-
前列腺癌是中老年男性常见的恶性肿瘤之一, 近年来其发病率在我国呈上升趋势。由于早期多无症状或症状不明显, 50%~80%的前列腺癌患者就诊时已经发生广泛转移, 失去了根治性手术的机会[1]。核素全身骨显像是目前诊断前列腺癌骨转移较好的方法, 前列腺特异性抗原(prostate-specific antigen, PSA)在前列腺癌的诊断和疗效观察等方面也具有重要的临床意义。因此, 我们对经病理证实的107例前列腺癌患者的血清PSA水平、病理分级和全身骨显像结果进行了回顾性分析, 旨在探讨全身骨显像诊断前列腺癌骨转移与血清PSA水平及病理分级的关系, 并研究前列腺癌发生骨转移的规律和特点。
-
2007年1月至2009年12月由我院收治的前列腺癌患者107例, 所有病例均经穿刺细胞学或手术病理证实。根据全身骨显像结果将患者分为骨转移组(49例)和非骨转移组(58例), 其中骨转移组年龄为49~86岁, 平均72.1岁; 非骨转移组年龄为59~87岁, 平均70.6岁。两组患者在年龄上的差异无统计学意义。
-
采用美国GE公司双探头MG型SPECT仪, 配低能高分辨准直器。99Tcm-MDP由原子高科股份有限公司提供, 标记率大于98%。经患者肘静脉注射925 MBq(25 mCi)99Tcm-MDP, 3~5 h后行全身骨前后位静态相, 扫描速度为15 cm/min, 必要时加局部静态相, 可疑部位可行局部采集, 局部采集500×103~1000×103的计数。由2位有经验的核医学医师分别阅片, 凡病变部位放射性浓集区高于健侧者诊断为阳性, 并排除骨良性病变所致的假阳性。
-
所有患者均空腹取静脉血3 ml并分离血清, 于-20℃冰箱中冻存, 采血前2周避免前列腺直肠指诊和膀胱镜检查, 并停用非那雄胺片。血样采集均先于骨显像, 但时间不超过8 d。采用美国宝特ELX-800NB型酶标仪, 放免试剂盒为瑞典康乃格诊断公司生产, 严格按说明书进行操作。血清PSA的正常范围为0~4 μg/L。
-
全部标本(前列腺穿刺活检、经尿道切除的组织或根治性前列腺切除组织)用10%甲醛液固定, 石蜡包埋后连续切片, 进行常规苏木精-伊红染色。根据染色切片中肿瘤的组织学形态, 按1992年修订的Gleason分级和评分标准进行病理分级, 根据分化程度的高低将所有患者分为高分化癌组(2~4分)、中分化癌组(5~7分)及低分化癌组(8~10分)3组。
-
采用SPSS13.0统计学软件对骨转移组和非骨转移组的PSA水平进行t检验, 组间的比较使用方差分析, 计数资料的比较采用χ2检验, P > 0.05为差异无统计学意义。
-
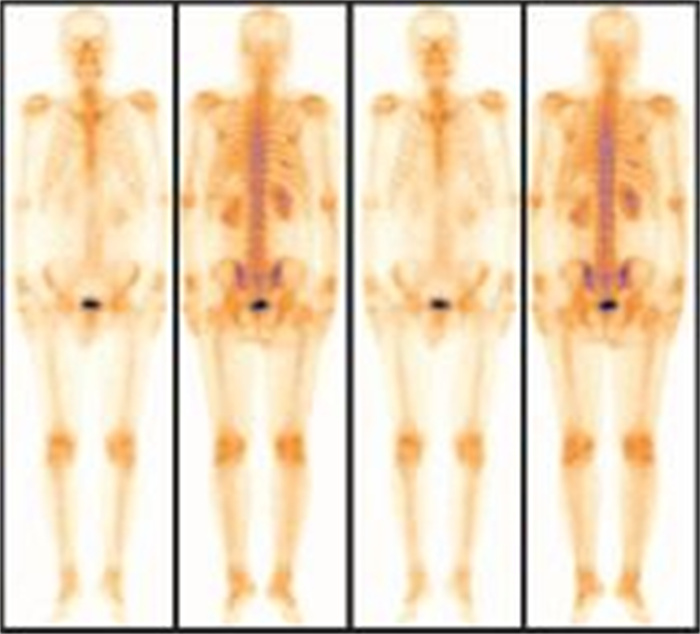
99Tcm-MDP全身骨显像发现前列腺癌骨转移49例, 占45.8%(49/107)。其中, 单个病灶或两个病灶者9例, 占18.4%(9/49)(图 1); 多个(≥3个)病灶者40例, 占81.6%(40/49)(图 2)。全身骨显像诊断骨转移的灵敏度为91.2%, 特异度为84.3%。
-
前列腺癌骨转移组与非骨转移组的PSA平均值分别为(120.3±12.3)μg/L和(28.6±9.3)μg/L, 前者显著高于后者(t=9.871, P < 0.01)。根据血清PSA的水平, 比较各个不同临界值对骨转移诊断的灵敏度、特异度、阳性预测值、阴性预测值和准确率, 结果见表 1。通过表 1发现, 以PSA > 20 μg/L为诊断临界值时, PSA≤20 μg/L和 > 20 μg/L两组诊断骨转移的差异有统计学意义(χ2=9.62, P < 0.01), 灵敏度达89.6%, 准确率达87.1%, 但特异度为59.1%;当以PSA > 40 μg/L为诊断临界值时, PSA≤40 μg/L和 > 40 μg/L两组诊断骨转移的差异具有统计学意义(χ2=8.69, P < 0.01), 灵敏度达79.2%, 准确率达82.3%, 特异度为91.6%, 有诊断骨转移的价值; 当以PSA > 60 μg/L为诊断临界值时, 特异度达100%, 表示PSA水平高于该值的患者几乎都已发生骨转移。
前列腺特异性抗原(μg/L) > 20 > 40 > 60 > 100 灵敏度 89.6 79.2 69.1 50.9 特异度 59.1 91.6 100 100 阳性预测值 90.1 95.3 100 100 阴性预测值 68.2 57.1 40.9 31.9 准确率 87.1 82.3 76.2 64.2 表 1 不同前列腺特异性抗原水平诊断骨转移发生的灵敏度、特异度、阳性预测值、阴性预测值和准确率(%)
-
根据前列腺癌原发病灶的病理分级进行分组后分析各自的骨转移发生率结果见表 2, 其中, 高分化组骨转移发生率为28.6%;中分化组为45.5%;低分化组为56.5%, 3组之间骨转移发生率比较, 差异有统计学意义(χ2=15.41, P < 0.01)。对上述3组之间骨转移发生率作两两比较: 低分化组分别与高分化组、中分化组相比, 差异有统计学意义(χ2=10.78和χ2=9.69, P均 < 0.01);而高分化组与中分化组相比, 差异无统计学意义(χ2= 1.33, P > 0.05)。
总例数 骨转移例数 骨转移发生率(%) 高分化组 28 8 28.6 中分化组 33 15 45.5 低分化组 46 26 56.5 表 2 前列腺癌病理分级与骨转移发生率的关系
-
随着人口寿命的延长、生活水平的提高, 前列腺癌的发病率可能会升高[2]。前列腺的静脉回流经前列腺静脉丛汇入髂内静脉, 与椎静脉下部有广泛交通, 椎静脉丛主要收集椎骨和骨髓的静脉血液, 亦是沟通上、下腔静脉的主要途径之一[3], 这构成了前列腺癌骨转移的解剖基础。目前, 骨转移的诊断主要通过核素全身骨显像和X线平片。核素全身骨显像可以比X线平片早3~6个月发现骨转移病变[4]。由于前列腺癌的骨转移绝大多数是成骨性转移, 全身骨显像对早期诊断骨转移有很高的临床价值。有文献报道, 24%~35%的前列腺癌患者在初诊时就已经有骨转移的征象[5], 骨显像对前列腺癌最初的分期、监测治疗的反应和评估骨痛也是很有价值的。本研究中, 107例前列腺癌患者在手术前行99Tcm-MDP全身骨显像, 发现49例骨转移, 阳性率为45.8%, 其中多发性骨转移占81.6%, 因此, 本研究以全身骨显像结果作为诊断骨转移的金标准。
PSA是仅由前列腺上皮分泌的一种糖蛋白, 具有中性丝氨酸蛋白酶活性, 有较高的特异性。目前临床上将其在血清中的质量浓度作为诊断前列腺癌的一种较有效的方法。本研究结果发现, PSA对早期发现及诊断骨转移有其特有的价值, 当PSA < 20 μg/L时, 骨转移的发生机会极小。Chybowski等[6]总结了512例未经治疗的前列腺癌患者的资料后认为, PSA < 10 μg/L时, 全身骨显像的阳性率接近零。Schulman[7]分析了852例未经治疗的前列腺癌患者PSA检测结果发现, PSA < 20 μg/L时, PSA水平预测骨显像结果的假阴性率为0.8%。因此可以认为, PSA < 20 μg/L时, 全身骨显像的阳性率极低。从本研究结果可以看出, 当PSA > 20 μg/L时, 患者可能发生早期骨转移, 该临界值的灵敏度达到89.6%, 但特异度不高(59.1%), 故我们建议, 对于PSA > 20 μg/L的患者, 应常规行全身骨显像, 用以早期确诊前列腺癌骨转移。有文献分析认为, PSA > 50 μg/L时前列腺癌多已发生广泛浸润或转移[8]。对于PSA > 60 μg/L特别是PSA > 100 μg/L的患者, PSA水平诊断骨转移的特异度达到100%, 可以认为患者已存在骨转移, 此时全身骨显像的诊断价值已不大。
前列腺癌中, 绝大多数为腺癌, 少数为鳞状上皮癌或移行上皮癌, 癌组织转移的发生与癌组织细胞分化的级别、癌组织的体积有明显的关系[9]。对前列腺癌的病理研究结果表明, 肿瘤细胞分化程度越低, 其恶性程度越高, 越具侵袭能力, 癌细胞更易入血而造成全身转移[10-11]。在多种化学因子、细胞因子及膜蛋白的共同参与和作用下, 骨骼是前列腺癌发生远处转移最常见的靶器官。本研究中前列腺癌患者的病理类型均为腺癌, 对不同分化组之间的骨转移发生率进行比较分析显示, 它们之间差异有统计学意义(χ2=15.41, P < 0.01)。与高分化组患者、中分化组患者相比, 低分化组患者骨转移发生率显著升高, 而高分化组与中分化组之间差异无统计学意义(χ2=1.33, P > 0.05), 提示随着前列腺癌分化程度降低, 患者更易发生骨转移。因此, 当前列腺疾病患者病理证实为低分化前列腺癌时, 应定期进行骨显像, 以期早期发现骨转移灶并进行治疗。
总之, 通过本研究我们发现, 前列腺癌患者骨转移发生率与前列腺癌的分化程度、血清PSA水平有一定的关系。前列腺癌分化程度越低, 骨转移发生率越高。对于初诊而未治疗的前列腺癌患者, PSA < 20 μg/L者应密切随访PSA水平的变化; PSA > 20 μg/L者应行骨显像以确诊; PSA > 60 μg/L者可临床诊断为骨转移。
全身骨显像诊断前列腺癌骨转移与PSA水平及病理分级的相关性研究
Study of relationship between the bone imaging, level of PSA and pathology grade in diagnosis of prostate carcinoma bone metastasis
-
摘要:
目的 探讨核素全身骨显像诊断前列腺癌骨转移与血清前列腺特异性抗原(PSA)水平及前列腺癌病理分级的关系,并研究前列腺癌发生骨转移的规律和特点。 方法 对107例前列腺癌患者术前用放免法测定其血清PSA水平,并进行99Tcm-亚甲基二膦酸盐全身骨显像,术后对其进行病理分级,分析前列腺癌骨转移与3种方法检查结果的相关性。 结果 107例前列腺癌患者全身骨显像发现49例发生骨转移,占45.8%(49/107),其中,不同病理分组之间的骨转移发生率差异有统计学意义(χ2=15.41,P < 0.01),前列腺癌分化程度越低,骨转移发生率越高;随着血清中PSA水平的升高,骨转移的发生率明显增加,PSA > 20μg/L者有骨转移可能,PSA > 60μg/L者骨转移的可能性极大。 结论 前列腺癌患者骨转移发生率与前列腺癌的分化程度、血清PSA水平有一定的关系。低分化者或PSA > 20μg/L者,骨转移发生率高。对于初诊的前列腺癌患者,PSA < 20μg/L者应密切随访其PSA水平变化;PSA > 20μg/L者应行骨显像以确诊;PSA > 60μg/L者临床可诊断为骨转移。 Abstract:Objective To investigate the relationship between the level of serum prostate-specific antigen(PSA), pathology grade and bone imaging in diagnosis of bone metastasis in prostatic carcinoma. Methods Serum PSA, whole body bone imaging with99Tcm-methylene diphosphonate, pathology grade in 107 cases of prostatic carcinoma were retrospectively reviewed as to their correlation with bone metastasis. Results Fourty-nine of 107 cases demonstrated bone metastasis by whole body bone imaging.There were significant difference in bone metastasis incidence rate among high differentiation group, middle differentiation group and low differentiation group.The results showed that when PSA < 20 μg/L, the possibility of bone metastasis was extremely low and follow-up studies were reuired.When serum PSA > 20 μg/L, bone metastasis should be considered and bone imaging was necessary. Conclusions There were some relationship among the level of PSA, pathology grade and bone metastasis.The possibility of bone metastasis became high when PSA > 20 μg/L.If PSA > 60 μg/L, a clinical diagnosis of bone metastasis might be made. -
表 1 不同前列腺特异性抗原水平诊断骨转移发生的灵敏度、特异度、阳性预测值、阴性预测值和准确率(%)
前列腺特异性抗原(μg/L) > 20 > 40 > 60 > 100 灵敏度 89.6 79.2 69.1 50.9 特异度 59.1 91.6 100 100 阳性预测值 90.1 95.3 100 100 阴性预测值 68.2 57.1 40.9 31.9 准确率 87.1 82.3 76.2 64.2 表 2 前列腺癌病理分级与骨转移发生率的关系
总例数 骨转移例数 骨转移发生率(%) 高分化组 28 8 28.6 中分化组 33 15 45.5 低分化组 46 26 56.5 -
[1] Armstrong AJ, Tannock IF, de Wit R, et al. The development of risk groups in men with metastatic castration-resistant prostate cancer based on risk factors for PSA decline and survival. Eur J Cancer, 2010, 46(3): 517-525. doi: 10.1016/j.ejca.2009.11.007 [2] 罗勇, 张林琳, 贺大林. 前列腺癌的骨转移倾向. 国外医学泌尿系统分册, 2004, 24(2): 145-149.
[3] Chiu JS, Wang YF, Su YC, et al. Artificial neural network to predict skeletal metastasis in patients with prostate cancer. J Med Syst, 2009, 33(2): 91-100. doi: 10.1007/s10916-008-9168-2 [4] Gomes RR Jr, Buttke P, Paul EM, et al. Osteosclerotic prostate cancer metastasis to murine bone are enhanced with increased bone formation. Clin Exp Metastasis, 2009, 26(7): 641-651. doi: 10.1007/s10585-009-9263-x [5] Klotz L. Active surveillance for prostate cancer: a review. Curr Urol Rep, 2010, 11(3): 165-171. doi: 10.1007/s11934-010-0110-z [6] Chybowski FM, Bergstralh EJ, Oesterling JE. The effect of digital rectal examination on the serum prostate specific antigen concentration: results of a randomized study. J Urol, 1992, 148(1): 83-86. doi: 10.1016/S0022-5347(17)36517-5 [7] Schulman C. PSA: an update. Arch Ital Urol Androl, 2006, 78(4): 161-162. [8] Henry N, Sebe P, Cussenot O. Inappropriate treatment of prostate cancer caused by heterophilic antibody interference. Nat Clin Pract Urol, 2009, 6(3): 164-167. [9] Schilling D, Hennenlotter J, Munz M, et al. Interpretation of the prostate cancer gene 3 in reference to the individual clinical background: implications for daily practice. Urol Int, 2010, 85(2): 159-165. doi: 10.1159/000314078 [10] Maeda H, Yonou H, Yano K, et al. Prostate-specific antigen enhances bioavailability of insulin-like growth factor by degrading insulin-like growth factor binding protein 5. Biochem Biophys Res Commun, 2009, 381(3): 311-316. doi: 10.1016/j.bbrc.2009.01.096 [11] K. o. llermann J, Weikert S, Schostak M, et al. Prognostic significance of disseminated tumor cells in the bone marrow of prostate cancer patients treated with neoadjuvant hormone treatment. J Clin Oncol, 2008, 26(30): 4928-4933. doi: 10.1200/JCO.2007.15.0441 -






 下载:
下载: