Napterville, IL, USA) were washed with PBS and fixed with 2% paraformaldehyde in PBS for 15 min. After three washes of PBS with 10 min each, the cells were treated with 0.2% Triton X-100 solution in PBS for 5 min, and stained with mouse monoclonal antibody for Serine 139 phospho-H2AX from Millipore (Cat. 05636, 1: 200) overnight at 4℃. After extensive washings, FITC-labeled rabbit anti-mouse antibodies were added, and fluorescent images of cells were captured using a fluorescence microscope.
-
Cell viability was measured by the reduction of yellow tetrazolium salt[MTT, 3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide) by dehydrogenase of metabolically active cells, to generate reducing purple formazan that can be solubilized and quantified by spectrophotometry. The MTT substrate was prepared in a physiologically balanced solution, added to cell culture at a final concentration of 0.2 mg/ml, and incubated for 2 hours. The quantity of the resultant purple formazan was measured by recording changes in absorbance at 570 nm using a plate reading spectrophotometer.
-
Data are expressed as mean ± SEM of two independent experiments in triplicates. One-way analyses of variance (ANOVA) followed by the Tukey′s test for multiple comparisons were used to compare between different groups. A P value less than 0.05 was considered statistically significant.
-
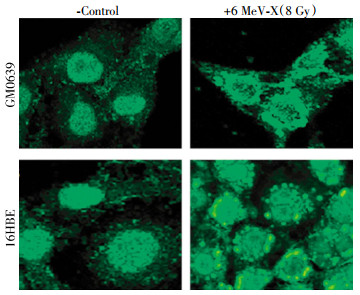
To investigate the impact of X-ray irradiation on possible HMGB1 release, we first determined its effect on HMGB1 cytoplasmic translocation-an essential step for subsequent HMGB1 release. Quiescent tumor cells constitutively expressed HMGB1 and maintained an intracellular "pool" of HMGB1 predominantly in the nucleus(Fig. 1, left panels). At 24 h post X-ray irradiation(8.0 Gy), large amount of HMGB1 staining was also noticed in numerous cytoplasmic vesicles (Fig. 1, right panels), suggesting that radiation stimulated tumor cells to actively translocate nuclear HMGB1 into the cytoplasmic vesicles before releasing into the extracellular milieu.
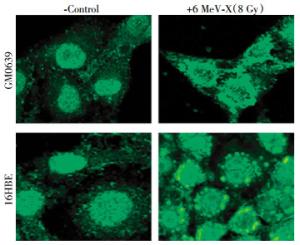
Figure 1. Radiation induced cytoplasmic HMGB1 translocation in tumor cells. Human skin fibroblast(GM0639) and bronchial epithelial(16HBE) cells were subjected to 6 MeV X-ray radiation at a dose of 8 Gy for 24 h, and assayed for HMGB1 cytoplasmic translocation by immunohistochemistry using HMGB1-specific antibodies. Note that HMGB1 was predominantly localized in the nuclear region of un-treated cells("control"), but found in both cytoplasmic and nuclear regions of X-ray radiated cells ("6 MeV-X").
To confirm the cytoplasmic HMGB1 translocation, whole cell lysates were fractionated and the levels of HMGB1 in the cytoplasmic and nuclear fractions were determined by Western blotting analysis. The relative levels of HMGB1(with reference to Lamin B1) in the nuclear fractions were significantly reduced in both GM0639 and 16HBE tumor cells after X-ray irradiation(Fig. 2A). In parallel, the relative levels of HMGB1(with reference to β-actin) in the cytoplasmic fraction were significantly elevated after irradiation (Fig. 2B), confirming that X-ray irradiation induced significant HMGB1 cytoplasmic translocation in these tumor cells.
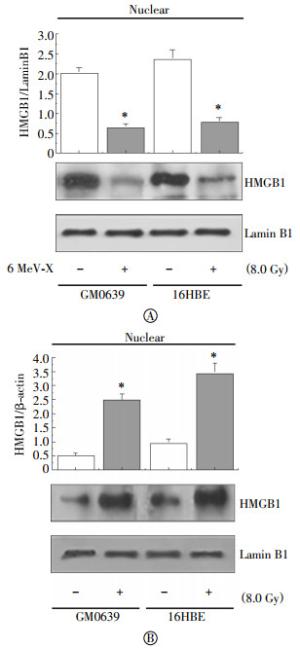
Figure 2. Radiation inversely altered nuclear and cytoplasmic HMGB1 levels. Following X-ray radiation, cytoplasmic and nuclear fractions were isolated, and assayed for levels of HMGB1 along with a nuclear(Lamin B1) or cytoplasmic(β-actin) marker by Western blotting analysis. Equal loading of samples was confirmed by Western blotting analysis of respective fractions with cytoplasmic (β-actin) or nuclear (Lamin B1) protein markers.
-
To determine whether X-ray radiation induces HMGB1 release, extracellular levels of HMGB1 in cell-conditioned culture medium were determined by Western blotting analysis. The levels of HMGB1 in the culture medium conditioned by the quiescent tumor cells were relatively low. Following X-ray irradiation, extracellular HMGB1 levels were elevated in a dose- and time-dependent fashion (Fig. 3). At a dose as low as 4-8 Gy, X-ray radiation induced HMGB1 release as early as 6 h post stimulation(Fig. 3).
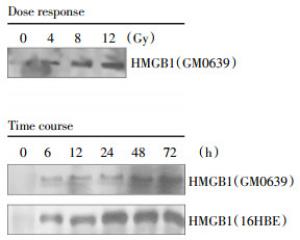
Figure 3. Radiation induced a dose- and time-dependent HMGB1 release. Human skin fibroblast(GM0639) and/or bronchial epithelial (16HBE) were exposed to X-ray radiation at various doses for different time periods, and extracellular levels of HMGB1 were determined by Western blotting analysis. Note that proteins were recovered from equal volume of cell-conditioned medium, and sample loading was normalized by equal volume of cell-conditioned medium.
In addition to active secretion, HMGB1 could also be passively released by injured cells. It is known that ionizing radiation can cause double-stranded breaks of chromosomal DNA, which activates histone γ-H2AX phosphorylation, and results in the recruitment of DNA repair proteins to form the γ-H2AX foci, a biomarker for chromosomal DNA damage. To test the impact of radiation on DNA damage, we examined the effect of X-ray irradiation on the formation of γ-H2AX foci in both tumor cell lines. As indicated in Fig. 4A, X-ray irradiation, at a dose as low as 4 Gy, induced marked DNA damage as judged by the formation of γ-H2AX foci(Fig. 4A). Consistently, the cell viability was significantly reduced by X-ray irradiation in both GM0639 and 16HBE tumor cells (Fig. 4B), suggesting that X-ray irradiation induced HMGB1 release partly through passive leakage from these dying cells.

Figure 4. Radiation caused DNA damage and loss of cell viability. Human skin fibroblast (GM0639) and bronchial epithelial (16HBE) cells exposed to X-ray at a dose of 4 Gy, and cells were stained with γ-H2AX-specific antibodies to detect DNA damage. In parallel, the cell viability was determined by MTT assay, and expressed as a % of controls in the absence of X-ray radiation. *, P < 0.05 versus untreated control at respective time points.
-
To examine whether X-ray irradiation induces HMGB1 release in vivo, we subjected male and female rats to X-ray irradiation at different doses, and measured circulating levels of HMGB1 by ELISA. In agreement with the in vitro findings, X-ray irradiation induced systemic HMGB1 accumulation in a dose- and time-dependent fashion (Fig. 5). At a dose as low as 6 Gy, X-ray irradiation induced significant HMGB1 accumulation in the circulation as early as 6 h post stimulation (Fig. 5).
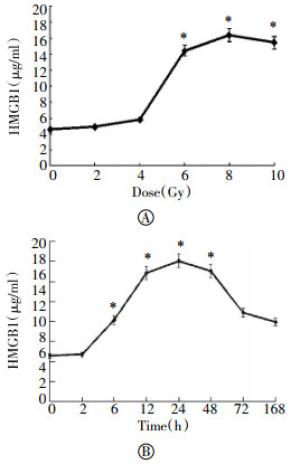
Figure 5. Radiation elevated circulating HMGB1 levels in vivo. Male Sprague-Dawley rats were exposed to X-ray radiation at various doses and for different time periods, and blood samples were collected to measure serum HMGB1 levels by ELISA. *, P < 0.05 versus untreated controls(no radiation, Panel A; or immediately prior to X-ray radiation, Panel B).
-
As a form of radiation, X-rays emit high energy photons that can donate energy to cellular molecules, kicking out atomic electrons from the inner orbit to produce unstable and highly reactive free radicals. These radicals quickly react with nearby molecules, resulting in breakage of chemical bonds or oxidation (addition of oxygen atoms) of the affected molecules. In the present study, we demonstrated that X-ray radiation induced DNA damage as manifested by the formation of γ-H2AX foci in the nuclei, and induced cytoplasmic HMGB1 translocation and release in human skin fibroblast(GM0639) and bronchial epithelial(16HBE) cell lines. Similar findings have been demonstrated using the breast(MCF-7, data not shown), lung(NCI-H1703), prostate(DU-145 and PC-3) and colorectal(HCT 15 and SW480)[30] and glioblastoma (T98G and U251MG)[31] tumor cell lines.
As aforementioned, similar cytoplasmic HMGB1 translocation occurs in innate immune cells following stimulation with cytokines(IFN-γ)[11] or hydrogen peroxide[14, 32]. Because cytoplasmic HMGB1 translocation was not closely associated with the occurrence of cell death[11, 14, 32], it was believed that cytoplasmic HMGB1 translocation might be an active process regulated by chemical modifications of HMGB1 NLS. Notably, ionizing radiation also induces water radiolysis to produce radicals, which can be converted into hydrogen peroxide (H2O2). Because hydrogen peroxide can induce active HMGB1 secretion or passive leakage[14, 32], we propose that X-ray induces HMGB1 release through multiple mechanisms that are dependent on both active cytoplasmic translocation and passive leakage.
In animals, X-ray radiation causes injury initially at the skin, but goes beyond the surface and continues to damage inner tissues in the body. Although many irradiated cells could repair DNA and protein damage, some cells would die of necrosis or apoptosis. It is thus possible that radiation may induce HMGB1 secretion and leakage to amplify an inflammatory response. Indeed, HMGB1 carries three redox-sensitive cysteine residues(C23, C45 and C106), and can exist in three isoforms termed "HMGB1" (all thiol form), "disulfide HMGB1"(partially oxidized), and oxidized HMGB1[33-34]. The "all-thiol" HMGB1 binds to other chemokines (e.g., CXCL12) to facilitate leukocyte recruitment via the CXCR4 receptor[35] or other signaling molecules[36-38] to the sites of injury[39-40]. In a sharp contrast, the "disulfide" HMGB1 can activate immune cells to produce cytokines/chemokines via TLR4 or other receptors such as RAGE[41], TLR2, TLR4[42-44], TLR9[10, 41], cluster of differentiation 24 (CD24)/Siglec-10[45], Mac-1[38], thrombomodulin[46], or single transmembrane domain proteins(e.g., syndecans)[47]. Once fully oxidized, HMGB1 is devoid of either chemokine or cytokine activities[33-34]. Thus, extracellular HMGB1 could serve as a proinflammatory signal to recruit and activate innate immune cells to sustain a potentially injurious inflammatory response to radiation.
It has been well established that excessive HMGB1 release adversely contributes to the pathogenesis of infection- and injury-elicited inflammatory diseases, because HMGB1-neutralizing antibodies are protective in animal models of sepsis[9, 48-51], ischemia/reperfusion[22, 52-53], trauma[54-55], chemical toxemia[26, 56-57], atherosclerosis[58], gastric ulcer[59] and hyperoxia[60]. Even during lethal infection[9, 48-51], tissue injury is accompanied by massive HMGB1 release that further amplifies the cytokine storm to precipitate organ dysfunction. In fact, HMGB1 itself could trigger caspase-1-dependent programmed cell death, pyroptosis, which is characterized by rapid plasma membrane rupture, and release of proinflammatory intracellular contents (including HMGB1)[61]. Although it is difficult to distinguish between microbial infection-induced sepsis from injury-elicited systemic inflammatory response syndrome[62-63], it might be important to develop strategies to specifically attenuate radiation-mediated inflammatory responses.
Recently, many herbal components including the green tea polyphenolic catechins[64], tanshinones[29], carbenoxolone[65] have been shown to inhibit HMGB1 release through multiple mechanisms. These divergent mechanisms include the stimulation of autophagic degradation of cytoplasmic HMGB1[66], the enhancement of endocytosis of exogenous HMGB1 into cytoplasmic vesicles[67], or inhibition of key signaling molecules(e.g., PKR) involved in the regulation of HMGB1 release[68]. It may be important to assess whether these herbal HMGB1 inhibitors can similarly prevent radiation-induced excessive inflammation without compromising the efficacy of radiation in the treatment of malignant cancers.
Ionizing Radiation Induces HMGB1 Cyto-plasmic Translocation and Extracellular Release
Ionizing Radiation Induces HMGB1 Cyto-plasmic Translocation and Extracellular Release
-
Abstract:
Objective A nucleosomal protein, HMGB1, can be secreted by activated immune cells or passively released by dying cells, thereby amplifying rigorous inflammatory responses. In this study we aimed to test the possibility that radiation similarly induces cytoplasmic HMGB1 translocation and release. Methods Human skin fibroblast (GM0639) and bronchial epithelial (16HBE) cells and rats were exposed to X-ray radiation, and HMGB1 translocation and release were then assessed by immunocytochemistry and immunoassay, respectively. Results At a wide dose range(4.0-12.0 Gy), X-ray radiation induced a dramatic cytoplasmic HMGB1 translocation, and triggered a time- and dose-dependent HMGB1 release both in vitro and in vivo. The radiation-mediated HMGB1 release was also associated with noticeable chromosomal DNA damage and loss of cell viability. Conclusions Radiation induces HMGB1 cytoplasmic translocation and extracellular release through active secretion and passive leakage processes. -
Key words:
- X-ray /
- HMGB1 /
- Tumor cells /
- Inflammatory response /
- Damage-associated molecule pattern molecules
-
Figure 1. Radiation induced cytoplasmic HMGB1 translocation in tumor cells. Human skin fibroblast(GM0639) and bronchial epithelial(16HBE) cells were subjected to 6 MeV X-ray radiation at a dose of 8 Gy for 24 h, and assayed for HMGB1 cytoplasmic translocation by immunohistochemistry using HMGB1-specific antibodies. Note that HMGB1 was predominantly localized in the nuclear region of un-treated cells("control"), but found in both cytoplasmic and nuclear regions of X-ray radiated cells ("6 MeV-X").
Figure 2. Radiation inversely altered nuclear and cytoplasmic HMGB1 levels. Following X-ray radiation, cytoplasmic and nuclear fractions were isolated, and assayed for levels of HMGB1 along with a nuclear(Lamin B1) or cytoplasmic(β-actin) marker by Western blotting analysis. Equal loading of samples was confirmed by Western blotting analysis of respective fractions with cytoplasmic (β-actin) or nuclear (Lamin B1) protein markers.
Figure 3. Radiation induced a dose- and time-dependent HMGB1 release. Human skin fibroblast(GM0639) and/or bronchial epithelial (16HBE) were exposed to X-ray radiation at various doses for different time periods, and extracellular levels of HMGB1 were determined by Western blotting analysis. Note that proteins were recovered from equal volume of cell-conditioned medium, and sample loading was normalized by equal volume of cell-conditioned medium.
Figure 4. Radiation caused DNA damage and loss of cell viability. Human skin fibroblast (GM0639) and bronchial epithelial (16HBE) cells exposed to X-ray at a dose of 4 Gy, and cells were stained with γ-H2AX-specific antibodies to detect DNA damage. In parallel, the cell viability was determined by MTT assay, and expressed as a % of controls in the absence of X-ray radiation. *, P < 0.05 versus untreated control at respective time points.
Figure 5. Radiation elevated circulating HMGB1 levels in vivo. Male Sprague-Dawley rats were exposed to X-ray radiation at various doses and for different time periods, and blood samples were collected to measure serum HMGB1 levels by ELISA. *, P < 0.05 versus untreated controls(no radiation, Panel A; or immediately prior to X-ray radiation, Panel B).
-
[1] Chen G, Li J, Qiang X, et al. Suppression of HMGB1 release by stearoyl lysophosphatidylcholine:an additional mechanism for its therapeutic effects in experimental sepsis[J]. J Lipid Res, 2005, 46(4):623-627. DOI:10.1194/jlr.C400018-JLR200. [2] Bonaldi T, Talamo F, Scaffidi P, et al. Monocytic cells hyperacetylate chromatin protein HMGB1 to redirect it towards secretion[J]. EMBO J, 2003, 22(20):5551-5560. DOI:10.1093/emboj/cdg516. [3] Kang R, Chen R, Zhang Q, et al. HMGB1 in health and disease[J]. Mol Aspects Med, 2014, 40:1-116. DOI:10.1016/j.mam.2014.05.001. [4] Bustin M. At the crossroads of necrosis and apoptosis: signaling to multiple cellular targets by HMGB1[J/OL]. Sci STKE, 2002, 2002(151): pe39[2016-02-26]. http://www.ncbi.nlm.nih.gov/pubmed/12297673. [5] Yanai H, Matsuda A, An J, et al. Conditional ablation of HMGB1 in mice reveals its protective function against endotoxemia and bacterial infection[J]. Proc Natl Acad Sci USA, 2013, 110(51):20699-20704. DOI:10.1073/pnas.1320808110. [6] Kang R, Zhang Q, Hou W, et al. Intracellular Hmgb1 inhibits inflammatory nucleosome release and limits acute pancreatitis in mice[J]. Gastroenterology, 2014, 146(4):1097-1107. DOI:10.1053/j.gastro.2013.12.015. [7] Huang H, Nace GW, Mcdonald KA, et al. Hepatocyte-specific high-mobility group box 1 deletion worsens the injury in liver ischemia/reperfusion:a role for intracellular high-mobility group box 1 in cellular protection[J]. Hepatology, 2014, 59(5):1984-1997. DOI:10.1002/hep.26976. [8] Lu B, Wang C, Wang M, et al. Molecular mechanism and therapeutic modulation of high mobility group box 1 release and action:an updated review[J]. Expert Rev Clin Immunol, 2014, 10(6):713-727. DOI:10.1586/1744666X.2014.909730. [9] Wang H, Bloom O, Zhang M, et al. HMG-1 as a late mediator of endotoxin lethality in mice[J]. Science, 1999, 285(5425):248-251. doi: 10.1126/science.285.5425.248 [10] Ivanov S, Dragoi AM, Wang X, et al. A novel role for HMGB1 in TLR9-mediated inflammatory responses to CpG-DNA[J]. Blood, 2007, 110(6):1970-1981. DOI:10.1182/blood-2006-09-044776. [11] Rendon-Mitchell B, Ochani M, Li J, et al. IFN-gamma induces high mobility group box 1 protein release partly through a TNF-dependent mechanism[J]. J Immunol, 2003, 170(7):3890-3897. doi: 10.4049/jimmunol.170.7.3890 [12] Kim JH, Kim SJ, Lee IS, et al. Bacterial endotoxin induces the release of high mobility group box 1 via the IFN-beta signaling pathway[J]. J Immunol, 2009, 182(4):2458-2466. DOI:10.4049/jimmunol.0801364. [13] Qiang X, Yang WL, Wu R, et al. Cold-inducible RNA-binding protein(CIRP) triggers inflammatory responses in hemorrhagic shock and sepsis[J]. Nat Med, 2013, 19(11):1489-1495. DOI:10.1038/nm.3368. [14] Tang D, Shi Y, Kang R, et al. Hydrogen peroxide stimulates macrophages and monocytes to actively release HMGB1[J]. J Leukoc Biol, 2007, 81(3):741-747. DOI:10.1189/jlb.0806540. [15] Lu B, Antoine DJ, Kwan K, et al. JAK/STAT1 signaling promotes HMGB1 hyperacetylation and nuclear translocation[J]. Proc Natl Acad Sci USA, 2014, 111(8):3068-3073. DOI:10.1073/pnas.1316925111. [16] Youn JH, Shin JS. Nucleocytoplasmic shuttling of HMGB1 is regulated by phosphorylation that redirects it toward secretion[J]. J Immunol, 2006, 177(11):7889-7897. doi: 10.4049/jimmunol.177.11.7889 [17] Wang H, Ward MF, Sama AE. Targeting HMGB1 in the treatment of sepsis[J].Expert Opin Ther Targets, 2014, 18(3):257-268. DOI:10.1517/14728222.2014.863876. [18] Gardella S, Andrei C, Ferrera D, et al. The nuclear protein HMGB1 is secreted by monocytes via a non-classical, vesicle-mediated secretory pathway[J]. EMBO Rep, 2002, 3(10):995-1001. DOI:10.1093/embo-reports/kvf198. [19] Lu B, Nakamura T, Inouye K, et al. Novel role of PKR in inflammasome activation and HMGB1 release[J]. Nature, 2012, 488(7413):670-674. DOI:10.1038/nature11290. [20] Hett EC, Slater LH, Mark KG, et al. Chemical genetics reveals a kinase-independent role for protein kinase R in pyroptosis[J]. Nat Chem Biol, 2013, 9(6):398-405. DOI:10.1038/nchembio.1236. [21] Scaffidi P, Misteli T, Bianchi ME. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation[J]. Nature, 2002, 418(6894):191-195. DOI:10.1038/nature00858. [22] Tsung A, Sahai R, Tanaka H, et al. The nuclear factor HMGB1 mediates hepatic injury after murine liver ischemia-reperfusion[J]. J Exp Med, 2005, 201(7):1135-1143. DOI:10.1084/jem.20042614. [23] Andrassy M, Volz HC, Igwe JC, et al. High-mobility group box-1 in ischemia-reperfusion injury of the heart[J]. Circulation, 2008, 117(25):3216-3226. DOI:10.1161/CIRCULATIONAHA.108.769331. [24] Cohen MJ, Brohi K, Calfee CS, et al. Early release of high mobility group box nuclear protein 1 after severe trauma in humans:role of injury severity and tissue hypoperfusion[J]. Crit Care, 2009, 13(6):R174. DOI:10.1186/cc8152. [25] Peltz ED, Moore EE, Eckels PC, et al. HMGB1 is markedly elevated within 6 hours of mechanical trauma in humans[J]. Shock, 2009, 32(1):17-22. doi: 10.1097/SHK.0b013e3181997173 [26] Zhou RR, Liu HB, Peng JP, et al. High mobility group box chromosomal protein 1 in acute-on-chronic liver failure patients and mice with ConA-induced acute liver injury[J]. Exp Mol Pathol, 2012, 93(2):213-219. DOI:10.1016/j.yexmp.2012.05.006. [27] Antoine DJ, Dear JW, Lewis PS, et al. Mechanistic biomarkers provide early and sensitive detection of acetaminophen-induced acute liver injury at first presentation to hospital[J]. Hepatology, 2013, 58(2):777-787. DOI:10.1002/hep.26294. [28] Seo YS, Kwon JH, Yaqoob U, et al. HMGB1 recruits hepatic stellate cells and liver endothelial cells to sites of ethanol induced parenchymal cell injury[J]. Am J Physiol Gastrointest Liver Physiol, 2013, 305(11):G838-848. DOI:11.1152/ajpgi.00151.2013. [29] Li W, Li J, Ashok M, et al. A cardiovascular drug rescues mice from lethal sepsis by selectively attenuating a late-acting proinflammatory mediator, high mobility group box 1[J]. J Immunol, 2007, 178(6):3856-3864. doi: 10.4049/jimmunol.178.6.3856 [30] Schildkopf P, Frey B, Mantel F, et al. Application of hyperthermia in addition to ionizing irradiation fosters necrotic cell death and HMGB1 release of colorectal tumor cells[J]. Biochem Biophys Res Commun, 2010, 391(1):1014-1020. DOI:10.1016/j.bbrc.2009.12. 008. [31] Pasi F, Paolini A, Nano R, et al. Effects of single or combined treatments with radiation and chemotherapy on survival and danger signals expression in glioblastoma cell lines[J/OL]. Biomed Res Int, 2014, 2014: 453497[2016-02-26]. http://www.ncbi.nlm.nih.gov/pubmed/?term=25097859. DOI: 10.1155/2014/453497. [32] Tang D, Kang R, Xiao W, et al. Nuclear heat shock protein 72 as a negative regulator of oxidative stress(Hydrogen peroxide)-induced HMGB1 cytoplasmic translocation and release[J]. J Immunol, 2007, 178(11):7376-7384. doi: 10.4049/jimmunol.178.11.7376 [33] Venereau E, Casalgrandi M, Schiraldi M, et al. Mutually exclusive redox forms of HMGB1 promote cell recruitment or proinflammatory cytokine release[J]. J Exp Med, 2012, 209(9):1519-1528. DOI:10.1084/jem.20120189. [34] Yang H, Lundback P, Ottosson L, et al. Redox modification of cysteine residues regulates the cytokine activity of high mobility group Box-1(HMGB1)[J]. Mol Med, 2012, 18(2):250-259. DOI:10.2119/molmed.2011.00389. [35] Schiraldi M, Raucci A, Muñoz LM, et al. HMGB1 promotes recruitment of inflammatory cells to damaged tissues by forming a complex with CXCL12 and signaling via CXCR4[J]. J Exp Med, 2012, 209(3):551-563. DOI:10.1084/jem.20111739. [36] Yang D, Chen Q, Yang H, et al. High mobility group box-1 protein induces the migration and activation of human dendritic cells and acts as an alarmin[J]. J Leukoc Biol, 2007, 81(1):59-66. DOI:10.1189/jlb.0306180. [37] Dumitriu IE, Bianchi ME, Bacci M, et al. The secretion of HMGB1 is required for the migration of maturing dendritic cells[J]. J Leukoc Biol, 2007, 81(1):84-91. DOI:10.1189/jlb.0306171. [38] Orlova VV, Choi EY, Xie C, et al. A novel pathway of HMGB1-mediated inflammatory cell recruitment that requires Mac-1-integrin[J]. EMBO J, 2007, 26(4):1129-1139. DOI:10.1038/sj.emboj.7601552. [39] Degryse B, Bonaldi T, Scaffidi P, et al. The high mobility group (HMG) boxes of the nuclear protein HMG1 induce chemotaxis and cytoskeleton reorganization in rat smooth muscle cells[J]. J Cell Biol, 2001, 152(6):1197-1206. doi: 10.1083/jcb.152.6.1197 [40] Degryse B, de Virgilio M. The nuclear protein HMGB1, a new kind of chemokine?[J]. FEBS Lett, 2003, 553(1/2):11-17. [41] Tian J, Avalos AM, Mao SY, et al. Toll-like receptor 9-dependent activation by DNA-containing immune complexes is mediated by HMGB1 and RAGE[J]. Nat Immunol, 2007, 8(5):487-496. DOI:10.1038/ni1457. [42] Yu M, Wang H, Ding A, et al. HMGB1 signals through toll-like receptor (TLR) 4 and TLR2[J]. Shock, 2006, 26(2):174-179. DOI:10.1097/01.shk.0000225404.51320.82. [43] Ha T, Xia Y, Liu X, et al. Glucan phosphate attenuates myocardial HMGB1 translocation in severe sepsis through inhibiting NF-κB activation[J]. Am J Physiol Heart Circ Physiol, 2011, 301(3):H848-H855. DOI:10.1152/ajpheart.01007.2010. [44] Xiang M, Shi X, Li Y, et al. Hemorrhagic shock activation of NLRP3 inflammasome in lung endothelial cells[J]. J Immunol, 2011, 187(9):4809-4817. DOI:10.4049/jimmunol.1102093. [45] Chen GY, Tang J, Zheng P, et al. CD24 and siglec-10 selectively repress tissue damage-induced immune responses[J]. Science, 2009, 323(5922):1722-1725. DOI:10.1126/science.1168988. [46] Abeyama K, Stern DM, Ito Y, et al. The N-terminal domain of thrombomodulin sequesters high-mobility group-B1 protein, a novel antiinflammatory mechanism[J]. J Clin Invest, 2005, 115(5):1267-1274. DOI:10.1172/JCI22782. [47] Salmivirta M, Rauvala H, Elenius K, et al. Neurite growth-promoting protein (amphoterin, p30) binds syndecan[J]. Exp Cell Res, 1992, 200(2):444-451. doi: 10.1016/0014-4827(92)90194-D [48] Abraham E, Arcaroli J, Carmody A, et al. HMG-1 as a mediator of acute lung inflammation[J]. J Immunol, 2000, 165(6):2950-2954. doi: 10.4049/jimmunol.165.6.2950 [49] Yang H, Ochani M, Li J, et al. Reversing established sepsis with antagonists of endogenous high-mobility group box 1[J]. Proc Natl Acad Sci USA, 2004, 101(1):296-301. DOI:10.1073/pnas.2434651100. [50] Qin S, Wang H, Yuan R, et al. Role of HMGB1 in apoptosis-mediated sepsis lethality[J]. J Exp Med, 2006, 203(7):1637-1642. DOI:10.1084/jem.20052203. [51] Suda K, Kitagawa Y, Ozawa S, et al. Anti-high-mobility group box chromosomal protein 1 antibodies improve survival of rats with sepsis[J]. World J Surg, 2006, 30(9):1755-1762. DOI:10.1007/s00268-005-0369-2. [52] Wu H, Ma J, Wang P, et al. HMGB1 contributes to kidney ischemia reperfusion injury[J]. J Am Soc Nephrol, 2010, 21(11):1878-1890. DOI:10.1681/ASN.2009101048. [53] Qiu J, Nishimura M, Wang Y, et al. Early release of HMGB-1 from neurons after the onset of brain ischemia[J]. J Cereb Blood Flow Metab, 2008, 28(5):927-938. DOI:10.1038/sj.jcbfm.9600582. [54] Okuma Y, Liu K, Wake H, et al. Anti-high mobility group box-1 antibody therapy for traumatic brain injury[J]. Ann Neurol, 2012, 72(3):373-384. DOI:10.1002/ana.23602. [55] Shimazaki J, Matsumoto N, Ogura H, et al. Systemic involvement of high-mobility group box 1 protein and therapeutic effect of anti-high-mobility group box 1 protein antibody in a rat model of crush injury[J]. Shock, 2012, 37(6):634-638. DOI:10.1097/SHK.0b013e31824ed6b7. [56] Yang R, Zhang S, Cotoia A, et al. High mobility group B1 impairs hepatocyte regeneration in acetaminophen hepatotoxicity[J]. BMC Gastroenterol, 2012, 12:45. DOI:10.1186/1471-230X-12-45.:45-12. [57] Nadatani Y, Watanabe T, Tanigawa T, et al. High mobility group box 1 promotes small intestinal damage induced by nonsteroidal anti-inflammatory drugs through Toll-like receptor 4[J]. Am J Pathol, 2012, 181(1):98-110. DOI:10.1016/j.ajpath.2012.03.039. [58] Hirata Y, Kurobe H, Higashida M, et al. HMGB1 plays a critical role in vascular inflammation and lesion formation via toll-like receptor 9[J]. Atherosclerosis, 2013, 231(2):227-233. DOI:10.1016/j.atherosclerosis.2013.09.010. [59] Nadatani Y, Watanabe T, Tanigawa T, et al. High-Mobility Group Box 1 Inhibits Gastric Ulcer Healing through Toll-Like Receptor 4 and Receptor for Advanced Glycation End Products[J/OL]. PLoS One, 2013, 8: e80130[2016-02-26]. https://www.ncbi.nlm.nih.gov/pubmed/?term=24244627. [60] Patel VS, Sitapara RA, Gore A, et al. High mobility group Box-1 mediates hyperoxia-induced impairment of pseudomonas aeruginosa clearance and inflammatory lung injury in mice[J]. Am J Respir Cell Mol Biol, 2013, 48(3):280-287. DOI:10.1165/rcmb.2012-0279OC. [61] Xu J, Jiang Y, Wang J, et al. Macrophage endocytosis of high-mobility group box 1 triggers pyroptosis[J]. Cell Death Differ, 2014, 21(8):1229-1239. DOI:10.1038/cdd.2014.40. [62] Sursal T, Stearns-Kurosawa DJ, Itagaki K, et al. Plasma bacterial and mitochondrial DNA distinguish bacterial sepsis from sterile systemic inflammatory response syndrome and quantify inflammatory tissue injury in nonhuman primates[J]. Shock, 2013, 39(1):55-62. DOI:10.1097/SHK.0b013e318276f4ca. [63] Vincent JL, Opal SM, Marshall JC, et al. Sepsis definitions:time for change[J]. Lancet, 2013, 381(9868):774-775. DOI:10.1016/S0140-6736(12)61815-7. [64] Li W, Ashok M, Li J, et al. A Major Ingredient of Green Tea Rescues Mice from Lethal Sepsis Partly by Inhibiting HMGB1[J/OL]. PLoS One, 2007, 2: e1153[2016-02-26]. http://www.ncbi.nlm.nih.gov/pubmed/?term=17987129. [65] Li W, Li J, Sama AE, et al. Carbenoxolone blocks endotoxin-induced protein kinase R(PKR) activation and high mobility group box 1(HMGB1) release[J]. Mol Med, 2013, 19(19):203-211. DOI:10.2119/molmed.2013.00064. [66] Li W, Zhu S, Li J, et al. EGCG stimulates autophagy and reduces cytoplasmic HMGB1 levels in endotoxin-stimulated macrophages[J]. Biochem Pharmacol, 2011, 81(9):1152-1163. DOI:10.1016/j.bcp.2011.02.015. [67] Zhang Y, Li W, Zhu S, et al. Tanshinone IIA Sodium sulfonate facilitates endocytic HMGB1 uptake[J]. Biochem Pharmacol, 2012, 84(11):1492-1500. DOI:10.1016/j.bcp.2012.09.015. [68] Macmicking JD, Nathan C, Hom G, et al. Altered responses to bacterial infection and endotoxic shock in mice lacking inducible nitric oxide synthase[J]. Cell, 1995, 81(4):641-650. -





 下载:
下载: