-
结直肠息肉包括腺瘤性息肉和非腺瘤性息肉,其中腺瘤性息肉是公认的结直肠癌的癌前病变,大约85%的结直肠癌都是由腺瘤发展而来的。结直肠腺瘤发展成浸润性腺癌的年转化率约为0.25%,但高风险腺瘤发展为浸润性腺癌的年转化率高达2.6%~5.7%[1],值得临床高度重视。早期发现结直肠腺癌或腺瘤(尤其是高风险腺瘤)对于结直肠癌的预防及治疗意义重大。据文献报道,18F-FDG PET/CT应用于肿瘤诊疗时,有1%~3%的患者会出现偶发胃肠道局灶性摄取增高灶[2],结直肠局灶性摄取增高对诊断结直肠癌及癌前病变有较高的灵敏度[3]。目前的相关研究大多按腺瘤的组织形态分类(管状腺瘤或绒毛管状腺瘤等)来进行对照研究[4],本研究综合纳入腺瘤的组织形态、大小及分级信息,初步探讨偶发18F-FDG PET/CT结直肠局灶性摄取增高对结直肠早期腺癌及高风险腺瘤的预测价值。
-
回顾性收集2016年1月至2020年6月在北京医院PET/CT中心因非结直肠病变行全身18F-FDG PET/CT显像并偶然发现结直肠局灶性摄取增高的56例患者[男性31例、女性25例,年龄(66±11)岁,范围38~84岁]的临床资料,包括一般资料、肠道病灶PET显像资料(包括病灶位置、SUVmax)、PET/CT检查后3个月内的结肠镜及组织病理学检查结果。结直肠局灶性摄取增高的诊断标准是通过视觉分析判断:高于正常肝脏的结节状局灶性摄取[5];若行延迟显像,则延迟显像需仍为位置固定的结节状局灶性摄取增高。纳入标准:(1)因非结直肠病变行全身18F-FDG PET/CT显像;(2)18F-FDG PET/CT显像中偶然发现结直肠局灶性摄取增高;(3)18F-FDG PET/CT检查后3个月内获得明确的结肠镜及组织病理学检查结果。排除标准:18F-FDG PET/CT检查前有明确的结直肠癌病史。本研究符合《赫尔辛基宣言》的原则。因本研究为回顾性临床研究,豁免签署患者知情同意书。
-
显像仪器为德国Siemens公司Biograph mCT PET/CT仪。显像前所有患者均至少禁食6 h以上,血糖控制在<11.1 mmol/L,静脉注射18F-FDG(北京原子高科股份有限公司,放射化学纯度>95%)5.18 MBq/kg。患者于注射18F-FDG前、注射18F-FDG后30 min和上检查床前分别口服0.6%碘海醇(通用电气药业上海有限公司)250、250和500 ml,注射18F-FDG后约60 min开始采集体部图像。先行CT平扫,范围为颅底部至股骨上段,扫描参数:电压120 kV,采用自动毫安秒技术,矩阵512×512,层厚3 mm。随后行PET扫描,患者体位保持不变,采用三维模式,矩阵128×128,体部采集5~7个床位,范围为颅底部至股骨上段,2 min/床位。其中30例患者于注射18F-FDG 后120 min左右行局部延迟显像(3 min/床位)。PET图像重建采用有序子集最大期望值法。采用Siemens TrueD工作站自带的软件在偶发结直肠局灶性摄取增高处勾画ROI,获得SUVmax。
-
收集患者在18F-FDG PET/CT检查后3个月内的结肠镜及组织病理学检查结果作为最终诊断标准,统计偶发结直肠局灶性摄取增高灶的病理类型。结直肠早期腺癌定义为浸润深度局限于黏膜及黏膜下层的任意大小的结直肠上皮性肿瘤,无论是否存在淋巴结转移[3]。高风险腺瘤定义为具备长径≥10 mm、有25%以上的绒毛成分、有高级别异型增生中的任一条的腺瘤[6]。分析18F-FDG PET/CT偶发结直肠局灶性摄取增高对结直肠早期腺癌及高风险腺瘤的诊断价值,并与非高风险腺瘤及非腺瘤性息肉进行比较。
-
采用IBM SPSS 24.0软件进行统计学分析。符合正态分布的计量资料以
$\bar x \pm s $ 表示,偏态分布的计量资料以M(Q1,Q3)表示,计数资料以例数(百分比)表示。正态分布的计量资料的两组间比较采用独立样本t检验(方差齐),多组间比较采用方差分析;偏态分布的计量资料的两组间比较采用Mann-Whitney检验,多组间比较采用Kruskal-Wallis检验。计数资料的比较采用卡方检验。勾画ROC曲线,分析SUVmax对结直肠早期腺癌及高风险腺瘤的诊断效能及最佳诊断临界值。P<0.05为差异有统计学意义。 -
56例患者中,18F-FDG PET/CT共发现74处结直肠局灶性摄取增高灶,结肠镜共发现140处阳性病灶。18F-FDG PET/CT显像前的临床诊断结果:恶性肿瘤37例(其中实体肿瘤31例、血液系统肿瘤6例)、疑诊肿瘤10例、癌胚抗原水平增高4例、健康查体5例。
-
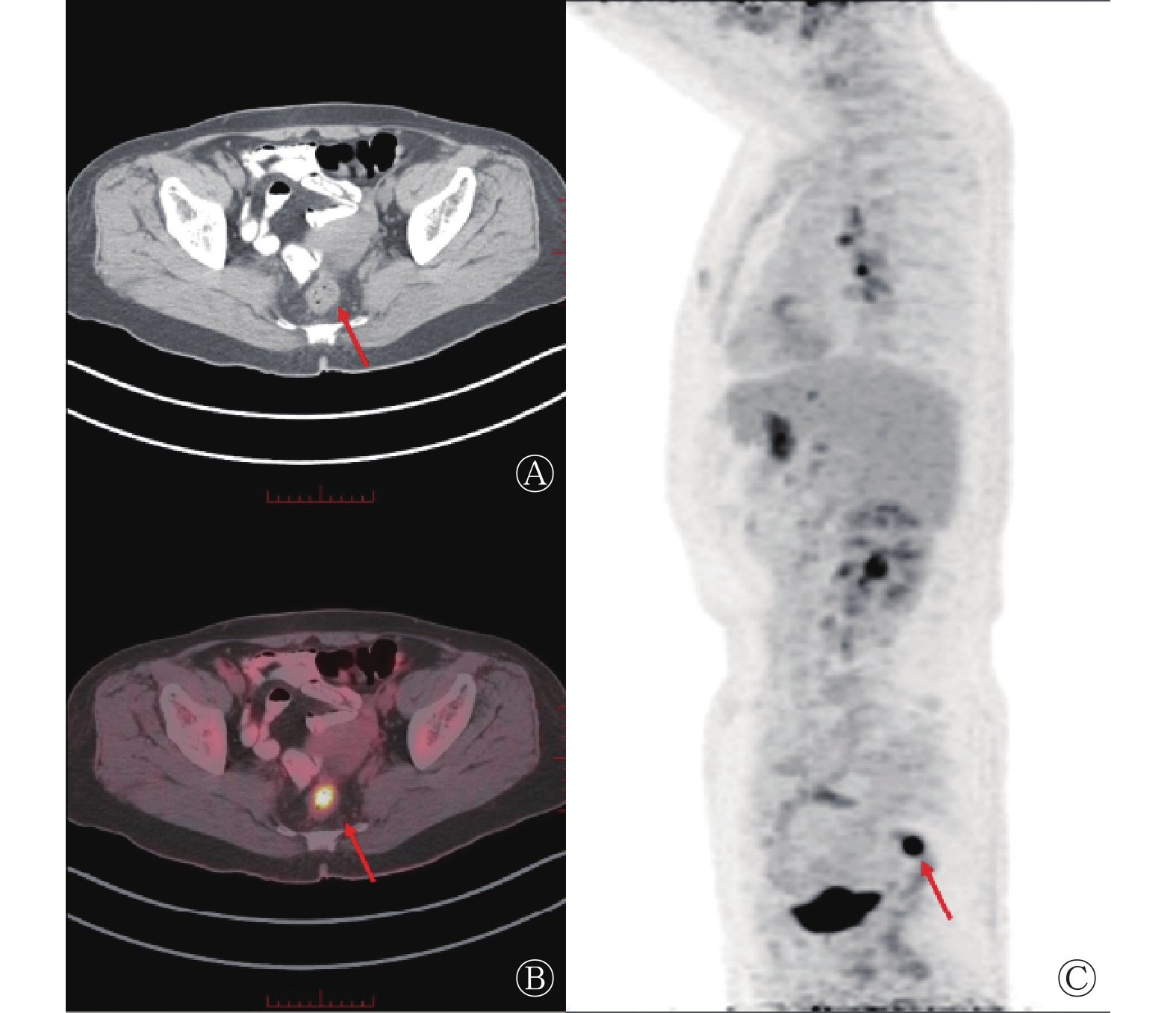
18F-FDG PET/CT发现的74处结直肠局灶性摄取增高灶(PET/CT阳性病灶)中,45处行延迟显像且仍表现为位置固定的结节状局灶性摄取增高;23处在同机CT平扫图像上表现为局部肠壁增厚或结节状软组织密度灶,其余51处在CT图像上均未见形态改变。74处结直肠局灶性摄取增高灶中,结肠镜阳性59处、阴性15处,结肠镜阳性病灶中,左半结肠44处、右半结肠15处;结肠镜结果显示,59处阳性病灶中,早期腺癌7处(11.9%,图1)、高风险腺瘤34处(57.6%,图2;其中绒毛管状腺瘤28处、长径≥10 mm 30处、伴高级别异型增生15处)、非高风险腺瘤5处(8.5%,图3)、非腺瘤性息肉8处(13.6%,其中错构瘤性息肉5处、幼年性息肉2处、增生性息肉1处)、转移灶3处(5.1%)、神经内分泌癌1处(1.7%)、憩室1处(1.7%)。结肠镜阳性者SUVmax显著高于阴性者,且差异有统计学意义[12.0(8.5,19.9)对7.8(5.3,10.7),Z=−2.65,P=0.008]。

图 1 胃MALT淋巴瘤患者(女性,67岁)治疗前18F-FDG PET/CT显像图
Figure 1. Pre-treatment 18F-FDG PET/CT images of a gastric MALT lymphoma patient (female, 67 years old)

图 2 腹部不适、腹泻数年患者(男性,53岁)的18F-FDG PET/CT显像图
Figure 2. 18F-FDG PET/CT images of a patient (male, 53 years old) with several years of abdominal discomfort and diarrhea

图 3 输尿管恶性肿瘤术后复发患者(男性,51岁)的18F-FDG PET/CT显像图
Figure 3. 18F-FDG PET/CT images of a patient (male, 51 years old) with recurrence after surgery for ureteral malignancy
18F-FDG PET/CT漏诊的81处病灶(PET/CT阴性病灶)中,左半结肠47处、右半结肠34处,结肠镜结果显示,高风险腺瘤13处(16.0%,其中绒毛管状腺瘤6处、长径≥10 mm 7处、伴高级别异型增生2处)、非高风险腺瘤45处(55.6%)、非腺瘤性息肉22处(27.2%,其中增生性息肉15处、炎性息肉7处)、转移灶1处。结肠镜显示,PET/CT阳性病灶与阴性病灶的长径分别为1.50(1.00,2.00)cm和0.40(0.30,0.60)cm,且差异有统计学意义(Z=−8.37,P<0.001)。
18F-FDG PET/CT结直肠局灶性摄取增高对结直肠早期腺癌和高风险腺瘤的阳性预测值为55.4%(41/74)、灵敏度为75.9%(41/54)。
-
由表1可知,早期腺癌、高风险腺瘤、非高风险腺瘤和非腺瘤性息肉的SUVmax的差异无统计学意义(F=0.78,P=0.511),18F-FDG PET/CT诊断灵敏度的差异有统计学意义(χ2=35.09,P<0.001),其中,18F-FDG PET/CT诊断高风险腺瘤的灵敏度明显高于非高风险腺瘤,且差异有统计学意义(χ2=39.20,P<0.001)。另外,腺瘤中绒毛管状腺瘤和管状腺瘤的SUVmax分别为17.1±10.4和12.3±8.3,二者的差异无统计学意义(t=−1.30,P=0.216),但18F-FDG PET/CT 诊断绒毛管状腺瘤的灵敏度明显高于管状腺瘤,且差异有统计学意义[82.4%(28/34)对14.8%(9/61),χ2=41.96,P<0.001]。
病理类型 病灶数
(处)SUVmax( $\bar x \pm s $ 

18F-FDG PET/CT
诊断灵敏度早期腺癌 7 15.4±7.7 100%(7/7) 高风险腺瘤 47 16.5±10.2 72.3%(34/47)a 非高风险腺瘤 50 7.7±0.6 10.0%(5/50) 非腺瘤性息肉 30 12.1±6.4 26.7%(8/30) 检验值 F=0.78 χ2=35.09 P值 P=0.511 P<0.001 注:a表示与非高风险腺瘤相比,差异有统计学意义(χ2=39.20,P<0.001)。SUVmax为最大标准化摄取值;FDG为氟脱氧葡萄糖;PET为正电子发射断层显像术;CT为计算机体层摄影术 表 1 不同病理类型的结直肠病灶的SUVmax及18F-FDG PET/CT诊断灵敏度比较
Table 1. Comparison of maximum standardized uptake value and sensitivity of 18F-FDG PET/CT among different pathologic types of colorectal lesions
ROC曲线分析结果(图4)显示,SUVmax诊断结直肠早期腺癌和高风险腺瘤的AUC为0.705(95%CI为0.551~0.858,P=0.027),最佳临界值为11.6,灵敏度为66.0%,特异度为76.0%。
-
长径≥10 mm的病灶与<10 mm病灶的SUVmax分别为16.3±9.1和9.6±7.5,二者的差异有统计学意义(t=2.246,P=0.029)。18F-FDG PET/CT诊断长径≥10 mm的病灶的灵敏度明显高于<10 mm的病灶,且差异有统计学意义(84.6%对13.3%,χ2=67.40,P<0.001)。
-
在一项对5850例18F-FDG PET/CT受检者的回顾性研究中,145例受检者(含180处病灶)意外发现了结直肠局灶性摄取增高灶,并于3个月内行结肠镜检查,结果显示18F-FDG PET/CT对高风险肿瘤性病灶的阳性预测值为47.8%[7]。另一项对1911例18F-FDG PET/CT肿瘤受检者的回顾性研究结果显示,5.2%的受检者中偶然发现了结直肠局灶性摄取增高灶,最终的结肠镜结果显示37.8%的摄取增高灶为腺癌和高风险腺瘤,且后续行结肠镜的受检者的总生存期高于未行结肠镜的受检者(38个月对14个月)[8]。本研究结果显示,偶发18F-FDG PET/CT结直肠局灶性摄取增高诊断结直肠早期腺癌和高风险腺瘤的阳性预测值为55.4%(41/74),与上述研究结果相似。关于18F-FDG PET/CT对结直肠早期腺癌和癌前病变的检出效能,不同研究报道的结果不尽相同,这可能与病灶的大小及类别有关。Hwang等[9]对614例18F-FDG PET/CT受检者的回顾性研究结果显示,18F-FDG PET/CT 对25例受检者的27处肠道局灶性摄取增高灶总的诊断灵敏度和特异度分别为5.6%和96.8%,但是对于≥1 cm的病灶,其诊断灵敏度为25.8%。本研究结果亦显示,18F-FDG PET/CT对长径≥10 mm的病灶的诊断灵敏度明显高于长径<10 mm的病灶(84.6%对13.3%,P<0.001),对高风险腺瘤的诊断灵敏度明显高于非高风险腺瘤(72.3%对10.0%,P<0.001)。Xu等[10]的研究结果显示,病灶大小可能是导致18F-FDG PET/CT漏诊结直肠高风险腺瘤的唯一因素。本研究结果显示,与结肠镜相比,虽然18F-FDG PET/CT有较高的漏诊率(57.9%,81/140),但其中大部分为非高风险腺瘤(45处,55.6%)及非腺瘤性息肉(22处,27.2%),说明18F-FDG PET/CT在检出高风险腺瘤上具有较高的价值,提示对于偶发18F-FDG PET/CT结直肠局灶性摄取增高灶,临床上需要给予更多的关注。
关于SUVmax对偶发结直肠局灶性摄取增高灶不同组织病理学类型间的鉴别诊断价值以及其对结直肠腺癌和高风险腺瘤诊断的最佳临界值,研究者们报道的结果不一。Alongi等[11]发现,结直肠癌前病变和腺癌的SUVmean显著高于炎症病灶和结肠镜阴性的病灶。Gilhotra 等[12]认为,局灶性且摄取显著增高的结直肠病灶通常与高风险息肉和恶性病灶相关。Chung等[13]认为,SUVmax>8是高风险腺瘤的独立危险因素。Purandare等[14]认为SUVmax>12.9是诊断结直肠腺癌的最佳临界值。一项较大样本量(7318例患者,在其中 359例患者中意外发现了404处结直肠局灶性摄取增高灶,最终242处病灶经结肠镜随诊结果证实,其中25例为腺癌、90例为腺瘤、127例为良性病灶)的回顾性研究结果显示,腺癌的SUVmax明显高于其他类型的病灶,但SUVmax不能区分腺瘤与良性病灶,该研究结果提示SUVmax ≥11.4时需要及时进行结肠镜检查[15]。本研究结果显示,早期腺癌、高风险腺瘤、非高风险腺瘤和非腺瘤性息肉的SUVmax的差异均无统计学意义,这可能与PET/CT阳性病灶中早期腺癌(7处)、非高风险腺瘤(5处)和非腺瘤性息肉(8处)的样本量较少有关。进一步的ROC曲线分析结果显示,SUVmax>11.6为诊断结直肠早期腺癌和高风险腺瘤的最佳临界值,灵敏度为66.0%,特异度为76.0%。鉴于目前大部分研究结果显示SUVmax并不能鉴别腺瘤与良性病灶,因此,不少学者认为对于每个结直肠局灶性摄取增高灶,后续均需行结肠镜检查[16]。
本研究的不足之处:本研究为回顾性分析研究,部分组织病理学类型的样本量较小,如早期腺癌,可能对于早期腺癌、腺瘤及非腺瘤性息肉的SUVmax之间的比较有一定影响,未来仍需更大样本量的研究来进一步探讨。
综上,本研究结果显示,偶发18F-FDG PET/CT结直肠局灶性摄取增高灶中有55.4%为结直肠早期腺癌和高风险腺瘤,且其在高风险腺瘤和早期腺癌中的诊断灵敏度明显增高。考虑到18F-FDG PET/CT在结直肠早期腺癌和高风险腺瘤的诊断中具有较高的阳性预测值和灵敏度,对于偶发18F-FDG PET/CT结直肠局灶性摄取增高灶,后续的结肠镜检查是非常有必要的,特别是对于高SUVmax的病灶。
利益冲突 所有作者声明无利益冲突
作者贡献声明 李文婵负责研究的设计、数据的收集与统计分析、论文的撰写;刘甫庚、姚稚明负责论文的审阅与修改;王宇芃负责数据的统计分析;刘秀芹负责数据的收集与分析;张竹负责论文撰写的指导
偶发18F-FDG PET/CT结直肠局灶性摄取增高对结直肠早期腺癌及高风险腺瘤的预测价值
Predictive value of incidental focal colorectal FDG uptake on 18F-FDG PET/CT in early colorectal adenocarcinoma and advanced adenomas
-
摘要:
目的 探讨偶发18F-氟脱氧葡萄糖(FDG) PET/CT结直肠局灶性摄取增高对结直肠早期腺癌及高风险腺瘤的预测价值。 方法 回顾性分析2016年1月至2020年6月在北京医院因非结直肠病变行全身18F-FDG PET/CT显像并偶然发现结直肠局灶性摄取增高的56例患者[男性31例、女性25例,年龄(66±11)岁,范围38~84岁]的临床资料,以18F-FDG PET/CT检查后3个月内的结肠镜及组织病理学检查结果作为最终诊断标准,分析18F-FDG PET/CT对结直肠早期腺癌及高风险腺瘤的预测价值。正态分布的计量资料的两组间比较采用独立样本t检验,多组间比较采用方差分析;偏态分布的计量资料的两组间比较采用Mann-Whitney检验,多组间比较采用Kruskal-Wallis检验。计数资料的比较采用卡方检验。勾画受试者工作特征(ROC)曲线,分析最大标准化摄取值(SUVmax)对结直肠早期腺癌和高风险腺瘤的诊断效能及最佳诊断临界值。 结果 56例受检者中,18F-FDG PET/CT共发现74处结直肠局灶性摄取增高灶,结肠镜共发现140处阳性病灶。18F-FDG PET/CT发现的74处结直肠局灶性摄取增高灶中,结肠镜发现阳性病灶59处[其中69.5%(41/59)为早期腺癌(7处)和高风险腺瘤(34处)]、阴性病灶15处。18F-FDG PET/CT共漏诊81处病灶,其中55.6%为非高风险腺瘤(45处),27.2%为非腺瘤性息肉(22处)。18F-FDG PET/CT诊断非腺瘤性息肉、非高风险腺瘤、高风险腺瘤、早期腺癌的灵敏度分别为26.7%(8/30)、10.0%(5/50)、72.3%(34/47)和100%(7/7)(χ2=35.09,P<0.001)。18F-FDG PET/CT诊断结直肠早期腺癌和高风险腺瘤的阳性预测值为55.4%(41/74)。ROC曲线分析结果显示,SUVmax诊断结直肠早期腺癌及高风险腺瘤的最佳临界值为11.6。 结论 偶发18F-FDG PET/CT结直肠局灶性摄取增高对结直肠早期腺癌及高风险腺瘤的诊断灵敏度及阳性预测值均较高。对于偶发18F-FDG PET/CT结直肠局灶性摄取增高灶,后续的结肠镜检查是非常必要的,特别是对于高SUVmax病灶。 Abstract:Objective To investigate the predictive values of 18F-fluorodeoxyglucose (FDG) PET/CT in patients with incidental focal colorectal FDG uptake in diagnosing early colorectal adenocarcinoma and advanced adenomas by comparing such values with colonoscopic and histopathologic findings. Methods Fifty-six patients (31 males and 25 females, aged (66±11) years, range 38–84 years) were retrospectively enrolled for the study. These patients underwent 18F-FDG PET/CT for noncolorectal lesions with incidental focal colorectal FDG uptake in Beijing Hospital between January 2016 and June 2020. Taking the results of colonoscopy and histopathology within 3 months after 18F-FDG PET/CT examination as the final diagnostic criteria, the predictive values of 18F-FDG PET/CT for early colorectal adenocarcinoma and advanced adenoma were analyzed. The normally distributed measurement data were compared between two groups using independent sample t-test (with equal variance) and among multiple groups using analysis of variance. Skewed distribution measurement data were compared between these two groups using Mann-Whitney test, whereas those for multiple groups were compared using the Kruskal-Wallis test. Rate enumeration data were also compared using the Chi-square test. Furthermore, the receiver operating characteristic (ROC) curve was delineated to analyze the diagnostic efficacy and optimal diagnostic threshold of maximum standardized uptake value (SUVmax) for diagnosing early colorectal adenocarcinoma and advanced adenomas. Results Fifty-six patients were enrolled in this study. A total of 74 incidental colorectal 18F-FDG uptake foci were found, while colonoscopy revealed 140 lesions. Among 74 incidental colorectal 18F-FDG uptake foci, 59 and 15 foci were found to be positive and negative, respectively, during colonoscopy. Among 59 positive colonoscopic findings, 69.5%(41/59) were early colorectal adenocarcinoma (n=7) and advanced adenomas (n=34). Among 81 lesions that were considered missed diagnoses via 18F-FDG PET/CT, 55.6% were nonadvanced adenomas (n=45) and 27.2% were nonadenomatous polyps (n=22). The sensitivities of 18F-FDG PET/CT in nonadenomatous polyps, nonadvanced adenomas, advanced adenomas, and early colorectal adenocarcinoma were 26.7% (8/30), 10.0% (5/50), 72.3% (34/47), and 100% (7/7), respectively (χ2=35.09, P<0.001). The positive predictive value of 18F-FDG PET/CT in early colorectal adenocarcinoma and advanced adenomas was 55.4% (41/74). In terms of the ROC analysis, the optimal cut-off value of SUVmax for diagnosing early colorectal adenocarcinoma and advanced adenomas was 11.6. Conclusions In this study, 18F-FDG PET/CT showed high positive predictive value and sensitivity in diagnosing early colorectal adenocarcinoma and advanced adenomas. According to the findings, follow-up colonoscopy is needed, especially in incidental focal colorectal 18F-FDG uptake lesions with high SUVmax. -
Key words:
- Colorectal neoplasms /
- Adenocarcinoma /
- Adenoma /
- Fluorodeoxyglucose F18 /
- Positron-emission tomography /
- Colonoscopy /
- Focal uptake
-
表 1 不同病理类型的结直肠病灶的SUVmax及18F-FDG PET/CT诊断灵敏度比较
Table 1. Comparison of maximum standardized uptake value and sensitivity of 18F-FDG PET/CT among different pathologic types of colorectal lesions
病理类型 病灶数
(处)SUVmax( $\bar x \pm s $ 

18F-FDG PET/CT
诊断灵敏度早期腺癌 7 15.4±7.7 100%(7/7) 高风险腺瘤 47 16.5±10.2 72.3%(34/47)a 非高风险腺瘤 50 7.7±0.6 10.0%(5/50) 非腺瘤性息肉 30 12.1±6.4 26.7%(8/30) 检验值 F=0.78 χ2=35.09 P值 P=0.511 P<0.001 注:a表示与非高风险腺瘤相比,差异有统计学意义(χ2=39.20,P<0.001)。SUVmax为最大标准化摄取值;FDG为氟脱氧葡萄糖;PET为正电子发射断层显像术;CT为计算机体层摄影术 -
[1] Brenner H, Hoffmeister M, Stegmaier C, et al. Risk of progression of advanced adenomas to colorectal cancer by age and sex: estimates based on 840 149 screening colonoscopies[J]. Gut, 2007, 56(11): 1585−1589. DOI: 10.1136/gut.2007.122739. [2] Kousgaard SJ, Thorlacius-Ussing O. Incidental colorectal FDG uptake on PET/CT scan and lesions observed during subsequent colonoscopy: a systematic review[J]. Tech Coloproctol, 2017, 21(7): 521−529. DOI: 10.1007/s10151-017-1652-6. [3] Shmidt E, Nehra V, Lowe V, et al. Clinical significance of incidental [18F]FDG uptake in the gastrointestinal tract on PET/CT imaging: a retrospective cohort study[J/OL]. BMC Gastroenterol, 2016, 16(1): 125[2022-11-09]. https://bmcgastroenterol.biomedcentral.com/articles/10.1186/s12876-016-0545-x. DOI: 10.1186/s12876-016-0545-x. [4] 中华医学会消化内镜学分会消化系早癌内镜诊断与治疗协作组, 中华医学会消化病学分会消化道肿瘤协作组, 中华医学会消化内镜学分会肠道学组, 等. 中国早期结直肠癌及癌前病变筛查与诊治共识(2014年, 重庆)[J]. 中华消化内镜杂志, 2015, 32(2): 69−85. DOI: 10.3760/cma.j.issn.1007-5232.2015.02.001.
Collaborative Group on Endoscopic Diagnosis and Treatment of Early Gastrointestinal Cancer, Digestive Endoscopy Branch, Chinese Medical Association, Collaborative Group on Gastrointestinal Tumors, Digestive Disease Branch, Chinese Medical Association, Gastroenterology Group, Digestive Endoscopy Branch, Chinese Medical Association, et al. Chinese consensus on screening, diagnosis and treatment of early colorectal cancer and precancerous lesions (2014, Chongqing)[J]. Chin J Dig Endosc, 2015, 32(2): 69−85. DOI: 10.3760/cma.j.issn.1007-5232.2015.02.001.[5] Gökden Y, Özülker F, Özülker T. Prevalence and clinical significance of incidental focal 18F-FDG uptake in colon on PET/CT imaging[J]. Mol Imaging Radionucl Ther, 2022, 31(2): 96−103. DOI: 10.4274/mirt.galenos.2022.38247. [6] 周海萍, 沈忠磊, 赵坚培, 等. 结直肠腺瘤分布特征及危险因素分析[J]. 中华胃肠外科杂志, 2018, 21(6): 678−684. DOI: 10.3760/cma.j.issn.1671-0274.2018.06.011.
Zhou HP, Shen ZL, Zhao JP, et al. Distribution characteristics and risk factors of colorectal adenomas[J]. Chin J Gastrointest Surg, 2018, 21(6): 678−684. DOI: 10.3760/cma.j.issn.1671-0274.2018.06.011.[7] Albertsen LN, Jaensch C, Tornbjerg SM, et al. Correlation between incidental focal colorectal FDG uptake on PET/CT and colonoscopic and histopathological results[J]. Scand J Gastroenterol, 2022, 57(2): 246−252. DOI: 10.1080/00365521.2021.1998602. [8] Dutta AK, Mitchell-Hay R, Baio G, et al. Clinically significant findings in patients with focal incidental colorectal abnormalities on positron emission tomography-CT scans[J]. J Med Imaging Radiat Oncol, 2022, 66(6): 749−754. DOI: 10.1111/1754-9485.13338. [9] Hwang JP, Woo SK, Yoon SY, et al. The potential usefulness of 18F-FDG PET/CT for detecting colorectal carcinoma and adenoma in asymptomatic adults[J]. Ann Nucl Med, 2015, 29(2): 157−163. DOI: 10.1007/s12149-014-0922-x. [10] Xu WM, Li HS, Guo ZQ, et al. Combined SUVmax and localized colonic wall thickening parameters to identify high-risk lesions from incidental focal colorectal 18F-FDG uptake foci[J/OL]. Front Oncol, 2022, 12: 972096[2022-11-09]. https://www.frontiersin.org/articles/10.3389/fonc.2022.972096/full. DOI: 10.3389/fonc.2022.972096. [11] Alongi P, Laudicella R, Gentile R, et al. Potential clinical value of quantitative fluorine-18-fluorodeoxyglucose-PET/computed tomography using a graph-based method analysis in evaluation of incidental lesions of gastrointestinal tract: correlation with endoscopic and histopathological findings[J]. Nucl Med Commun, 2019, 40(10): 1060−1065. DOI: 10.1097/MNM.0000000000001062. [12] Gilhotra RA, Song LS, Remedios M, et al. Clinical significance of incidental 18FDG PET uptake in the gastrointestinal tract: a retrospective cohort study[J]. Intern Med J, 2022, DOI: 10.1111/imj.16002. [13] Chung SM, Kim KO, Cho IH, et al. Clinicopathological analysis and risk factors of advanced colorectal neoplasms incidentally detected by 18F-FDG PET-CT[J]. Eur J Gastroenterol Hepatol, 2017, 29(4): 407−413. DOI: 10.1097/MEG.0000000000000808. [14] Purandare NC, Gawade SK, Puranik AD, et al. Etiology and significance of incidentally detected focal colonic uptake on FDG PET/CT[J]. Indian J Radiol Imaging, 2012, 22(4): 260−266. DOI: 10.4103/0971-3026.111476. [15] van Hoeij FB, Keijsers RGM, Loffeld BCAJ, et al. Incidental colonic focal FDG uptake on PET/CT: can the maximum standardized uptake value (SUVmax) guide us in the timing of colonoscopy?[J]. Eur J Nucl Med Mol Imaging, 2015, 42(1): 66−71. DOI: 10.1007/s00259-014-2887-3. [16] Kirchner J, Schaarschmidt BM, Kour F, et al. Incidental 18F-FDG uptake in the colon: value of contrast-enhanced CT correlation with colonoscopic findings[J]. Eur J Nucl Med Mol Imaging, 2020, 47(4): 778−786. DOI: 10.1007/s00259-019-04579-y. -





 下载:
下载: