-
乳腺癌是一种常见的严重威胁妇女健康的亲骨性恶性肿瘤,其发病率呈逐年上升,骨转移是其最常见的并发症之一,而发生骨转移将极大地影响患者的生存质量,且预后不佳。判断乳腺癌有无骨转移,对制定治疗方案、判断预后有重要意义[1]。全身骨显像是诊断骨转移的重要方法,其通过放射性核素的异常分布发现转移灶。糖类抗原15-3(carbohydrate antigen, CA15-3)和癌胚抗原(carcinoembryonic antigen, CEA)是乳腺癌诊断中常用的血清肿瘤标志物。本研究探讨全身骨显像联合血清CA15-3、CEA检测在乳腺癌骨转移诊断中的价值,现报道如下。
-
良性病变组:45例,女性,年龄21~65岁,为本院收治的良性乳腺疾病患者,包括纤维瘤、乳腺囊肿、乳腺增生等。
乳腺癌骨转移组:46例,女性,年龄36~75岁,为在我院门诊或住院治疗的乳腺癌患者,包括浸润性导管癌23例、浸润性单纯癌及腺癌15例、浸润性小叶癌4例、髓样癌3例、其他病理类型癌1例。
乳腺癌无骨转移组:51例,女性,年龄31~69岁,为在我院门诊或住院治疗的乳腺癌患者,包括浸润性导管癌26例、浸润性单纯癌及腺癌17例、浸润性小叶癌4例、髓样癌2例、其他病理类型2例。
所有患者均经临床、病理、血清肿瘤标志物检测以及影像学检查确诊,并排除其他系统良恶性肿瘤以及可能对骨显像和肿瘤标志物检测造成干扰的疾病。
-
按常规方法静脉注射显像剂99Tcm-MDP 740 MBq(20 mCi,由北京原子高科股份有限公司提供),3 h后行全身前、后位平面显像。图像采集使用日本东芝公司的GCA7100A/DI型SPECT仪,配低能高分辨准直器,采集条件为:能峰140 keV,能窗20%,矩阵256×1024,扫描速度15 cm/min。对骨显像阴性但骨痛明显患者再行CT或MRI,用以确诊。
-
由3位有经验的医师共同阅片,病变部位放射性高于或低于对侧或邻近正常骨组织视为异常分布。
-
受检者均空腹采集静脉血3 ml,室温放置1h后,分离血清。血清CA15-3、CEA检测采用德国罗氏公司Elecsys2010型电化学发光免疫分析仪及其配套试剂盒。CA15-3的正常参考值为0~25 kU/L,CEA的正常参考值为0~15 μg/L。
-
血清CA15-3、CEA数据以x±s表示,采用SPSS 10.0软件进行统计学分析,组间数据比较采用配对t检验,P < 0.05为差异有统计学意义。
-
3组患者全身骨显像和血清CA15-3、CEA水平测定结果见表 1,结果显示:乳腺癌骨转移组血清CA15-3、CEA水平明显高于无骨转移组和对照组,差异有统计学意义;乳腺癌无骨转移组血清CA15-3、CEA水平略高于对照组,差异无统计学意义。
例数 全身骨显像阳性率(%) CA15-3 CEA 实测值[(x±s) kU/L] 阳性率(%) 实测值[(x±s)μg/L] 阳性率(%) 良性病变组 45 6.67(3/45) 13.41±4.26 2.22(1/45) 6.7±3.1 6.67(3/45) 乳腺癌骨转移组 46 95.65(44/46) 95.51±32.93△ 76.09(35/46) 58.6±20.4△△ 80.43(37/46) 乳腺癌无骨转移组 51 13.73(7/51) 17.31±6.91* 17.65(9/51) 9.5±4.8** 11.76(6/51) 注:乳腺癌骨转移组与无骨转移组比较,△:t=2.891,P < 0.01;△△:t=2.952,P < 0.01。乳腺癌骨转移组与良性病变组比较,△:t=3.125,P < 0.01;△△:t=3.248,P < 0.01;乳腺癌无骨转移组与良性病变组比较,*:t=1.694,P > 0.05;**:t=1.681,P > 0.05。 表 1 乳腺癌骨转移组、无骨转移组和良性病变组患者全身骨显像和血清CA15-3、CEA水平测定结果
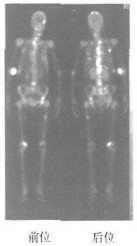
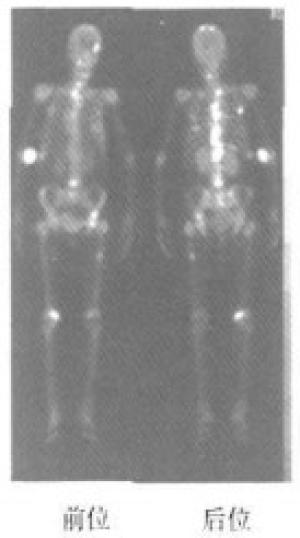
乳腺癌骨转移组患者大多数全身骨显像结果为阳性(图 1),仅2例为阴性,骨显像阴性患者全身核素分布未见明显异常,但是患者骨痛明显,结合CT或MRI检查发现骨转移;无骨转移组有7例发现核素异常浓聚,其中5例为术后改变,2例为外伤引起;良性病变组3例放射性异常浓聚均由外伤引起,该3例患者的血清CA15-3、CEA水平在正常范围;骨转移组全身骨显像阴性的2例患者,血清CA15-3、CEA水平明显高于正常范围;无骨转移组全身骨显像阳性的7例患者中,有2例CA15-3、CEA水平轻度增高,5例在正常范围。
血清CA15-3、CEA水平测定和全身骨显像联检对乳腺癌骨转移的诊断价值见表 2。结果显示,虽然CA15-3+CEA两项联检诊断乳腺癌骨转移的特异度较单项肿瘤标志物检测时低,但其灵敏度和准确率却明显提高;而CA15-3+CEA+全身骨显像三项联检比CA15-3+CEA两项联检在诊断乳腺癌骨转移中的灵敏度、特异度和准确率均明显提高。
诊断指标 灵敏度 特异度 准确率 全身骨显像 95.65(44/46) 89.85(86/96) 91.55(130/142) CA15-3 76.09(35/46) 89.85(86/96) 85.21(121/142) CEA 80.43(37/46) 90.63(87/96) 87.32(124/142) CA15-3+CEA 93.45(43/46) 88.54(85/96) 90.14(128/142) CA15-3+CEA+全身骨显像 100(46/46) 95.83(92/96) 97.18(138/142) 表 2 血清CA15-3、CEA水平和全身骨显像联检对乳腺癌骨转移的诊断价值
(%) -
乳腺癌为亲骨性肿瘤,许多患者无骨痛症状时已有转移,一旦出现明显症状,则多数已处于病程的晚期。因此,早期判断乳腺癌有无骨转移,对乳腺癌患者的临床分型、治疗方案制定、预后判断等有十分重要的临床意义。本研究中乳腺癌患者骨转移率为47.42%(46/97)。
血清肿瘤标志物是指肿瘤组织和细胞内由于癌基因及其产物的异常表达所产生的抗原和生物活性物质,其能反映肿瘤发生过程中相关基因的变化,现已经成为临床常规肿瘤的诊断方法,但其含量在不同类型肿瘤中存在较大差异,临床多采用针对相应肿瘤的、特异性较高的多项肿瘤标志物联合检测来提高诊断的灵敏度和特异度[2]。CA15-3是一种大分子糖蛋白,主要存在于正常乳腺上皮管腔面,细胞恶变时其含量明显增多,是一种特异性较高的标志物。本研究结果显示,有骨转移的乳腺癌患者血清CA15-3水平明显升高,提示CA15-3对乳腺癌骨转移具有较高的诊断价值[3]。
CEA属胚胎性抗原,只在胚胎期产生,其分泌受胚胎细胞的有关基因调控,健康成人血清中很少检测到,CEA主要用于诊断有内胚层分化而来的恶性肿瘤,对腺癌的诊断价值较大。本研究结果显示,乳腺癌患者CEA水平在骨转移后明显增高,表明监测血清CEA水平变化对评价疗效、监测肿瘤复发及转移有一定的临床意义[4]。
99Tcm-MDP全身骨显像能早期反映骨骼的病理变化,甚至当骨组织代谢发生改变时即可发现病灶,因此对转移性骨肿瘤有很高的灵敏度。其机制可能是因为骨组织被肿瘤侵犯时,肿瘤部位代谢旺盛,血流加快,破骨和成骨细胞活跃,导致骨显像剂沉积增多,在病变区呈放射性浓聚。由于X线片诊断骨肿瘤的基础是骨小梁被肿瘤侵犯后引起脱钙而致局部解剖密度差异,一般认为骨骼发生溶骨性损伤,病变局部需脱钙50%以上时X线片才能发现病变局部密度或形态的变化;而且,由于X线摄片有时受摄片范围的限制,难免会遗漏拍摄范围以外的骨转移灶。而一次全身骨显像即可发现不同部位的多个病灶,对于进一步确定X线摄片部位具有一定的参考价值。本研究中,骨转移组病例全身骨显像多数为阳性(占95.65%),说明99Tcm-MDP全身骨显像对骨转移的早期诊断具有重要价值。
全身骨显像对于多个放射性浓聚灶不难作出骨转移的诊断,一般认为骨的良性病变很少呈现典型的多灶性。少数非转移性浓聚的常见原因有生理变异、病理因素、骨折、创伤、感染和手术等因素,且常表现为单发病灶,因此,判断骨显像示单个病灶的病例是否为假阳性,对于治疗方案的确定具有重要价值。本研究中的骨转移组病例全身骨显像有2例为阴性(4.35%),血清CA15-3、CEA水平测定疑似骨转移,后经CT或MRI确诊,说明骨显像结合血清CA15-3、CEA水平测定,能够提高骨转移诊断的准确性。另外,肿瘤标志物检测还有助于鉴别诊断,良性病变组3例骨显像阳性者血清CA15-3、CEA水平均在正常范围,而乳腺癌未转移组7例阳性者中有2例血清CA15-3、CEA水平轻度增高,5例在正常范围内。
研究结果表明,乳腺癌骨转移组血清CA15-3、CEA水平明显高于乳腺癌无骨转移组和良性病变组,乳腺癌无骨转移组血清CA15-3、CEA水平略高于良性病变组,但差异无统计学意义,说明血清CA15-3、CEA水平增高程度与乳腺癌骨转移具有相关性,提示乳腺癌发生骨转移,应尽快进行全身骨显像,及时调整患者治疗方案;但血清CA15-3、CEA水平在乳腺癌早期较低,早期诊断意义不大。本研究提示,全身骨显像与血清CA15-3、CEA联合检测的临床实用性强,可有效提高乳腺癌骨转移诊断的敏感性和特异性。
血清CA15-3、CEA检测联合全身骨显像在乳腺癌骨转移中的诊断价值
The value of combined examination of serum CA15-3, CEA level and whole body bone scan in the diagnosis of bone metastasis in breast cancer
-
摘要:
目的 评价血清肿瘤标志物糖类抗原15-3(CA15-3)、癌胚抗原(CEA)检测联合全身骨显像在乳腺癌骨转移诊断中的价值。 方法 对97例乳腺癌患者(其中,骨转移组46例、无骨转移组51例)及45例良性乳腺疾病患者(良性病变组)行SPECT全身骨显像,对骨显像阴性但骨痛明显患者,再行CT或MRI检查以确诊。同时,用电化学发光法测定其血清CA15-3、CEA水平,并将两者的结果进行比较。 结果 骨转移组CA15-3、CEA水平明显高于无骨转移组和良性病变组,两项指标诊断骨转移的阳性率分别为76.09%、80.43%;骨转移组全身骨显像44例为阳性(占95.65%),仅2例为阴性(占4.35%),该2例骨显像阴性患者再经CT或MRI确诊为骨转移;无骨转移组中的7例及良性病变组中的3例骨显像为阳性,均由手术或外伤引起。CA15-3+CEA+全身骨显像三项联检比CA15-3+CEA两项联检及各单项检查在诊断乳腺癌骨转移中的灵敏度、特异性和准确率均明显提高。 结论 全身骨显像和CA15-3、CEA联合检测有助于提高乳腺癌骨转移的检出率。 Abstract:Objective To explore the value of combined examination of serum tumormarkers carbohydrate antigen 15-3 (CA15-3), carcinoembryonic antigen (CEA) and whole body bone scan in the diagnosis of bone metastasis in breast cancer. Methods Whole body bone scan and serum CA15-3 and CEA levels with a electrochemical luminescence assay were performed in 97 patients with breast cancer (46 cases with bone metastasis and 51 cases without bone metastasis) and 45 patients with benign breast diseases. As for the negative cases who had significant pains in bones, CT or MRI was performed to make sure. Results The serum level of CA15-3 and CEA were significantly higher in patients with bone metastasis than those in patients without bone metastasis and the benign lesions. The positive predicting values were 76.09% and 80.43%. Most patients with bone metastasis had positive results in bone scan(95.65%), only 2 cases had negative results (4.35%), which is positive by CT or MRI Seven. Seven patients without bone metastasis and Three patients with the benign lesions had positive results in bone scan, that may be caused by previous operation or injury. The combined determination of CA15-3, CEA and whole body bone scan had a better performance in sensitivity, specificity and accuracy than each single way. Conclusion The combined determination of CA15-3, CEA and whole body bone scan were valuable in the diagnosis of bone metastasis in breast cancer. -
表 1 乳腺癌骨转移组、无骨转移组和良性病变组患者全身骨显像和血清CA15-3、CEA水平测定结果
例数 全身骨显像阳性率(%) CA15-3 CEA 实测值[(x±s) kU/L] 阳性率(%) 实测值[(x±s)μg/L] 阳性率(%) 良性病变组 45 6.67(3/45) 13.41±4.26 2.22(1/45) 6.7±3.1 6.67(3/45) 乳腺癌骨转移组 46 95.65(44/46) 95.51±32.93△ 76.09(35/46) 58.6±20.4△△ 80.43(37/46) 乳腺癌无骨转移组 51 13.73(7/51) 17.31±6.91* 17.65(9/51) 9.5±4.8** 11.76(6/51) 注:乳腺癌骨转移组与无骨转移组比较,△:t=2.891,P < 0.01;△△:t=2.952,P < 0.01。乳腺癌骨转移组与良性病变组比较,△:t=3.125,P < 0.01;△△:t=3.248,P < 0.01;乳腺癌无骨转移组与良性病变组比较,*:t=1.694,P > 0.05;**:t=1.681,P > 0.05。 表 2 血清CA15-3、CEA水平和全身骨显像联检对乳腺癌骨转移的诊断价值
(%) 诊断指标 灵敏度 特异度 准确率 全身骨显像 95.65(44/46) 89.85(86/96) 91.55(130/142) CA15-3 76.09(35/46) 89.85(86/96) 85.21(121/142) CEA 80.43(37/46) 90.63(87/96) 87.32(124/142) CA15-3+CEA 93.45(43/46) 88.54(85/96) 90.14(128/142) CA15-3+CEA+全身骨显像 100(46/46) 95.83(92/96) 97.18(138/142) -
[1] Parkin DM, Bray F, Ferlay J, et al. Global cancer statistics. CA Cancer J Clin, 2005, 55(2): 74-108. doi: 10.3322/canjclin.55.2.74 [2] 邹雄. 肿瘤标志在肿瘤早期诊断中的研究与应用进展. 中华检验医学杂志, 2002, 25(2) : 71-72.
[3] 郭旭霞, 段满乐, 杜肖刚, 等. 蛋白质芯片技术检测肿瘤标志物及临床应用. 临床检验杂志, 2004, 22(3) : 224.
[4] 刘晨梅, 侯敏, 张连祥, 等. 肺癌患者血清和胸水中肿瘤标志物联合检测的意义. 临床检验杂志, 2005, 23(2): 150.
-





 下载:
下载: