-
肺隔离症(pulmonary sequestration,PS)是一种先天性肺部发育畸形,患者肺组织内无正常支气管血管束,但存在1根异常供血动脉[1],其发病率为1.1%~1.8%[2]。PS的临床表现缺乏特异性,因此常被误诊为肺脓肿、囊腺瘤样畸形或肺癌等其他肺部疾病[3],患者常无法得到及时有效的治疗。目前,18F-FDG PET/CT被广泛应用于肿瘤细胞的分子显像和代谢显像,对肿瘤的诊断和鉴别诊断有很大帮助,同机诊断性CT的应用可以更好地降低18F-FDG PET/CT显像的假阳性率和假阴性率。本文旨在探讨增强CT联合18F-FDG PET/CT在伴肿瘤标志物水平升高的PS患者中的应用并对其图像特征进行分析,以提高18F-FDG PET/CT诊断医师对该病的认识。
-
回顾性分析2007年10月至2020年12月于青岛大学附属青岛市中心医院经手术证实的6例PS患者的临床资料,其中男性2例、女性4例,年龄(49.8±17.5)岁。纳入标准:(1)经组织病理学检查结果诊断为PS;(2)肿瘤标志物水平升高;(3)行CT增强扫描和18F-FDG PET/CT显像,且期间未行任何治疗;(4)无其他部位占位性病变。排除标准:(1)有CT增强扫描禁忌证;(2)有18F-FDG PET/CT显像禁忌证;(3)18F-FDG PET/CT检查前接受过治疗;(4)有其他部位的占位性病变。6例患者中,临床表现为反复发作的发热伴咳嗽的患者3例,胸闷、胸痛患者3例。6例患者的肿瘤标志物[CA199、CA125、神经特异性烯醇化酶(NSE)]水平均有不同程度的升高,其中2例患者的CA199水平升高,分别为50.16、179.85 U/ml(正常值范围:0~37.00 U/ml);2例患者的CA125水平升高,分别为59.74、60.15 kU/L(正常值范围:0~35.00 kU/L) ;2例患者的神经特异性烯醇化酶(NSE)水平升高,分别为19.85、24.77 ng/ml(正常值范围:0~16.30 ng/ml)。所有患者均行18F-FDG PET/CT显像并于检查前签署了知情同意书。本研究符合《赫尔辛基宣言》的原则。
-
使用德国西门子公司生产的Biograph 16型 PET/CT仪和美国CTI 公司生产的RDS111 型回旋加速器及FDGNA合成模块生产的18F-FDG对所有患者进行显像。显像前患者禁食4~6 h以上, 注射18F-FDG前测量患者的身高、体重, 血糖水平控制在<11.1 mmol/L。患者按照3.70 MBq/kg的剂量静脉注射18F-FDG后, 于安静、避光的室内平卧1 h后行18F-FDG PET/CT显像,采集范围自颅顶至股骨中上段。采集条件:管电压120 kV,管电流50 mA,层厚5 mm,层间距3 mm,螺距0.75。采用三维模式进行PET扫描, 2.5 min/床位,共采集6~8个床位。图像采用有序子集最大期望值法进行迭代重建, 利用CT扫描数据对PET图像进行衰减校正, 将校正后的PET图像与CT图像融合, 分别得到横断面、矢状面和冠状面的PET图像、CT图像及PET/CT融合图像, 并得到最大密度投影(MIP)图像。
使用PET/CT的同机CT行胸部双期(动脉期、静脉期)增强扫描。患者行PET/CT常规显像后,在肘关节留置20 G套管针,使用双通道高压注射器(美国Bayer公司Medrad Incorporated Mark 7 Arterion)以3.0 ml/s的注射速率注入90 ml非离子型碘佛醇(350 mgI/ml,北京北陆药业股份有限公司)。扫描范围为胸廓入口处至肺底。扫描条件:管电压120 kV,管电流100 mA,层厚5 mm,重建层厚1.0 mm。图像分别进行二维和三维重建,以观察病灶的强化程度、强化方式及是否存在异常的供血动脉。
-
由2名具有副高以上职称的PET/CT诊断医师对所有患者的18F-FDG PET/CT和CT图像特征(包括病变位置、形态、密度、CT强化特点及18F-FDG代谢情况)进行独立分析,意见不一致时经协商达成一致。病灶内CT成像强化区域的18F-FDG也呈代谢增高则判断为CT增强方式与18F-FDG PET显像结果匹配,否则认为二者不匹配。CT的强化程度(病灶动脉期或静脉期与平扫期CT值的差值)包括轻度强化(CT值增加<20 HU);中度强化(20 HU≤CT值增加≤40 HU);重度强化(CT值增加>40 HU)。采用四点法[4]对18F-FDG代谢增高的程度进行评价,包括以下4种情况。无18F-FDG代谢增高:病灶的18F-FDG代谢程度≤肺本底;18F-FDG代谢轻度增高:病灶的18F-FDG代谢程度≤纵隔血池;18F-FDG代谢中度增高:病灶的18F-FDG代谢程度>纵隔血池;18F-FDG代谢重度增高:病灶的18F-FDG代谢程度明显>纵隔血池。
-
采用SPSS 24.0对数据进行统计学分析,符合正态分布的计量资料以
$\bar x\pm s $ 表示。 -
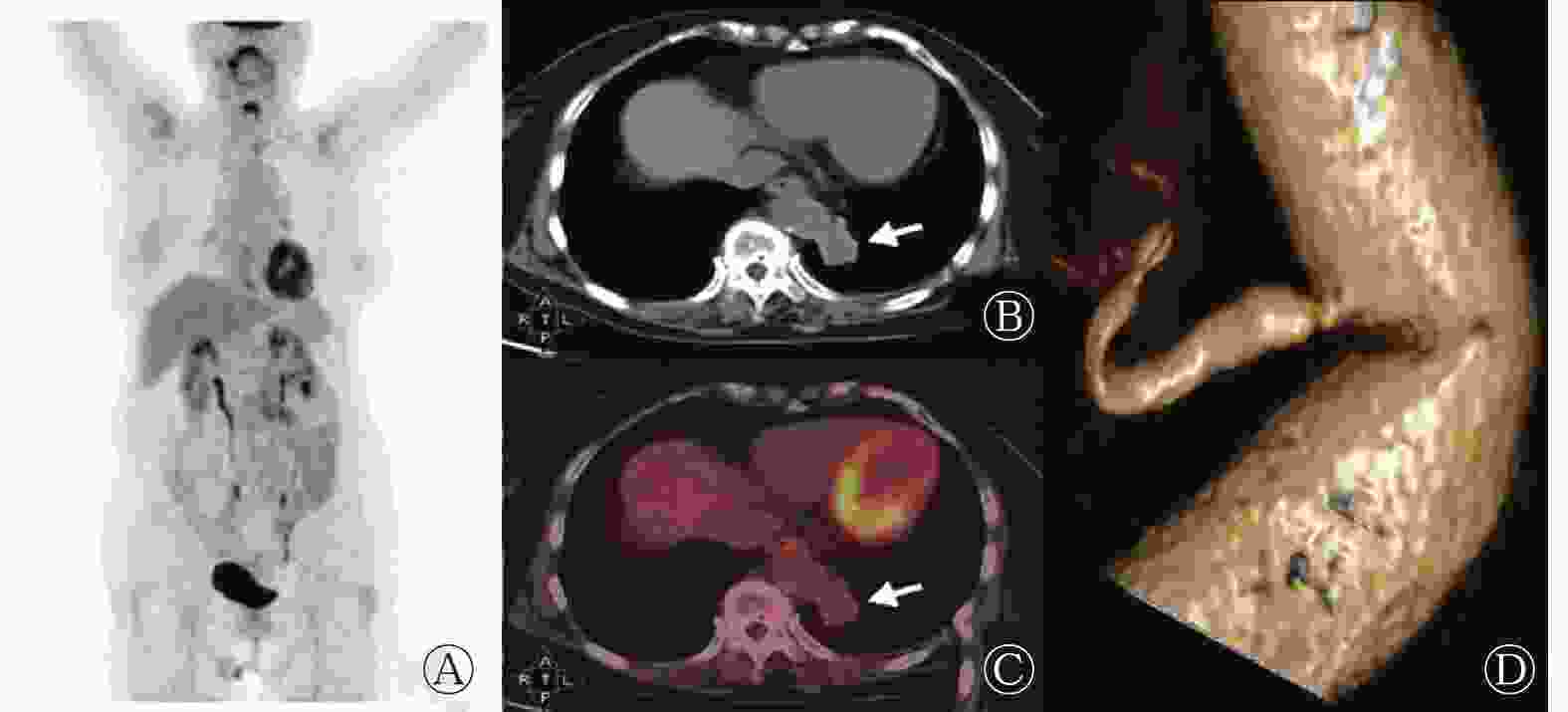
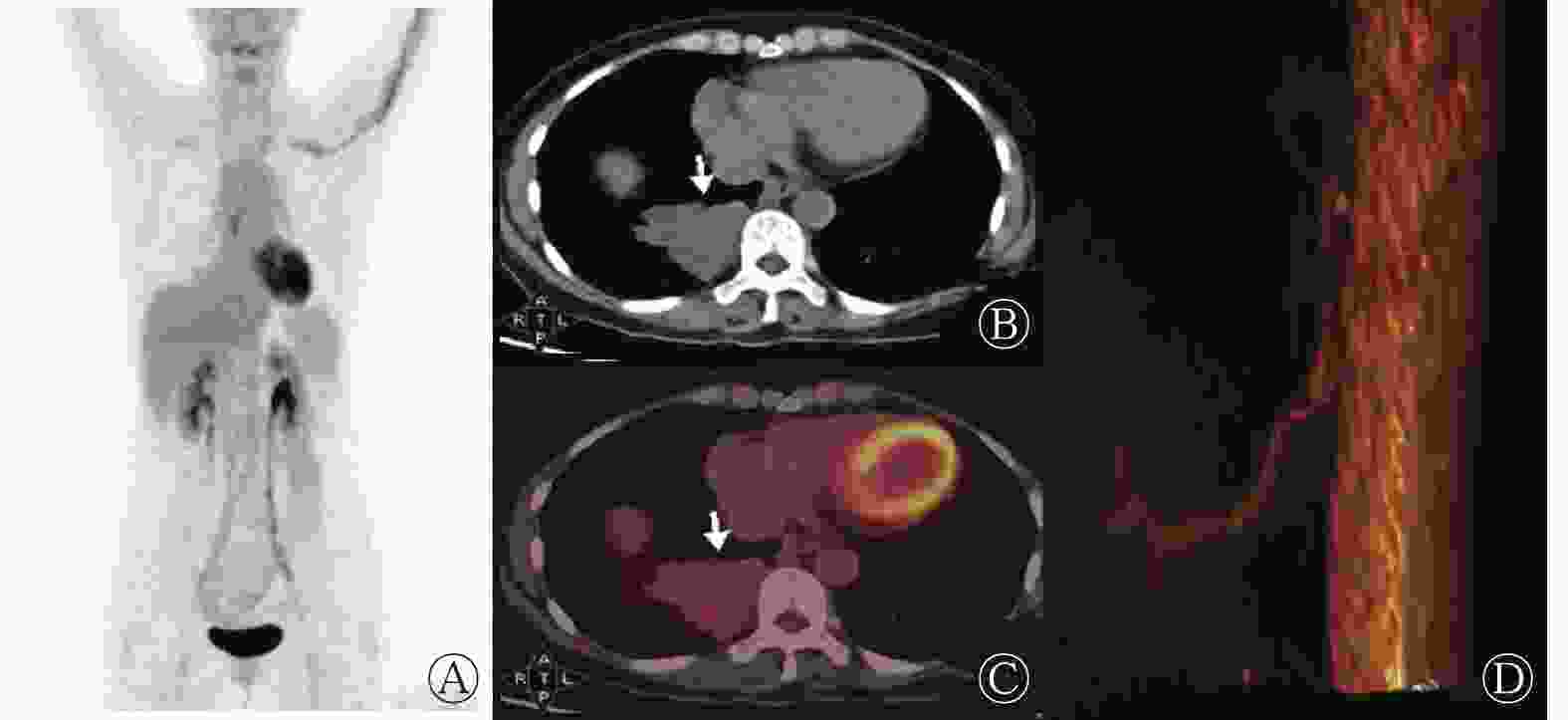
6例PS患者的病灶均位于肺下叶后基底段,其中右肺4例、左肺2例;病灶最大径为(4.3±2.0) cm;平扫CT值为(27.2±13.9) HU。病灶形态呈椭圆形的患者2例(图1)、类圆形2例(图2)、类三角形2例(图3)。1例患者的病灶内可见钙化灶。1例患者的病灶无明显强化、1例明显强化、4例中度强化。6例患者均存在起自胸主动脉的异常供血动脉。根据CT表现,囊性肿块型患者2例、实性肿块型患者3例、囊实性肿块型患者1例。局部肺叶内血管增多、增粗、紊乱患者3例。18F-FDG代谢中度增高患者2例、轻度代谢增高患者3例、无代谢增高患者1例,SUVmax为2.10~7.50。6例患者病灶CT的增强方式与18F-FDG PET显像均不匹配。6例患者的18F-FDG PET/CT显像均未发现肺外恶性肿瘤征象。

图 1 肺隔离症患者(女性,27岁)的18F-FDG PET/CT显像图
Figure 1. 18F-FDG PET/CT images of a patient (female, 27 years old) with pulmonary sequestration
-
PS是一种畸形的异常肺组织,其内无正常支气管血管束,但有1根来自体循环、胸主动脉或腹主动脉的异常供血动脉[5]。关于PS成因的假说较多,研究人员认为其与血管牵引异常、副肺芽、血管发育不全、异常动脉和肺囊肿等有关[6]。根据PS病灶是否位于同一脏层胸膜内[7],可将其分为肺叶内型和肺叶外型,其中前者占75%~85%,多数PS患者的病灶位于肺下叶较偏后部位,双侧PS较少见[8]。本研究中的6例患者均为肺叶内型PS且病灶均位于肺下叶后基底段。
PS的临床表现为反复感染、呼吸窘迫、咳嗽、痰中带血或无明显症状[9],其临床表现缺少特异性,影像学检查是其最主要的诊断方法。PS的CT表现为膈肌上区、肺下叶基底部、脊柱旁可见密度不均匀的软组织密度影,典型表现为软组织呈蜂窝状改变;增强CT扫描呈不均匀强化,实质部分强化明显。PS病灶主要为类圆形、椭圆形、类三角形的囊性、囊实性或实性肿块。本研究结果显示,2例囊性肿块、3例实性肿块、1例囊实性肿块患者的增强CT表现为实性区域或囊壁轻中度强化,与既往文献[9]报道一致。有文献报道,PS可引起肿瘤标志物(如 CA50、CA199、CA242等)水平的升高[10-11],但组织病理学检查结果均未发现恶性肿瘤,其原因可能是由于PS患者发育不良的肺组织内有大量炎症细胞的慢性浸润,慢性炎症的长期刺激促使肿瘤标志物释放入血,同时上皮细胞产生大量黏液,造成患者肿瘤标志物水平升高[7, 12]。本研究中6例患者的肿瘤标志物水平均有不同程度的地升高,其中1例患者的CA199水平高达179.85 U/ml。肿瘤标志物水平升高是行PET/CT检查的适应症之一。18F-FDG PET/CT是目前肿瘤诊断及分期最常用的一种无创性的检查手段,但存在假阳性的可能。有文献报道,PS患者病灶的18F-FDG PET/CT显像呈不均匀代谢增高,病灶周围18F-FDG代谢增高,病灶中心代谢较低,延迟显像的18F-FDG代谢无明显变化[13]。本研究结果显示,6例患者中,无18F-FDG PET/CT显像代谢增高患者1例、轻度代谢增高3例、中度代谢增高2例;且6例患者的CT增强方式与18F-FDG代谢方式不匹配。我们分析原因如下:发育不良的肺组织如不伴有炎症,其18F-FDG代谢不增高或轻度增高;如肺组织伴有炎症可表现为18F-FDG代谢明显增高,此时需结合增强CT确定有无来自体循环的供血动脉[14]。本研究中,所有患者均因发现了来自体循环的供血动脉而确诊,且与手术结果相吻合。有文献报道,PS的平均SUVmax为5.83±4.73,最高可达11.29[15]。本研究中除1例患者无明显18F-FDG代谢增高外,其余5例患者的SUVmax为2.10~7.50,其中1例患者的SUVmax高达7.5,与文献[15]的结果一致。
吴楠等[16]通过查阅文献发现有9例肺叶内型PS合并肺癌病例的报道,他们认为发生肺癌的原因可能与长期的炎症刺激导致病灶易发生癌变有关,但具体机制尚不明确。通过18F-FDG PET/CT可以鉴别PS是否合并肺癌并对其进行分期。Lee等[17]报道了1例误诊为PS的肺癌患者,最终通过18F-FDG PET/CT进行了明确的诊断。本研究中6例患者均通过手术证实未合并恶性肿瘤,且未发现肺外恶性肿瘤征象。
PS还需与以下疾病相鉴别。(1)肺癌:CT成像可见分叶、毛刺及胸膜凹陷征,肺门纵隔内淋巴结增大;18F-FDG PET/CT表现为18F-FDG代谢增高。(2)肺炎:临床表现为咳嗽、咳痰或伴发热;CT表现为边界欠清晰的片样影,可见支气管充气征,行抗感染治疗后病灶变化明显;18F-FDG PET/CT表现为18F-FDG代谢增高。(3)肺脓肿:病灶周围有严重的炎性改变,行抗炎治疗效果明显; 18F-FDG PET/CT表现为18F-FDG代谢增高。(4)肺囊腺瘤样畸形:CT表现为肺内呈大小不等的囊腔和结节样、蜂窝状小囊肿影,部分囊腔周围可见少量实性成分,占位效应较强,邻近肺组织含气不良,18F-FDG PET/CT表现为无18F-FDG代谢增高或18F-FDG代谢轻度增高。
综上所述,当双肺下叶近脊柱旁发现类圆形、椭圆形、类三角形结节或肿块,且伴有不同程度肿瘤标志物水平升高,18F-FDG代谢方式与CT增强方式不匹配时需结合增强CT寻找有无来自体循环的供血动脉。CT增强扫描联合18F-FDG PET/CT显像可以提高PS诊断的准确性。
利益冲突 所有作者声明无利益冲突
作者贡献声明 隋玉栋负责命题的提出与设计、论文的撰写;王清、高山、郭兴隆负责数据的采集、统计与分析;路月、林帅、张静负责文献资料的获取、图像的分析;王艳丽负责论文的指导、修订与审阅
增强CT联合18F-FDG PET/CT在肺隔离症诊断中的应用及其图像特征分析
Application and image feature analysis of enhanced CT combined with 18F-FDG PET/CT in diagnosis of pulmonary sequestration
-
摘要:
目的 探讨增强CT联合18F-FDG PET/CT在肺隔离症(PS)诊断中的应用并对其图像特征进行分析。 方法 回顾性分析2007年10月至2020年12月于青岛大学附属青岛市中心医院经手术证实伴有肿瘤标志物水平升高的6例PS患者的临床资料,其中男性2例、女性4例,年龄(49.8±17.5)岁。6例患者均行18F-FDG PET/CT显像及增强CT扫描,观察病变位置、形态、密度、CT强化特点及18F-FDG代谢情况。 结果 6例患者的病灶均位于肺下叶后基底段,其中右肺4例、左肺2例。病灶最大径为(4.3±2.0) cm,平扫CT值为(27.2±13.9) HU。病灶形态呈椭圆形、类圆形、类三角形的患者各2例;1例患者的病灶内可见钙化灶。1例患者的CT图像呈明显强化,4例呈中度强化,1例无明显强化。6例患者均发现了起自胸主动脉的异常供血动脉。囊性肿块型患者2例,实性肿块型患者3例,囊实性肿块型患者1例。局部肺叶内血管增多、增粗、紊乱患者3例。18F-FDG代谢中度增高患者2例、轻度增高患者3例、无代谢增高患者1例。 结论 当患者有不同程度的肿瘤标志物水平升高,双肺下叶脊柱旁发现类圆形、椭圆形、类三角形结节或肿块,18F-FDG代谢增高且CT增强扫描发现异常供血动脉并伴轻到中度强化或病灶无18F-FDG代谢增高且CT增强扫描无明显强化但发现异常供血动脉时,均要考虑PS的可能性。CT增强联合18F-FDG PET/CT显像可以提高PS诊断的准确性。 -
关键词:
- 肺肿瘤 /
- 氟脱氧葡萄糖F18 /
- 正电子发射断层显像术 /
- 计算机体层摄影术,X线计算机 /
- 肺隔离症
Abstract:Objective To explore the application of enhanced CT combined with 18F-FDG PET/CT in the diagnosis of pulmonary sequestration (PS) and analyze the image characteristics. Methods The clinical data of 6 patients with PS, including 2 males and 4 females, aged (49.8±17.5) years, who were surgically confirmed to be accompanied by elevated levels of tumor markers at the Affiliated Qingdao Central Hospital of Qingdao University from October 2007 to December 2020 were retrospectively analyzed.18F-FDG PET/CT imaging and enhanced CT scanning were performed in the 6 patients, and the location of the lesions, morphology, density, CT enhancement characteristics and 18F-FDG metabolism were observed. Results The lesions in the six patients were all located in the posterior basal segment of the lower lobe of the lung, including four cases in the right lung and two cases in the left lung. The maximum diameter of the lesion was (4.3±2.0) cm, and the CT value on plain scan was (27.2±13.9) HU. 2 patients had oval, rounded, or triangular-shaped lesions, and 1 patient had calcified foci within the lesion; 1 patient had marked enhancement on the CT image, 4 had moderate enhancement, and 1 had no marked enhancement. 6 patients were found to have an abnormal arterial blood supply originating from the thoracic aorta. Two patients had cystic masses, three patients had solid masses, and one patient had a cystic-solid mass. There were 3 patients with localized vascularization, coarsening, and disorganization in the lobes of the lungs, 2 patients with moderately increased 18F-FDG metabolism, 3 patients with mildly increased metabolism, and 1 patient with no metabolism increased metabolism. Conclusion The possibility of PS should be considered when patients have different degrees of elevated levels of tumour markers, round-like, oval-like, triangular-like nodules or masses found adjacent to the spine in the lower lobes of both lungs, increased 18F-FDG metabolism and abnormal blood-supplying arteries detected on CT-enhanced scans with mild-to-moderate enhancement, or when the lesion has no increased 18F-FDG metabolism and there is no significant enhancement on CT-enhanced scans, but abnormal blood-supplying arteries were detected. CT enhancement combined with 18F-FDG PET/CT imaging improves the accuracy of PS diagnosis. -
-
[1] Ryujin K, Akamine T, Miura N, et al. An adult case of multiple extralobar pulmonary sequestrations in the thoracic and abdominal cavities[J]. Ann Thorac Surg, 2022, 113(1): e17−e20. DOI: 10.1016/j.athoracsur.2021.03.083. [2] Martínez-Besteiro E, Eiros Bachiller JM, Solís García M, et al. Pulmonary sequestration[J]. Med Clin (Barc), 2022, 158(9): 449. DOI: 10.1016/j.medcli.2021.11.004. [3] Matsuoka H, Nohara H. Pulmonary sequestration with high levels of tumor markers tending to be misdiagnosed as lung cancer[J]. Jpn J Thorac Cardiovasc Surg, 2006, 54(3): 117−119. DOI: 10.1007/BF02744874. [4] Callister MEJ, Baldwin DR, Akram AR, et al. British thoracic society guidelines for the investigation and management of pulmonary nodules[J]. Thorax, 2015, 70(Suppl 2): Sii1−ii54. DOI: 10.1136/thoraxjnl-2015-207168. [5] Kong FW, Wang WM, Gong LB, et al. Extralobar pulmonary sequestration with elevated serum neuron-specific enolase: a case report and review of the literature[J]. Medicine (Baltimore), 2020, 99(40): e22574. DOI: 10.1097/MD.0000000000022574. [6] 孔煜, 熊佶, 樊洁, 等. 成人肺隔离症16例临床病理学分析[J]. 复旦学报(医学版), 2022, 49(3): 390−394. DOI: 10.3969/j.issn.1672-8467.2022.03.011.
Kong Y, Xiong J, Fan J, et al. Clinical and pathological features of adult pulmonary sequestration: an analysis of 16 cases[J]. Fudan Univ J (Med Sci), 2022, 49(3): 390−394. DOI: 10.3969/j.issn.1672-8467.2022.03.011.[7] Teng GJ, Nie XH, Wang DD. Association of pulmonary sequestration with elevated serum cancer antigen 125 levels: a case report[J]. J Int Med Res, 2020, 48(2): 300060520903871. DOI: 10.1177/0300060520903871. [8] Arjun P, Palangadan S, Haque A, et al. Intralobar sequestration[J]. Lung India, 2017, 34(6): 559−561. DOI: 10.4103/lungindia.lungindia_51_17. [9] Gabelloni M, Faggioni L, Accogli S, et al. Pulmonary sequestration: what the radiologist should know[J]. Clin Imaging, 2021, 73: 61−72. DOI: 10.1016/j.clinimag.2020.11.040. [10] Fu SZ, Wang HT. Pulmonary sequestration associated with a synchronous elevation of carbohydrate antigen 50 and 19-9: a case report[J]. Ann Transl Med, 2018, 6(11): 212. DOI: 10.21037/atm.2018.05.18. [11] Ge L, Wang S, Liu HR, et al. Intralobar pulmonary sequestration with aspergillus infection and elevated serum CA19-9 and CA242: a case report[J]. Transl Cancer Res, 2021, 10(2): 1169−1176. DOI: 10.21037/tcr-20-2434. [12] Chen Q, Lu QJ, Fei X, et al. Elevated tumor markers in a benign lung disease[J/OL]. J Cardiothorac Surg, 2021, 16(1): 308[2022-11-23]. https://cardiothoracicsurgery.biomedcentral.com/articles/10.1186/s13019-021-01688-4. DOI: 10.1186/s13019-021-01688-4. [13] Şen S, Kanlıoğlu Kuman N, Şentürk E, et al. Pulmonary sequestration with renal aplasia and elevated SUV level in PET/CT[J]. Case Rep Pulmonol, 2012, 2012: 276012. DOI: 10.1155/2012/276012. [14] 赵家宁, 张欲翔, 董倩, 等. 胸主动脉供血与腹主动脉供血肺隔离症的影像学表现对比分析[J]. 中国中西医结合影像学杂志, 2021, 19(5): 429−431. DOI: 10.3969/j.issn.1672-0512.2021.05.006.
Zhao JN, Zhang YX, Dong Q, et al. Comparison analysis of imaging features of pulmonary sequestration with blood supply from thoracic aorta and abdominal aorta[J]. Chin Imaging J Integr Tradit West Med, 2021, 19(5): 429−431. DOI: 10.3969/j.issn.1672-0512.2021.05.006.[15] Li XJ, He WL, Li JH, et al. Pulmonary sequestration associated with increased serum tumor markers and elevated standard uptake value level in PET/CT: a case report and literature review[J]. Medicine (Baltimore), 2018, 97(31): e11714. DOI: 10.1097/MD.0000000000011714. [16] 吴楠, 孙宇, 郑庆锋, 等. 叶内型肺隔离症的诊断、治疗及分子病理学特点[J]. 中华医学杂志, 2007, 87(37): 2627−2631. DOI: 10.3760/j.issn:0376-2491.2007.37.009.
Wu N, Sun Y, Zheng QF, et al. Diagnosis and treatment of intralobar pulmonary sequestration[J]. Natl Med J China, 2007, 87(37): 2627−2631. DOI: 10.3760/j.issn:0376-2491.2007.37.009.[17] Lee BF, Chang HY, Yan JJ, et al. Carcinoma of the lung misinterpreted as pulmonary sequestration on contrast CT but correctly identified on FDG PET/CT[J]. Clin Nucl Med, 2010, 35(5): 343−345. DOI: 10.1097/RLU.0b013e3181d6276d. -





 下载:
下载: