-
炎症性肠病是病因不明的慢性非特异性肠道疾病,目前主要依据肠镜和组织病理学检查来确诊。其在国内的发病率有增高的趋势,尤其是溃疡性结肠炎[1]。由于该疾病临床表现复杂多样,炎症性肠病的漏诊率为60.9%,误诊率为36.8%[2];溃疡性结肠炎的漏诊率为32.1%,误诊率为27.5%[3]。炎症性肠病由于不能早期及时得到正确的治疗,容易成为慢性活动性病或带来严重并发症[4]。探寻特异的炎症性肠病的早期诊断方式显得极为重要。
近些年随着分子影像学技术的发展,PET/CT作为一种新型的非侵入性的诊断手段受到越来越多的关注,因此各种不同类型PET显像剂的研究也逐渐成为研究热点。丁酸盐是一种短链脂肪酸,是肠上皮细胞的主要能量来源,可促进水、钠吸收,改善上皮细胞紧密连接功能,加速上皮修复,对维持肠道粘膜稳态具有重要意义[5-6],还可以通过抑制促炎细胞因子信号通路发挥抗炎作用[7]。2013年,有研究者指出在动物模型中,丁酸盐可能成为治疗炎症性肠病的药物[8]。11C-乙酸可用作前列腺癌的显像剂,18F-氟乙酸是乙酸的类似物,它具有更长的半衰期,在PET诊断前列腺癌时可以成为11C-乙酸的替代显像剂[9]。若将丁酸中的一个氢原子以氟原子取代,做为丁酸的类似物,氟代丁酸(fluorobutyric acid,FBA)很可能仍具有和丁酸相似的体内活性。因此,为寻找一种新型特异性炎症性肠病的显像剂,本研究拟对正丁酸及其甲酯进行18F标记,初步探讨18F-FBA及其甲酯作为PET显像剂的可行性。
-
4-溴丁酸甲酯(纯度 > 97%)、无水K2CO3(纯度99.995%)购自美国Aldrich公司;无水乙腈(99.9%,extra dry)购自比利时Acros公司;K222(纯度98%)购自德国ABX公司;用于高压液相色谱(high pressure liquid chromatography,HPLC)的乙腈(色谱纯)购自瑞士Fisher公司;其余试剂均为国产分析纯。QMA(Sep-pak Light)和C18(Sep-pak Light)购自美国Waters公司;Symemetry300TM C18色谱柱,5 μ,4.6 mm×250.0 mm,购自美国Waters公司;Alltima C18,250.0 mm×10.0 mm,10 μ,购自美国Grace公司。
-
RDS111型回旋加速器购自美国CTI公司;PET-MF-2V-IT-I型18F正电子药物合成模块购自北京派特科技有限公司;高效液相色谱仪(配515型泵)、486型高效液相色谱紫外检测器均购自美国Waters公司;高效液相色谱放射性检测器购自美国Bio-Scan公司;Mini-Scan和flow counter购自美国Bio-Scan公司;CRC-15R型放射性活度计、micro PET购自德国西门子公司。
-
C57BL/6J小鼠,体质量20~24 g,来源于北京华阜康生物技术有限公司。
-
4-溴丁酸甲酯先与18F-发生亲核取代,生成中间体4-18F-氟代丁酸甲酯(methyl 4-[18F] fluorobutyrate,4-18F-FBAM),再经NaOH水解、HCl中和后即得到4-18F-FBA。
反应式如下:
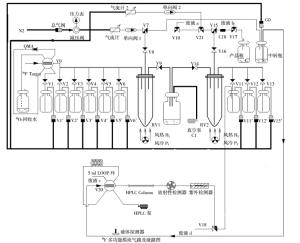
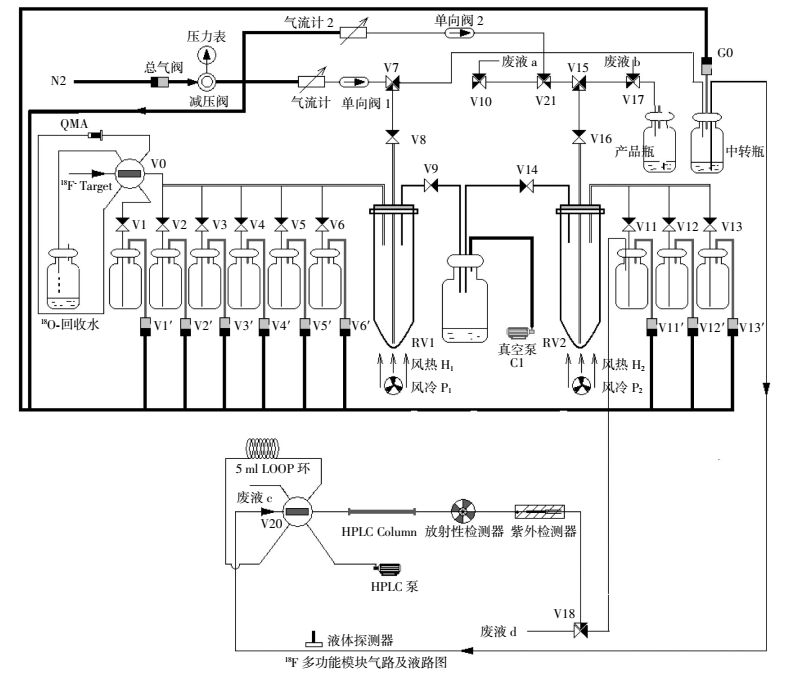
18F多功能合成模块合成4-18F-FBA结构示意图见图 1。具体反应过程如下:
图 1 4-18F-氟代丁酸合成示意图 图中,HPLC:高压液相色谱。
Figure 1. Sketch map of the synthesis of 4-[18F] fluorobutyric acid
(1)1号瓶中的1.5 ml淋洗液(K222:13 mg/ml、K2CO3:3 mg/ml)将富集在QMA柱上的18F-淋洗至1号反应管(RV1)中,于116℃下加热蒸干溶液。
(2)将2号瓶中的2 ml无水乙腈加入到1号反应管中,于116℃下加热,蒸干溶液后冷却。
(3)加入3号瓶中的液体(5 μl 4-溴丁酸甲酯溶于1.0 ml无水乙腈)至1号反应管中,于90℃下加热反应5 min。
(4)1号反应管冷却后加入4 ml 4号瓶中的水,转移至中转瓶中。
(5)打开G0开关,将中转瓶中的液体转移至LOOP环,HPLC(色谱柱Alltima C18,250.0 mm×10.0 mm,10 μ,流动相,乙腈/水=40/60,流速4 ml/min)进行分离纯化。收集保留时间6.8~7.8 min的组分至11号瓶中。
(6)将11号瓶中收集到的组分加入至2号反应管(RV2)中,再加入12号瓶中的0.5 ml 1 mol/L的NaOH溶液,于115℃下加热10 min后,然后加入13号瓶中的0.5 ml 1 mol/L HCl进行中和。
(7)将2号反应管中的液体转移至收集瓶中,得到4-18F-FBA。
-
4-18F-FBAM是合成4-18F-FBA的中间体,它的反应式如下:
18F多功能合成模块合成4-18F-FBAM结构示意图见图 2。
在用HPLC分离得到4-18F-FBAM后,因是乙腈溶液,需用固相萃取的方法除去有机溶剂。具体过程如下:用30 ml注射用水稀释,分3次加入至2号反应管中,液体流经C18柱进入废液瓶中,再将12号瓶中20 ml注射用水分两次加入至2号反应管中,冲洗C18柱。将13号瓶中的0.5 ml乙醇加入至2号反应管中,然后流经C18柱,洗脱产品至已预先加好4.5 ml生理盐水的收集瓶中,得到4-18F-FBAM。
-
(1)目测两种产品的颜色及澄明度,分别用精密pH试纸测定pH值。
(2)分别检测两种产品的放射化学纯度。采用HPLC法,色谱条件:色谱柱(Symemetry300TM C18,5 μ,4.6 mm×250.0 mm),紫外检测波长210 nm,4-18F-FBA的流动相为0.5 mol/L NaH2PO4溶液,流速0.5 ml/min;4-18F-FBAM的流动相为乙腈/水=40/60,流速1 ml/min。
(3)室温下测定两种产品3个半衰期内(6 h)的放射化学纯度,检测其稳定性。
-
取C57BL/J小鼠尾静脉注射7.4~11.1 MBq的4-18F-FBA,注射前后测量注射器放射性,精确计算注射活度。注射药物后30 min在2%异氟烷麻醉下(加2 L/min氧气)用西门子Inoven专用小动物PET进行动物显像。能量窗口设定为350~650 keV,符合时间设定为3.432 ns,采集时间设定为5 min。
-
4-18F-FBA合成过程需时约40 min,产品的放化收率为35%;4-18F-FBAM合成过程需时约20 min,产品的放化收率为50%(均未经时间校正)。放射性比活度分别为12 GBq/μmol和15 GBq/μmol。
-
4-18F-FBA与4-18F-FBAM均澄明无颗粒。精密pH试纸测定的4-18F-FBA的pH值为6.5,4-18F-FBAM的为7.1。
-
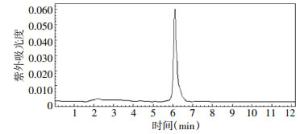
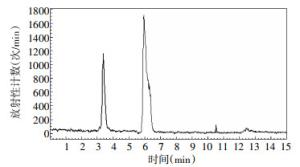
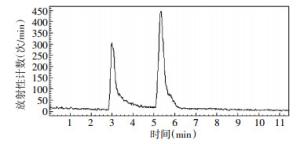
4-18F-FBA的的放化纯度 > 95%,4-18F-FBAM的放化纯度 > 99%,二者的保留时间分别为6.3 min和5.4 min,色谱图分别见图 3、图 4。4-氟代丁酸的紫外吸收峰的保留时间是6.2 min,色谱图见图 5。

图 3 4-18F-氟代丁酸的放射化学纯度色谱图
Figure 3. The radiochemical purity chromatogram of 4-[18F] fluorobutyric acid with high pressure liquid chromatography
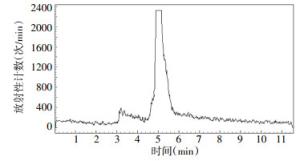
图 4 4-18F-氟代丁酸甲酯的放射化学纯度色谱图
Figure 4. The radiochemical purity chromatogram of methyl 4-[18F] fluorobutyrate with high pressure liquid chromatography
由图 3和图 5可见,4-18F-FBA的放射化学吸收峰的保留时间6.3 min与4-氟代丁酸的紫外吸收峰的保留时间6.2 min是对应的,说明合成的4-18F-FBA结构是正确的。
-
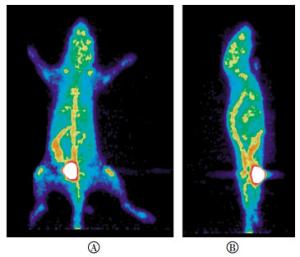
小鼠micro PET图像显示,注射4-18F-FBA 30 min后,肠道和脊柱放射性摄取均比较明显,膀胱亦有较多的放射性浓聚(图 8)。
-
PET显像是一种非侵入性的无创检测手段,在临床诊断中显示出越来越多的优势。PET显像依赖于PET显像剂。多种参与生物体内代谢的活性物质被开发成PET显像剂,例如,目前最广泛应用的18F-FDG,它是葡萄糖类似物,参与葡萄糖的代谢,进入细胞后,不能被进一步代谢而聚集于细胞中,从而达到诊断目的[11];11C-乙酸参与脂肪酸代谢[12];18F-氟代胸苷参与核苷酸代谢[13]等。其中脂肪酸代谢是其中重要的一种代谢途径。丁酸是一种短链脂肪酸,正电子同位素标记后的分子应该能够作为丁酸的类似物参与脂肪酸代谢,从而达到显像目的。将丁酸用正电子核素18F标记后,用作炎性肠病PET显像剂的工作尚未见文献报道。
本研究参照文献[10]中2-18F-氟代丙酸的合成方法,以4-溴丁酸甲酯为原料,经亲核取代反应生成4-18F-FBAM,4-18F-FBAM经NaOH水解、HCL中和后即得4-18F-FBA。4-18F-FBA经与标准品对照,确证其结构。4-18F-FBAM是水解前的原料,可以推断其结构也是正确的。整个反应简单易操作,且收率较高。在亲核取代反应后采用了最直接有效的HPLC进行中间体的分离纯化,不仅可以很好地除去反应中的催化剂K222,而且也能完全除掉未反应的前体4-溴丁酸甲酯,更好地保证产品的化学纯度。国产PET-MF-2V-IT-I型18F多功能模块在我国已推广使用很多年,可以根据放化合成路线设计自动化过程。本实验在此模块上实现了预期两种PET显像剂的自动化合成,合成时间短,而且大大减少了工作人员在合成过程中接受的放射性照射剂量。4-18F-FBAM是反应的中间体,它可以作为4-18F-FBA的前示踪剂(proradiotracer),因其具有更好的脂溶性,可能在体内具有与4-18F-FBA不完全相同的分布特性。有研究表明,18F-氟乙酸乙酯能很快穿过血脑屏障,可用于神经胶质代谢的PET显像研究[14],而18F-氟乙酸的水溶性大,不能穿透血脑屏障,不能用于脑内的相关PET显像研究。因此也将4-18F-FBAM作为PET显像剂进行了相关的质量控制研究。同时对4-18F-FBA进行了正常小鼠体内显像。质控结果表明,无论4-18F-FBA还是4-18F-FBAM,在室温放置30 min后均出现了明显的脱氟现象,表明它们均不稳定。推测其原因可能是在溶液中,由于羧基带负电,4位的碳原子由于18F-的强吸电作用带有部分正电荷,与羧基上带负电荷的氧(-COO-)发生分子内结合,能形成一个稳定的五元环结构,从而导致脱氟。4-18F-FBA正常小鼠体内30 min显像结果显示,脊柱有明显的放射性浓聚,说明该显像剂在体内也出现了脱氟现象。作为PET显像剂,要求至少在3个半衰期内是稳定的,因此4-18F-氟丁酸及其甲酯不符合作为PET显像剂的要求,不适合作为PET显像剂进行进一步的深入研究。
小鼠体内显像结果表明,虽然由于脱氟造成了脊柱和关节的放射性浓聚,但肠道亦有明显的放射性摄取,这与设计初衷是相吻合的,说明18F-FBA类似物可以做为肠道炎性疾病的显像剂。4位被18F取代的丁酸不稳定,改变18F的取代位置为2位或3位,因其不能再形成稳定的五元环结构,推测应该稳定。该研究正在进行中。
本工作利用PET-MF-2V-IT-I型18F多功能合成模块分别自动化合成了4-18F-FBA和4-18F-FBAM,合成时间分别为40 min和20min,产品的放化收率分别为35%和50%(均未经时间校正),放射化学纯度均大于95%。但二者稳定性均较差,室温放置30 min后即出现明显的脱氟现象,4-18F-FBA小鼠体内显像结果也表明脱氟。虽然合成方法简便快速,但因为4-18F-FBA和4-18F-FBAM的不稳定性,使得其不适合作为PET显像剂进行深入研究。但小鼠体内显像提示,18F-FBA类似物作为PET显像剂在肠道疾病的诊断中很可能具有潜在研究价值。2位和3位取代的18F-FBA正在研究中。
4-18F-氟代丁酸及其甲酯作为PET显像剂可行性的初步研究
The preliminary feasibility studies of 4-[18F] fluorobutyric acid and its methyl ester as PET imaging agents
-
摘要:
目的 利用PET-MF-2V-IT-I氟多功能合成模块自动化合成4-18F-氟代丁酸及其甲酯,并初步探讨其作为PET显像剂的可行性。 方法 前体4-溴代丁酸甲酯与18F-发生氟代反应,中间体4-18F-氟代丁酸甲酯用高压液相色谱法分离,收集保留时间在6.8~7.8 min的组分,将此组分与NaOH溶液于115 ℃下加热水解10 min,加入HCl调至中性,得到4-18F-氟代丁酸;稀释后过C18柱,再用20 ml注射用水清洗C18柱,0.5 ml乙醇洗脱后用生理盐水稀释,得到4-18F-氟代丁酸甲酯。分别目测两种产品的澄清度,精密试纸测定pH值、测定放射化学纯度和稳定性。正常小鼠尾静脉注射4-18F-氟代丁酸30 min后行micro PET显像。 结果 4-18F-氟代丁酸和4-18F-氟代丁酸甲酯的自动合成时间分别为40 min和20 min,放化收率分别为35%和50%(均未经时间校正),pH值分别为6.5和7.1,产品放射化学纯度均 > 95%。二者均澄清无颗粒,室温放置30 min后均出现脱氟现象。正常小鼠micro PET显像结果显示,脊柱有明显的放射性浓聚,表明4-18F-氟代丁酸在体内脱氟,肠道亦有较高的放射性摄取。 结论 4-18F-氟代丁酸及其甲酯的合成方法操作简便,合成时间短,且放化收率也较高,但因其不稳定,不适合用作PET显像剂进行进一步研究。但小鼠micro PET显像结果提示,18F-氟代丁酸类似物在肠道疾病诊断中有潜在研究价值。 -
关键词:
- 4-18F-氟代丁酸 /
- 4-18F-氟代丁酸甲酯 /
- 自动化合成 /
- PET显像剂
Abstract:Objective To synthesize 4-[18F] fluorobutyric acid and its methyl ester automatically using PET-MF-2V-IT-I multifunction synthesizer and to evaluate their feasibility as PET imaging agents. Methods The precursor, methyl 4-bromobutyrate was fluorinated with 18F-. The intermediate, methyl 4-[18F] fluorobutyrate was separated by high pressure liquid chromatography. The components which retention time was from 6.8 to 7.8 min were collected. The components were heated with NaOH solution at 115℃ for 10 min to hydrolyze, and then neutralized with HCl solution. The product, 4-[18F] fluorobutyric acid was obtained. The components were diluted and passed through a C18 cartridge, which was washed with 20 ml water then. The product, methyl 4-[18F] fluorobutyrate was obtained after 0.5 ml ethanol eluted the cartridge and was diluted with saline. The quality controls of both products were done. The clarity was tested visually, the pH value was tested with precise pH paper, and the radiochemical purity and stability were also measured. 4-[18F] fluorobutyric acid(7.4~11.1 MBq) was injected into the C57BL/J mice via tail vein for micro PET imaging at 30 min time-point. Results The whole synthetic procedure periods of 4-[18F] fluorobutyric acid and methyl 4-[18F] fluorobutyrate were 40 min and 20 min separately. The radiochemical yields of the two products were about 35% and 50%(both decay uncorrected) respectively. The pH values were 6.5 and 7.1. The radiochemical purities were both > 95%. Both the product solutions were clear and no particles. Defluorination was observed in these two product solutions 0.5 h later at room temperature. The micro PET imaging showed that the uptake of spine was clearly, which indicated that the imaging agent was defluorinated in vivo, the uptake of intestine was also higher. Conclusion 4-[18F] fluorobutyric acid and its methyl ester could be automatically synthesized on PET-MF-2V-IT-I multifunction synthesizer simply and rapidly. The synthetic yield, radiochemical purity were high. But for their instabilities at room temperature, the two agents were not suitable for further studies as PET imaging agents. But the micro PET imaging suggested the analogs of [18F] fluorobutyric acid may have its potential in diagnosis of intestinal diseases. -
-
[1] 邓长生, 夏冰.炎症性肠病[M].北京:人民卫生出版社, 1998.
[2] 常玉英, 欧阳钦, 胡仁伟.我国克罗恩病的漏诊误诊情况分析[J].中华消化内镜杂志, 2005, 22(6):372-375. doi: 10.3760/cma.j.issn.1007-5232.2005.06.003
[3] 常玉英, 欧阳钦, 胡仁伟.我国有关炎症性肠病诊断的文献复习[J].四川医学, 2005, 26(4):373-375. doi: 10.3969/j.issn.1004-0501.2005.04.008
[4] 郑家驹.炎症性肠病的流行病学及中国研究概况//中华医学会消化病学分会.中华医学会第九次全国消化系统疾病学术会议论文汇编[G], 广州, 2009.广州: 中华医学会消化病学分会, 2009: 29-33.
[5] Scheppach W, Weiler F. The butyrate story:old wine in new bottles?[J]. Curr Opin Clin Nutr Metab Care, 2004, 7(5):563-567. doi: 10.1097/00075197-200409000-00009 [6] Hamer HM, Jonkers D, Venema K, et al. Review article:the role of butyrate on colonic function[J]. Aliment Pharmacol Ther, 2008, 27(2):104-119. [7] Joseph J, Mudduluru G, Antony S, et al. Expression profiling of Sodium butyrate(NaB)-treated cells:identification of regulation of genes related to cytokine signaling and cancer metastasis by NaB[J]. Oncogene, 2004, 23(37):6304-6315. doi: 10.1038/sj.onc.1207852 [8] Furusawa Y, Obata Y, Fukuda S, et al. Commensal microbe-derived butyrate induces the differentiation of colonic regulatory T cells[J]. Nature, 2013, 504(7480):446-450. doi: 10.1038/nature12721 [9] Ponde DE, Dence CS, Oyama N, et al. 18F-fluoroacetate:a potential acetate analog for prostate tumor imaging--in vivo evaluation of 18F-fluoroacetate versus 11C-acetate[J]. J Nucl Med, 2007, 48(3):420-428. [10] Pillarsetty N, Punzalan B, Larson SM. 2-18F-Fluoropropionic acid as a PET imaging agent for prostate cancer[J]. J Nucl Med, 2009, 50(10):1709-1714. doi: 10.2967/jnumed.109.064212 [11] Som P, Atkins HL, Bandoypadhyay D, et al. A fluorinated glucose analog, 2-fluoro-2-deoxy-D-glucose(18F):nontoxic tracer for rapid tumor detection[J]. J Nucl Med, 1980, 21(7):670-675. [12] Yoshimoto M, Waki A, Yonekura Y, et al. Characterization of acetate metabolism in tumor cells in relation to cell proliferation:Acetate metabolism in tumor cells[J]. Nucl Med Biol, 2001, 28(2):117-122. [13] Shields AF, Grierson JR, Dohmen BM, et al. Imaging proliferation in vivo with[18F]FLT and positron emission tomography[J]. Nat Med, 1998, 4(11):1334-1336. doi: 10.1038/3337 [14] Mori T, Sun LQ, Kobayashi M, et al. Preparation and evaluation of ethyl[18F] fluoroacetate as a proradiotracer of[18F] fluoroacetate for the measurement of glial metabolism by PET[J]. Nucl Med Biol, 2009, 36(2):155-162. -





 下载:
下载: