-
PET/CT因其高灵敏度、可视化、定量精确和非侵入性等特性,已成为临床重要的影像技术,广泛应用于癌症的诊断和治疗评估[1]。18F-FDG是临床PET/CT应用最为广泛的显像剂,能够探测葡萄糖代谢异常情况。然而,18F-FDG PET/CT的特异度较低,且常常难以正确区分生理性与病理性摄取。此外,18F需要通过加速器生产,对于大部分医院来说,获取和维护一个正常运转的回旋加速器的成本太高[2]。
在正电子核素显像中,68Ga具有优良的性能,其可以通过68Ge-68Ga发生器淋洗获得,获得途径较简便且成本相对较低。母体核素68Ge的半衰期为270.8 d,所得子体核素68Ga的半衰期为67.7 min,68Ga的正电子衰变能量占其所有衰变能量的89%,仅次于11C和18F。另外,68Ga的半衰期适中,较为适于实际使用。与11C(半衰期为20.3 min)相比,对68Ga进行化学合成和注射扫描有较为充裕的时间,其放射性活度不会迅速降低。相比于18F标记显像剂,68Ga标记显像剂具有标记方法简单、反应时间短、纯化技术简便等特点,由于68Ga标记的多为多肽分子,标记产物的特异性更好,通常具有更好的显像质量和更高的靶向性。目前,68Ga标记显像剂已经广泛用于去势抵抗性前列腺癌、神经内分泌肿瘤等恶性肿瘤的临床诊断、疗效评估和肿瘤分期[3-6]。
目前,国内68Ge-68Ga发生器的使用仍依赖进口,尚无国产的68Ge-68Ga发生器投入使用。我们自主研发了国产68Ge-68Ga发生器,用其进行标记前列腺特异性膜抗原(prostate-specific membrane antigen,PSMA)-11的可行性研究和动物体内显像实验,并与进口68Ge-68Ga发生器进行了初步对比。
-
PSMA-11前体(德国ABX公司);盐酸(0.05、1 mol/L)由超纯盐酸(德国Merck公司,30%)和注射用水(四川美大康佳乐药业有限公司)配制而成;醋酸钠溶液(5 mol/L)由无水超纯醋酸钠(德国Merck公司,99.99%)和注射用水配制而成;乙腈(天津赛孚瑞公司);三氟乙酸(上海玻尔公司);胰酶(美国Gibco公司);PBS(北京索莱宝公司);异氟烷(河北一品制药有限公司);1640培养基和胎牛血清(美国Gibco公司);人前列腺癌22Rv1细胞(南京科佰生物科技有限公司)。
68Ge-68Ga发生器(德国ITG公司,规格为1 850 MBq);回旋加速器(美国TCC公司,CS30型);高纯锗伽马能谱仪(美国EG&G Ortec公司,GEM30P4-76型);电感耦合等离子体发射光谱仪(美国Perkin Elmer公司,ICP-OES型);小动物PET/CT活体影像系统(德国Siemens公司,Inveon MM型);动物麻醉机(美国Matrx公司,VIP3000型);电热恒温器(上海净信公司,JXMIN-80型);细胞培养箱(美国Thermo公司,311直热式);电子分析天平(瑞士Mettler-Toledo公司,AL104-IC型);高效液相色谱仪(日本岛津公司,M20A型);放射性检测器(美国Bioscan公司,FC3200型);医用放射性核素活度计(美国Capintec公司,CRC-55tPET型);C18 light柱(美国Waters公司);C18反相色谱柱(美国Agilent公司,ZORBAX Eclipse C18Plus),柱内径4.6 mm,柱高度250 mm,填料粒径5 μm。
-
5周龄雄性裸鼠6只,购自北京华阜康生物科技股份有限公司,实验动物生产许可证号:SCXK(京)2019-008,体重15~25 g;5~7周龄雄性昆明小鼠6只,购自西南医科大学实验动物中心,实验动物生产许可证号:SCXK(川)2023-0017,实验动物使用许可证号:SYXK(川)2023-0065,体重25~35 g。采用标准无特定病原体级条件饲养,温度25~28℃,湿度(55±5)%,小鼠均自由进食、饮水,每12 h黑暗和光照交替处理。
-
使用四川大学原子核科学技术研究所回旋加速器辐照Ga4Ni靶(自制),制备68Ge[69Ga(p,2n)68Ge],辐照时间10 min,辐照能量21 MeV,质子束流强度50 μA,辐照结束后使用双色谱柱(自制)分离纯化并获得68Ge。
-
依据文献[7]合成并制备SnO2,选用粒径为75~147 μm的SnO2进行发生器的装配。取1.3节中制备的68Ge溶解于1 mol/L盐酸中,并负载到SnO2柱上即得68Ge-68Ga发生器。采用1 mol/L盐酸溶液淋洗68Ge-68Ga发生器,获得68Ga3+。测定淋洗后初始时间(0)、1、4、12 h的淋洗产物68Ga3+的放射性活度。与进口68Ge-68Ga发生器在上述时间点的淋洗产物的放射性活度以及初始时间的淋洗效率、淋洗比活度、淋洗产物的放射化学纯度和细菌内毒素进行比较。
-
使用高纯锗伽马能谱仪检测1.4节中淋洗液的放射性核纯度,检测时间800 s。国产68Ge-68Ga发生器淋洗出的68Ga3+衰变5 d后,采用薄层色谱分析法连续8 d检测淋洗液中68Ga3+的放射化学纯度,展开剂选用生理盐水。使用电感耦合等离子体发射光谱仪检测国产68Ge-68Ga发生器淋洗液衰变后的Ga3+、Ni2+和Sn2+的质量浓度。
-
得到68Ga3+后,进行pH值、反应温度和时间对68Ga3+标记PSMA-11标记率的影响的研究,以确定最佳标记条件。取含68Ga3+的淋洗液1.5 ml(含68Ga3+ 60 MBq)与10 μl PSMA-11前体(10 μg)充分混匀,加入适量醋酸钠(5 mol/L)调节pH值至4.5,置于85℃电热恒温器中加热15 min。反应液通过C18 light柱后,取10 ml生理盐水冲洗C18 light柱,除去未标记上的游离68Ga3+,再用0.5 ml 50%乙醇冲洗C18 light柱,即得68Ga-PSMA-11。计算衰减校正后的产率。
-
使用约5 ml的0.05 mol/L盐酸淋洗进口68Ge-68Ga发生器,得到68Ga3+,取后端淋洗液3 ml(含68Ga3+1110 MBq)与10 μl PSMA-11前体(10 μg)、600 μl醋酸钠(0.25 mol/L)混合,置于80℃电热恒温器中加热15 min。反应液通过C18 light柱后,取10 ml生理盐水冲洗C18 light柱,除去未标记上的游离68Ga3+,再用2 ml 50%乙醇冲洗C18 light柱,即得68Ga-PSMA-11。计算衰减校正后的产率。
-
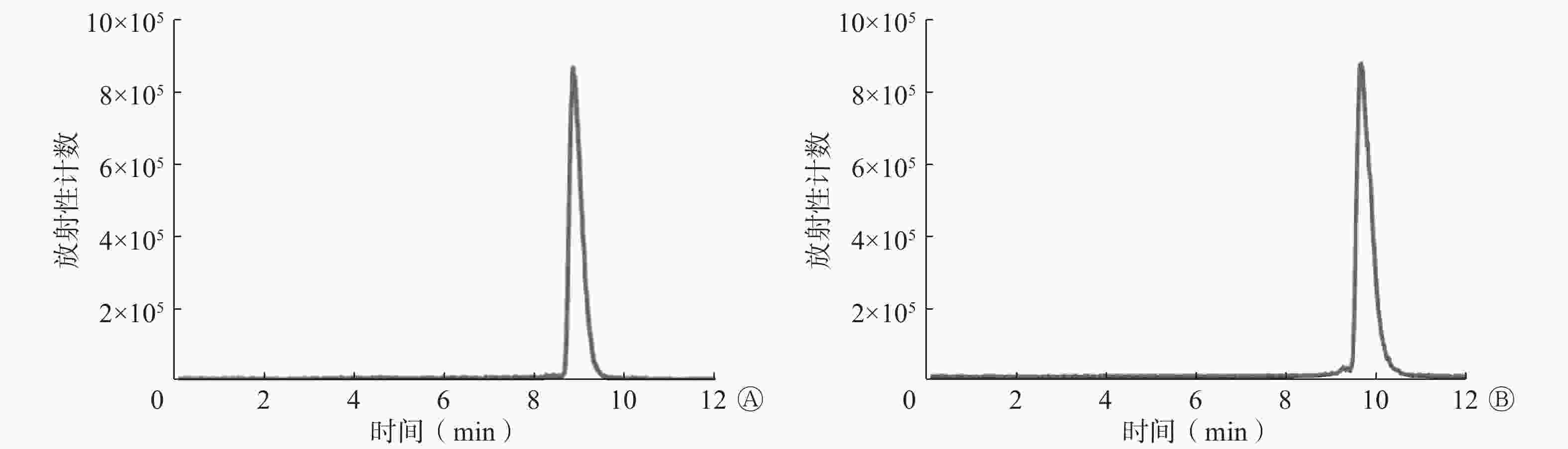
用高效液相色谱仪检测标记产物68Ga-PSMA-11的放射化学纯度。流动相A:体积分数为0.1%的三氟乙酸乙腈溶液;流动相B:体积分数为0.1%的三氟乙酸水溶液。采用C18反相色谱柱,检测波长为254 nm,洗脱梯度:在A+B溶液中,流动相A的百分比从10%逐渐上升至90%,流动相B的百分比从90%逐渐下降至10%,流速为1 ml/min,洗脱时间为0~15 min。
-
取6只昆明小鼠,采用随机数字分组方法分为2组,每组3只,禁食6 h以上,2组分别从尾静脉注射2.96 MBq国产68Ge-68Ga发生器生产的68Ga3+和进口68Ge-68Ga发生器生产的68Ga3+,30 min后进行小动物PET/CT显像,PET扫描时间为10 min。
-
将约5×106个22Rv1细胞于1640培养基(含10%胎牛血清和1%青霉素-链霉素),37℃及5% CO2细胞培养箱中培养,根据细胞密度及培养基情况进行换液或者传代。待细胞密度超过80%以上时,吸弃培养瓶中的培养液,加入2 ml 0.25%胰酶消化,吸出消化液,离心5 min(3000 转/min,离心半径10 cm)并计数,用PBS洗涤2次,加入无血清的1640培养基制成1×107个/ml细胞的悬液。取6只裸鼠,于左侧腋下接种,每只裸鼠皮下注射约0.1 ml的上述细胞悬液,接种细胞数量约为1×106个[8]。接种后15 d观察肿瘤模型是否构建成功。
-
取6只22Rv1肿瘤模型小鼠,采用随机数字分组方法分为2组,每组3只,禁食6 h以上,2组分别从尾静脉注射3.70 MBq国产68Ga-PSMA-11和进口68Ga-PSMA-11,于注射后30、60、90 min进行小动物PET/CT显像,每次扫描时间为20 min。
-
小鼠在异氟烷持续麻醉(动物麻醉机:氧气流量0.6 L/min,1.5档)状态下进行PET/CT显像。PET显像采用滤波反投影图像重构算法(图像大小128×128,扫描600 s),CT扫描采用标准定位(扫描参数:电压80 kV、电流500 μA,扫描时间8 min),采用德国Siemens公司 Inveon Research Workplace(IRW)4.2分析软件计算肿瘤部位的每克组织的注射剂量百分比(%ID/g)[9]。
-
应用SPSS 24.0软件进行统计学分析,符合正态分布的计量资料以
$ \bar x \pm s$ 表示,组间比较采用LSD法进行检验,P<0.05为差异有统计学意义。 -
国产68Ge-68Ga发生器在淋洗后初始时间(0)、1、4、12 h的淋洗产物68Ga3+的放射性活度分别为125.8、74.0、118.4、111.0 MBq;进口68Ge-68Ga发生器在上述时间点的淋洗产物68Ga3+的放射性活度分别为1554.0、925.0、1369.0、1184.0 MBq。
国产和进口68Ge-68Ga发生器在初始时间的淋洗效率分别为66.67%、84.00%,淋洗比活度分别为62.9、388.5 MBq/ml,淋洗产物的放射化学纯度均>99%,细菌内毒素均≤20 EU/ml。
-
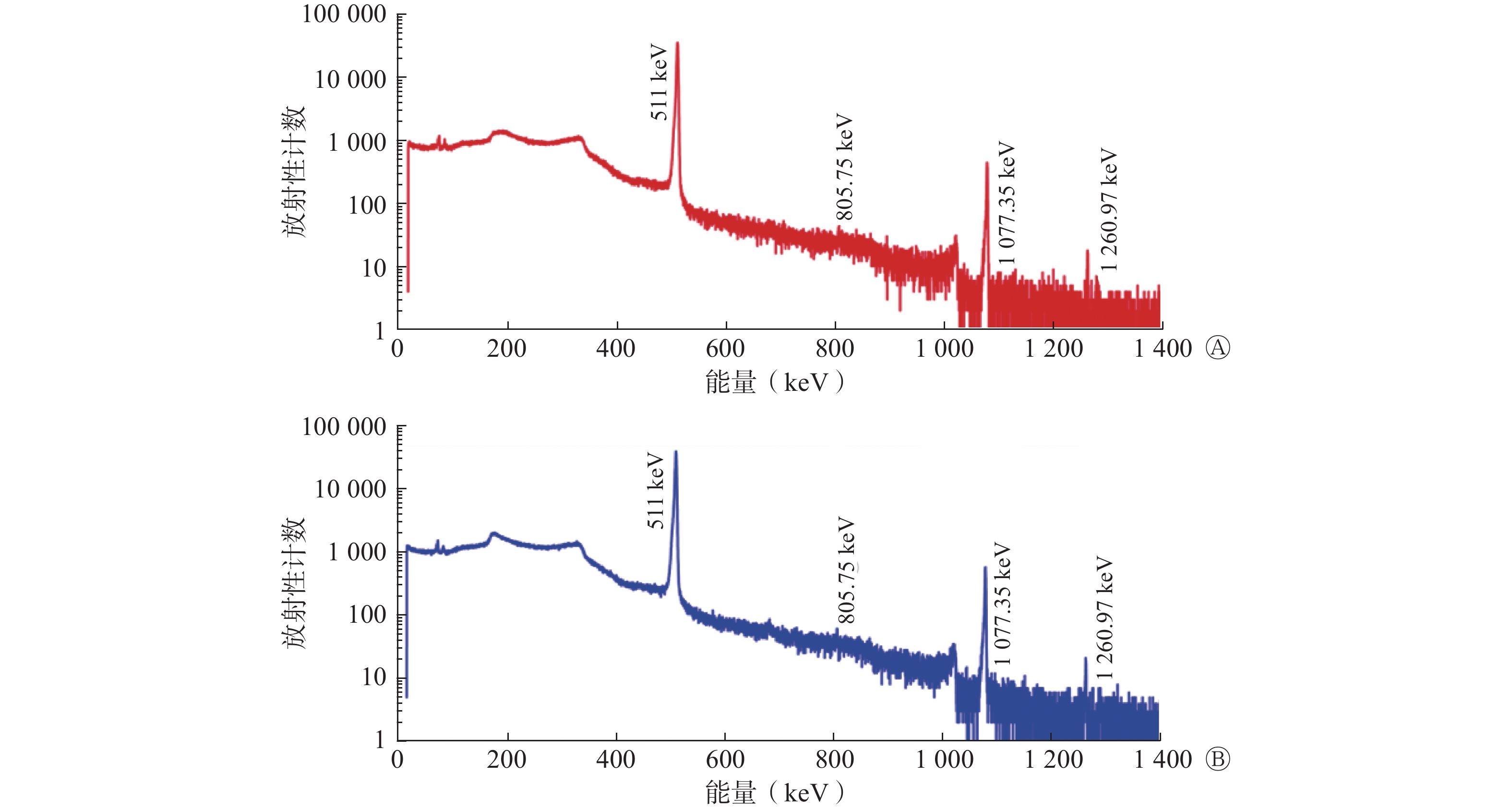
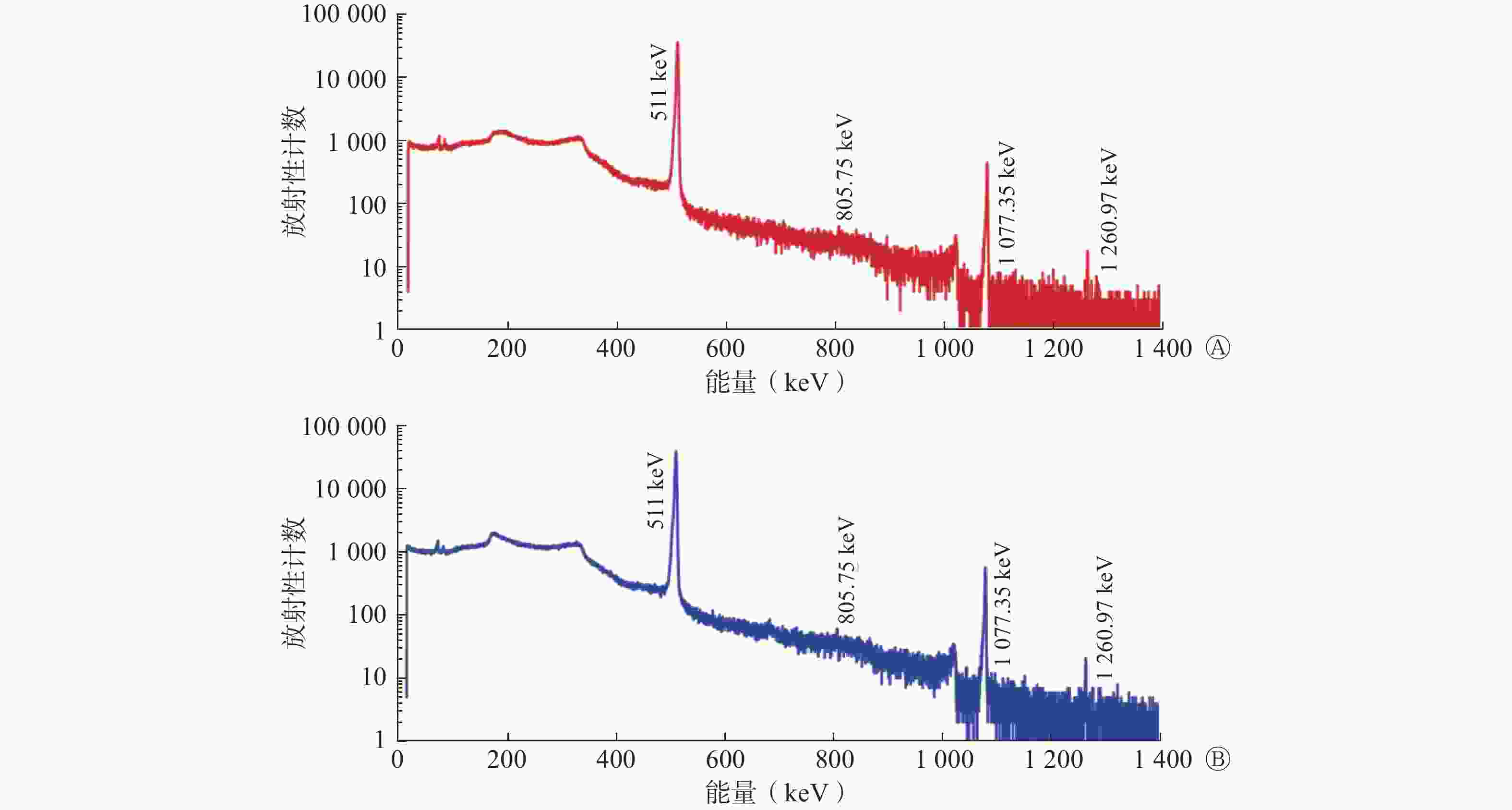
国产和进口68Ge-68Ga发生器淋洗产物68Ga3+的γ能谱图见图1,两者无明显差异,在淋洗产物中未见其他核素的γ能量峰,其放射性核纯度达到99.9%以上。国产68Ge-68Ga发生器淋洗产物68Ga3+衰变5 d后,长时间(8 d)薄层色谱分析结果显示,68Ga3+中有极微量的68Ge、56Co、57Co和65Zn存在,68Ga3+的放射化学纯度>99%。电感耦合等离子体发射光谱仪分析结果表明,在国产68Ge-68Ga发生器淋洗产物68Ga3+的衰变溶液中仅检测到Sn2+,质量浓度为0.02 mg/L。
-
国产68Ge-68Ga发生器淋洗产物标记PSMA-11的最佳标记条件为pH值4.5,85℃恒温反应15 min。国产和进口68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11经衰减校正后的产率分别为81.7%和82.8%。
-
国产和进口68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11的放射化学纯度均>95%,见图2。
-
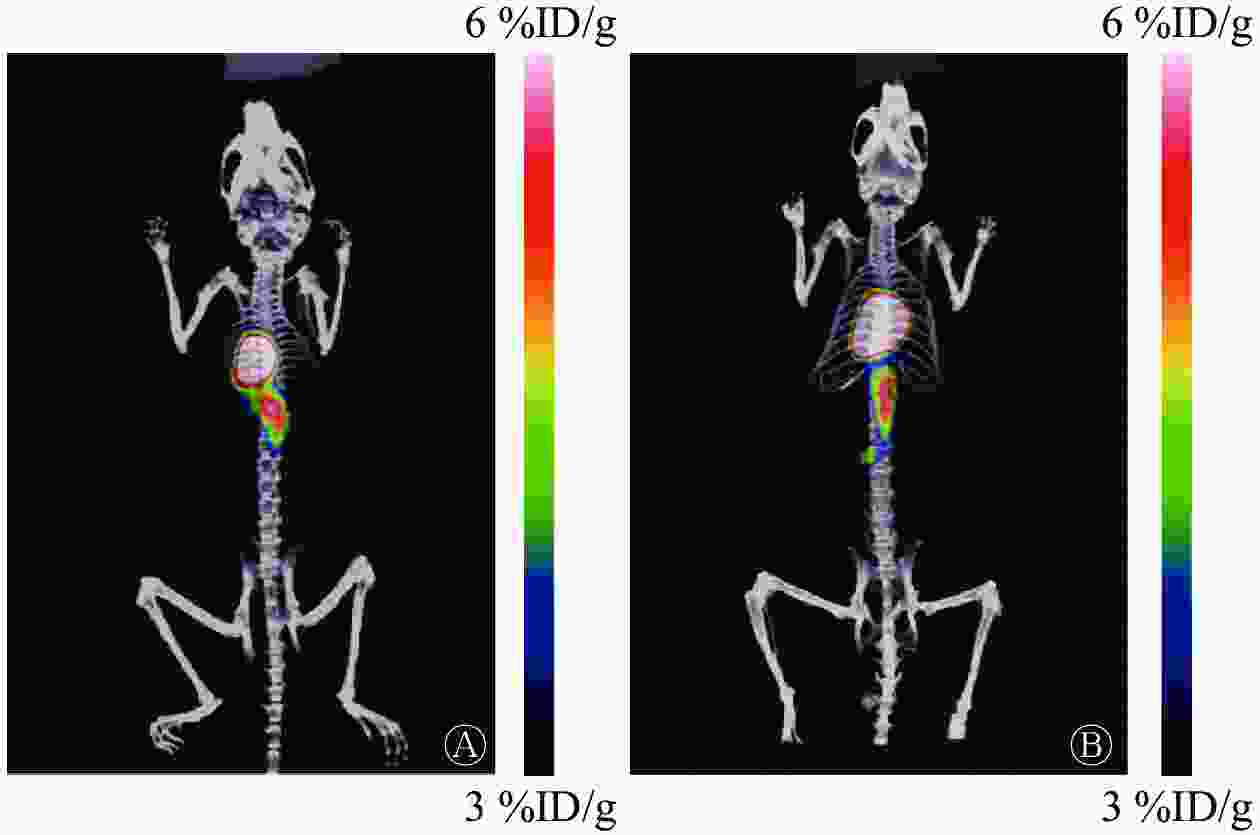
由图3可见,国产和进口68Ge-68Ga发生器淋洗产物68Ga3+在正常昆明小鼠体内的分布基本没有差异,均在心脏呈现放射性高摄取。
-
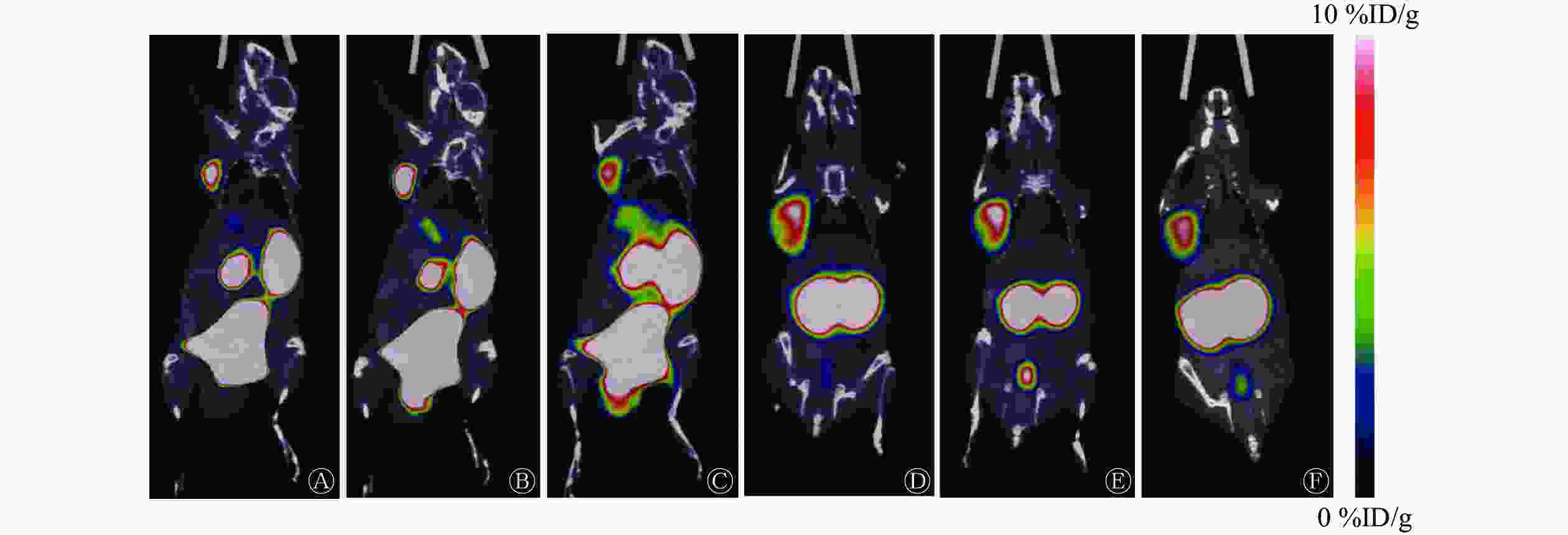
由图4可见,68Ga-PSMA-11注射后30、60、90 min,肿瘤部位均有明显放射性摄取,小鼠膀胱、肾脏也呈现放射性高摄取,其他脏器未见明显放射性摄取。国产68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11注射后30、60、90 min,肿瘤部位的放射性摄取值分别为(3.60±0.14)、(3.40±0.12)、(2.90±0.28) %ID/g。进口68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11注射后30、60、90 min,肿瘤部位的放射性摄取值分别为(5.30±0.42)、(5.90±0.36)、(5.50±0.33) %ID/g。国产68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11在注射后各时间点的肿瘤部位的放射性摄取值均低于进口68Ge-68Ga发生器,且差异均有统计学意义(LSD法,均P<0.05)。
-
近几年,以68Ga标记的小分子化合物和多肽逐渐成为肿瘤显像领域的研究热点,如68Ga-PSMA-11和68Ga-1,4,7,10- 四氮杂环十二烷-1,4,7,10-四乙酸-D-苯丙氨酸-1-酪氨酸-3-苏氨酸-8-奥曲肽(DOTATATE),已经成为临床诊断前列腺癌和内分泌肿瘤的“金标准”。68Ga能与多种含氮杂环形成稳定的络合物,如:1,4,7,10-四氮杂环十二烷-1,4,7,10-四乙酸(DOTA)、1,4,7-三氮杂环壬烷-N,N',N''-三乙酸(NOTA)、N,N'-双[2-羟基-5-(羧乙基)苄基]乙二胺-N,N'-二乙酸(HBEDCC)等,由于68Ga的标记条件温和、方法简单可控、标记率高等特点,被广泛应用于临床转化。同时,68Ga的配位性质与177Lu、90Y类似,可以标记同一化合物并分别进行显像和多肽受体放射性核素治疗,更加精准地筛选病例、制定个体化治疗方案、进行个体化预后评价。68Ga的临床应用不仅推动了核素显像的发展,同时也推动了核素诊疗一体化的进程,实现了精准诊疗的一体化。
68Ga标记的PSMA抑制剂是一类重要的68Ga标记显像剂,研究结果显示,PSMA的表达水平与前列腺癌的发生、发展密切相关,且随着疾病的进展,PSMA的表达水平显著升高[10-11]。靶向PSMA的核素标记小分子抑制剂主要集中在脲基类PSMA抑制剂中[12-17]。有研究者将68Ga-PSMA-11用于248例前列腺癌根治性切除术后生化复发患者的显像,结果显示前列腺特异性抗原(PSA)水平越高,病灶的检出率越高,该研究中发现了81个常规影像检查无法识别的病灶,68Ga-PSMA-11 PET/CT已成为前列腺癌诊断的重要方法[18]。最新的Ⅲ期临床研究报道,68Ga-PSMA-11 PET/CT与常用的CT和骨扫描相比,其诊断前列腺癌的准确率更高,辐射剂量更小,68Ga-PSMA-11 PET/CT对前列腺癌盆腔淋巴结或远处转移的诊断准确率(92%)高于传统影像(65%)[19]。
68Ga在核医学研究中成为热点,主要是由于以下两方面原因。一是68Ge-68Ga发生器的进步,使用68Ge-68Ga发生器供应68Ga,可以实现“药盒化”,在生产成本、使用方便性、辐射剂量控制等方面都有较大的优势。通常一台68Ge-68Ga发生器的售价约60万元,可以使用1年左右。2000年以后,68Ge-68Ga发生器的离子吸附、分离纯化、淋洗工艺取得了较大进步,68Ge-68Ga发生器的技术进步也促进了新型放射性药物的研发和应用。二是68Ga与177Lu的核物理性能非常适合临床诊断与治疗,其标记化合物是诊疗一体化研究的热点[20]。177Lu和68Ga可以组成“诊疗一体化”配对药物,能够将诊断和治疗结合起来,从而实现精准靶向治疗,确保疗效。其原因在于177Lu和68Ga有相似的化学配位特性,可以使用相同的螯合剂,因而具备相似的药代动力学特征,能将显像与治疗关联起来,实现“所见即所得”,形成诊疗一体化。
本研究制备了国产68Ge-68Ga发生器,并与进口68Ge-68Ga发生器淋洗产物68Ga3+的γ能谱、初始时间淋洗参数、标记PSMA-11流程、体外稳定性和动物显像方面进行了比较,结果显示,国产与进口68Ge-68Ga发生器淋洗产物68Ga3+的γ能谱基本相符,无明显差异。初始时间国产68Ge-68Ga发生器的淋洗效率、淋洗比活度低于进口68Ge-68Ga发生器,淋洗产物的放射化学纯度、细菌内毒素与进口68Ge-68Ga发生器基本一致。与进口68Ge-68Ga发生器比较,国产68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11衰减校正后的产率与其基本一致,放射化学纯度均达到了95%以上。在正常小鼠和22Rv1前列腺癌肿瘤模型小鼠中的显像结果显示,国产与进口68Ge-68Ga发生器淋洗产物68Ga3+在正常小鼠体内的分布情况无明显差异, 68Ga3+主要被心脏摄取,68Ga-PSMA-11主要被肿瘤部位摄取,并通过肾脏排泄,但国产68Ga-PSMA-11在肿瘤部位的放射性摄取值低于进口68Ga-PSMA-11,其主要原因是国产68Ge-68Ga发生器淋洗出的68Ga3+较少,导致68Ga-PSMA-11的比活度低,影响了肿瘤的摄取。本实验结果初步表明,国产68Ge-68Ga发生器与进口68Ge-68Ga发生器在生产的核素的放射化学纯度、标记条件、动物显像体内分布等方面均无明显差异,可用于后续实验研究。
基于68Ga优良的物理性质以及在核医学研究中的突出优势,加之目前国内尚无商品化的68Ge-68Ga发生器供应,相关单位使用的68Ge-68Ga发生器仍是依赖进口,因此,在国内开展68Ge-68Ga发生器的研制工作是非常有必要的。本研究制备的国产68Ge-68Ga发生器目前生产的产品放射性活度较小,不能达到临床使用需求,仍有待提升。
利益冲突 所有作者声明无利益冲突
作者贡献声明 陈琳负责实验的开展、论文的撰写;邢菲负责数据的获取与分析;冯悦负责现场实验的指导、数据的分析;刘宁、秦芝负责部分实验的开展和指导;陈跃负责研究内容的设计、论文的审阅
国产68Ge-68Ga发生器的制备及初步应用评价
Preparation and preliminary application evaluation of a domestic 68Ge-68Ga generator
-
摘要:
目的 制备国产68Ge-68Ga发生器,通过与进口68Ge-68Ga发生器(德国ITG公司)进行对比,对其进行初步应用评价。 方法 制备68Ge-68Ga发生器,测定其淋洗后初始时间(0)、1、4、12 h的淋洗产物68Ga3+的放射性活度;与进口68Ge-68Ga发生器淋洗产物68Ga3+ 的γ能谱及68Ga3+在正常小鼠体内的分布情况进行对比;确定国产68Ge-68Ga发生器淋洗产物标记前列腺特异性膜抗原(PSMA)-11的最佳标记条件及产物的放射化学纯度;对比国产与进口68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11在22Rv1前列腺癌肿瘤模型小鼠体内的分布情况。计量资料的组间比较采用LSD法进行检验。 结果 国产68Ge-68Ga发生器在淋洗后初始时间(0)、1、4、12 h的淋洗产物68Ga3+的放射性活度分别为125.8、74.0、118.4、111.0 MBq。国产与进口68Ge-68Ga发生器淋洗产物68Ga3+的γ能谱高度一致,在正常小鼠体内的分布基本没有差异,均在心脏呈现放射性高摄取。国产68Ge-68Ga发生器淋洗产物标记PSMA-11的最佳标记条件是pH值4.5,85℃恒温反应15 min,得到的68Ga-PSMA-11的放射化学纯度>95%。国产与进口68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11在22Rv1前列腺癌肿瘤模型小鼠体内的显像结果显示,68Ga-PSMA-11均在肿瘤部位呈现高摄取,其余主要分布于肾脏和膀胱。国产68Ge-68Ga发生器淋洗产物标记的68Ga-PSMA-11注射后30、60、90 min,肿瘤部位的放射性摄取值均低于进口68Ge-68Ga发生器[(3.60±0.14) %ID/g对(5.30±0.42) %ID/g、(3.40±0.12) %ID/g对(5.90±0.36) %ID/g、(2.90±0.28) %ID/g对(5.50±0.33) %ID/g],且差异均有统计学意义(LSD法,均P<0.05)。 结论 国产68Ge-68Ga发生器淋洗产物68Ga3+及其标记的68Ga-PSMA-11在正常小鼠和肿瘤模型小鼠体内的分布情况与进口68Ge-68Ga发生器无明显差异。 -
关键词:
- 放射性核素发生器 /
- 锗 /
- 镓放射性同位素 /
- 前列腺特异性膜抗原 /
- 正电子发射断层显像术 /
- 体层摄影术,X线计算机
Abstract:Objective To prepare a domestically produced 68Ge-68Ga generator and compare it with an imported 68Ge-68Ga generator (ITG Company, Germany) for preliminary application evaluation. Methods A 68Ge-68Ga generator was prepared, and the radioactivity of its elution product 68Ga3+ was measured at the initial time (0) and 1, 4, and 12 h after elution. The γ-spectra of the elution product 68Ga3+ and its distribution in normal mice were compared with those of an imported 68Ge-68Ga generator. The optimal labeling conditions for prostate-specific membrane antigen (PSMA)-11 labeled by domestic 68Ge-68Ga generator elution product and the radiochemical purity of the labeled product were determined, and the distribution of 68Ga-PSMA-11 labeled by domestic and imported 68Ge-68Ga generator elution products in 22Rv1 prostate cancer tumor model mice was compared. The intergroup comparison of measurement data was conducted using the LSD method. Results The radioactivity values of 68Ga3+ in the elution product of the domestic 68Ge-68Ga generator at the initial time (0), 1, 4, and 12 h after elution were 125.8, 74.0, 118.4, and 111.0 MBq, respectively. The γ-spectra of 68Ga3+ eluted by domestic and imported 68Ge-68Ga generators were highly consistent, and no difference was observed in the distribution of 68Ga3+ in normal mice, and both showed high uptake of radioactivity in the heart. The optimal labeling conditions for PSMA-11 labeled by domestic 68Ge-68Ga generator elution product were pH 4.5, reaction at 85 ℃ for 15 min, and the radiochemical purity of the product 68Ga-PSMA-11 was >95%. The imaging results of 68Ga-PSMA-11 labeled by domestic and imported generator elution products in 22Rv1 prostate cancer model mice showed high uptake of 68Ga-PSMA-11 at the tumor site, while the rest were mainly distributed in the kidneys and bladder. At 30, 60, and 90 min after injection, the radioactive uptake values of 68Ga-PSMA-11 labeled by the domestic 68Ge-68Ga generator were lower than those of the imported 68Ge-68Ga generator ((3.60±0.14) %ID/g vs. (5.30±0.42) %ID/g, (3.40±0.12) %ID/g vs. (5.90±0.36) %ID/g, (2.90±0.28) %ID/g vs. (5.50±0.33) %ID/g). The differences were statistically significant (LSD method, all P<0.05). Conclusion No significant difference was observed in the distribution of 68Ga3+ and its labeled 68Ga-PSMA-11 in normal mice and tumor model mice between the domestic 68Ge-68Ga generator and the imported 68Ge-68Ga generator. -
-
[1] Lopci E, Fanti S. Non-FDG PET/CT[M]//Schober O, Kiessling F, Debus J. Molecular imaging in oncology. Cham: Springer, 2020: 669−718. DOI: 10.1007/978-3-030-42618-7_20. [2] Martiniova L, De Palatis L, Etchebehere E, et al. Gallium-68 in medical imaging[J]. Curr Radiopharm, 2016, 9(3): 187−207. DOI: 10.2174/1874471009666161028150654. [3] Garzón JRG, de Arcocha Torres M, Delgado-Bolton R, et al. 68Ga-PSMA PET/CT in prostate cancer[J/OL]. Rev Esp Med Nucl Imagen Mol (Engl Ed), 2018, 37(2): 130−138[2022-10-07]. https://www.sciencedirect.com/science/article/abs/pii/S2253654X17300744?via%3Dihub. DOI: 10.1016/j.remn.2017.07.004. [4] Zhao RN, Li YJ, Nie LH, et al. The meta-analysis of the effect of 68Ga-PSMA-PET/CT diagnosis of prostatic cancer compared with bone scan[J]. Medicine (Baltimore), 2021, 100(15): e25417. DOI: 10.1097/MD.0000000000025417. [5] Hu XW, Wu Y, Yang PQ, et al. Performance of 68Ga-labeled prostate-specific membrane antigen ligand positron emission tomography/computed tomography in the diagnosis of primary prostate cancer: a systematic review and meta-analysis[J]. Int Braz J Urol, 2022, 48(6): 891−902. DOI: 10.1590/S1677-5538.IBJU.2020.0986. [6] Sanli Y, Garg I, Kandathil A, et al. Neuroendocrine tumor diagnosis and management: 68Ga-DOTATATE PET/CT[J]. AJR Am J Roentgenol, 2018, 211(2): 267−277. DOI: 10.2214/AJR.18.19881. [7] Brauer G. Handbook of preparative inorganic chemistry[M]. 2nd ed. New York: Academic Press, 1963: 738. [8] Kim SB, Song IH, Song YS, et al. Biodistribution and internal radiation dosimetry of a companion diagnostic radiopharmaceutical, [68Ga]PSMA-11, in subcutaneous prostate cancer xenograft model mice[J/OL]. Sci Rep, 2021, 11(1): 15263[2022-10-07]. https://www.nature.com/articles/s41598-021-94684-6. DOI: 10.1038/s41598-021-94684-6. [9] 冯悦, 钟萌, 罗见春, 等. 18F-FDG PET/CT技术监测雷公藤甲素对类风湿关节炎模型大鼠关节损伤的改善作用[J]. 中国药房, 2018, 29(22): 3059−3062. DOI: 10.6039/j.issn.1001-0408.2018.22.08.
Feng Y, Zhong M, Luo JC, et al. Improvement effects of triptolide on joint injury in rheumatoid arthritis model rats monitored by 18F-FDG PET/CT imaging[J]. China Pharm, 2018, 29(22): 3059−3062. DOI: 10.6039/j.issn.1001-0408.2018.22.08.[10] Hoffmann MA, Wieler HJ, Baues C, et al. The impact of 68Ga-PSMA PET/CT and PET/MRI on the management of prostate cancer[J]. Urology, 2019, 130: 1−12. DOI: 10.1016/j.urology.2019.04.004. [11] 陈跃, 霍力, 兰晓莉, 等. 68Ga-前列腺特异性膜抗原PET/CT前列腺癌显像操作指南[J]. 中国医学影像技术, 2019, 35(10): 1441−1444. DOI: 10.13929/j.1003-3289.201905087.
Chen Y, Huo L, Lan XL, et al. Guideline of 68Ga-PSMA PET/CT imaging in prostatic cancer[J]. Chin J Med Imaging Technol, 2019, 35(10): 1441−1444. DOI: 10.13929/j.1003-3289.201905087.[12] Banerjee SR, Kumar V, Lisok A, et al. 177Lu-labeled low-molecular-weight agents for PSMA-targeted radiopharmaceutical therapy[J]. Eur J Nucl Med Mol Imaging, 2019, 46(12): 2545−2557. DOI: 10.1007/s00259-019-04434-0. [13] Pernthaler B, Kulnik R, Gstettner C, et al. A prospective head-to-head comparison of 18F-fluciclovine with 68Ga-PSMA-11 in biochemical recurrence of prostate cancer in PET/CT[J]. Clin Nucl Med, 2019, 44(10): e566−e573. DOI: 10.1097/RLU.0000000000002703. [14] Giesel FL, Will L, Lawal I, et al. Intraindividual comparison of 18F-PSMA-1007 and 18F-DCFPyL PET/CT in the prospective evaluation of patients with newly diagnosed prostate carcinoma: a pilot study[J]. J Nucl Med, 2018, 59(7): 1076−1080. DOI: 10.2967/jnumed.117.204669. [15] Banerjee S, Pillai MRA, Knapp FF. Lutetium-177 therapeutic radiopharmaceuticals: linking chemistry, radiochemistry, and practical applications[J]. Chem Rev, 2015, 115(8): 2934−2974. DOI: 10.1021/cr500171e. [16] Hofman MS, Violet J, Hicks RJ, et al. [177Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): a single-centre, single-arm, phase 2 study[J]. Lancet Oncol, 2018, 19(6): 825−833. DOI: 10.1016/S1470-2045(18)30198-0. [17] Fendler WP, Calais J, Eiber M, et al. Assessment of 68Ga-PSMA-11 PET accuracy in localizing recurrent prostate cancer: a prospective single-arm clinical trial[J]. JAMA Oncol, 2019, 5(6): 856−863. DOI: 10.1001/jamaoncol.2019.0096. [18] Eiber M, Maurer T, Souvatzoglou M, et al. Evaluation of hybrid 68Ga-PSMA ligand PET/CT in 248 patients with biochemical recurrence after radical prostatectomy[J]. J Nucl Med, 2015, 56(5): 668−674. DOI: 10.2967/jnumed.115.154153. [19] Hofman MS, Lawrentschuk N, Francis RJ, et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study[J]. Lancet, 2020, 395(10231): 1208−1216. DOI: 10.1016/S0140-6736(20)30314-7. [20] Farolfi A, Lima GM, Oyen W, et al. Molecular imaging and theranostics—a multidisciplinary approach[J]. Semin Nucl Med, 2019, 49(4): 247−254. DOI: 10.1053/j.semnuclmed.2019.02.002. -





 下载:
下载: