-
肺癌的发病率呈逐年上升趋势,是目前所有恶性肿瘤中发病率最高的一种,且病死率较高[1]。肺癌是呼吸系统中较为常见的一种恶性肿瘤,按照病理分型可将其分为小细胞肺癌和非小细胞肺癌,其中小细胞肺癌的恶性程度较高,非小细胞肺癌较为常见,约占所有肺癌的75%[2-3]。肺癌的恶性程度高,发病较为隐匿,早期确诊困难,一旦发现时已处于中晚期,且患者的5年生存率很低[4-6]。因此,早期准确地对肺癌进行诊断并进行病理学分型对于后续治疗方案的制定、患者预后的改善和生存期的延长等尤为重要。血清肿瘤标志物的产生与恶性肿瘤细胞异常或是宿主对肿瘤的刺激反应有关,作为一种特征性的存在,其能反映肿瘤发生发展的过程,可作为发现肿瘤的依据,且其取样方便、创伤小,能够为肿瘤的分类和诊断提供参考,可以进一步指导治疗方案的制定及改善患者预后等。高分辨率CT(high resolution CT,HRCT)是肺部检查常用的影像方法,为了更好地寻找肺癌的发生、发展的原因及其与病理特征的相关性,我们探讨了HRCT联合多种血清肿瘤标志物对早期肺癌的诊断价值,以期为临床诊治肺癌提供参考。
-
回顾性分析2019年8月至2021年10月首都医科大学附属北京天坛医院收治的164例早期肺癌患者(肺癌组)的临床资料,同时选取同期参加体检的78名健康者作为对照组。所有患者均经组织病理学检查结果证实。肺癌组男性94例、女性70例,年龄(52.4±3.6)岁;其中,腺癌90例、鳞癌62例、小细胞癌12例。2组人员在性别、年龄上的差异均无统计学意义(
χ2=0.210,t=0.172,均P>0.05),具有可比性。 患者纳入标准:(1)均经手术、穿刺活检等组织病理学检查结果确诊为肺癌;(2)均行CT和HRCT扫描,且均于检查前签署了知情同意书;(3)未伴随其他恶性肿瘤;(4)临床资料和影像资料完整。排除标准:(1)有既往放化疗史;(2)伴有其他肺部疾病。
本研究符合《赫尔辛基宣言》的原则。
-
所有患者均采用荷兰飞利浦公司的Brilliance iCT机行胸部扫描。患者取仰卧位,扫描范围:肺尖至肺底,先行常规螺旋CT平扫,再行增强扫描。发现病灶后再行HRCT扫描。增强CT扫描所用对比剂为碘佛醇注射液(江苏恒瑞医药股份有限公司,350 mg I/ml),使用高压注射器以3~4 ml/s的速率静脉注射,注射总量为80~100 ml,延迟扫描时间120 s。常规平扫参数:管电压120 kV,管电流30 mA,层厚10 mm,螺距1.5。增强扫描参数:管电压120 kV,自动管电流,矩阵512×512,螺距1.0,层厚5 mm,层距5 mm。HRCT扫描参数:每圈1.5 s,螺距1.0,层厚1.5 mm,层距1.5,管电压120 kV,管电流180 mA,实施骨算法重建。将获得的图像上传至VS2后处理工作站,观察病灶形态及结构,同时测量病灶大小。所有图像均由2名副主任医师独立阅片,意见不同时协商达成一致。
-
肺癌组患者和对照组健康者均于清晨取空腹静脉血5 ml进行检测。采用肝素抗凝,分离血清,于2~8℃低温环境下保存。使用罗氏Elecsys-2010电化学发光全自动免疫分析仪检测血清学指标:神经元特异性烯醇化酶(neuron-specific enolase,NSE)、癌胚抗原(carcinoembryonic antigen,CEA)、鳞状细胞癌抗原(squamous cell carcinoma antigen,SCCA)、细胞角蛋白19片段抗原21-1(cyto-keratin 19 fragment antigen 21-1,CYFRA21-1)、糖类抗原125(carbohydrate antigen 125,CA125)。严格按照说明书中的操作规范对以上血清肿瘤标志物进行检测。血清肿瘤标志物阳性标准:CEA>4.7 ng/ml、NSE>16.3 ng/ml、SCCA>1.5 μg/L、CYFRA21-1>3.3 ng/ml、CA125>35 U/ml。
-
对比肺癌组与对照组以及不同病理类型肺癌患者的血清NSE、CEA、CYFRA21-1、SCCA、CA125水平;分别计算HRCT、5种肿瘤标志物、HRCT+5种肿瘤标志物诊断肺癌的灵敏度、特异度和准确率。
-
应用SPSS22.0软件对数据进行统计学分析。符合正态分布的计量资料以
$ \bar{x}\pm s $ 表示,组间比较采用两独立样本t检验(方差齐)。计数资料以率表示,组间比较采用χ2检验。P<0.05为差异有统计学意义。 -
由表1可知,肺癌组的各项血清肿瘤标志物水平均高于对照组,且差异有统计学意义(t=8.107~30.460,均P<0.001)。
组别 CEA(ng/ml) NSE(ng/ml) CA125(U/ml) CYFRA21-1(ng/ml) SCCA(μg/L) 肺癌组(n=164) 45.45±4.51 23.52±3.56 124.23±29.43 16.78±2.56 7.36±2.56 对照组(n=78) 2.14±0.58 10.23±1.62 20.37±1.26 1.63±2.12 1.13±0.48 t值 30.460 8.107 14.619 10.363 14.532 P值 <0.001 <0.001 <0.001 <0.001 <0.001 注:CEA为癌胚抗原;NSE为神经元特异性烯醇化酶;CA125为糖类抗原125; CYFRA21-1为细胞角蛋白19片段抗原21-1;SCCA为鳞状细胞癌抗原 表 1 肺癌组患者与对照组健康者的血清肿瘤标志物水平比较(
$ \bar{x}\pm s $ Table 1. Comparison of serum tumor markers between lung cancer group and control group (
$ \bar{x}\pm s $ -
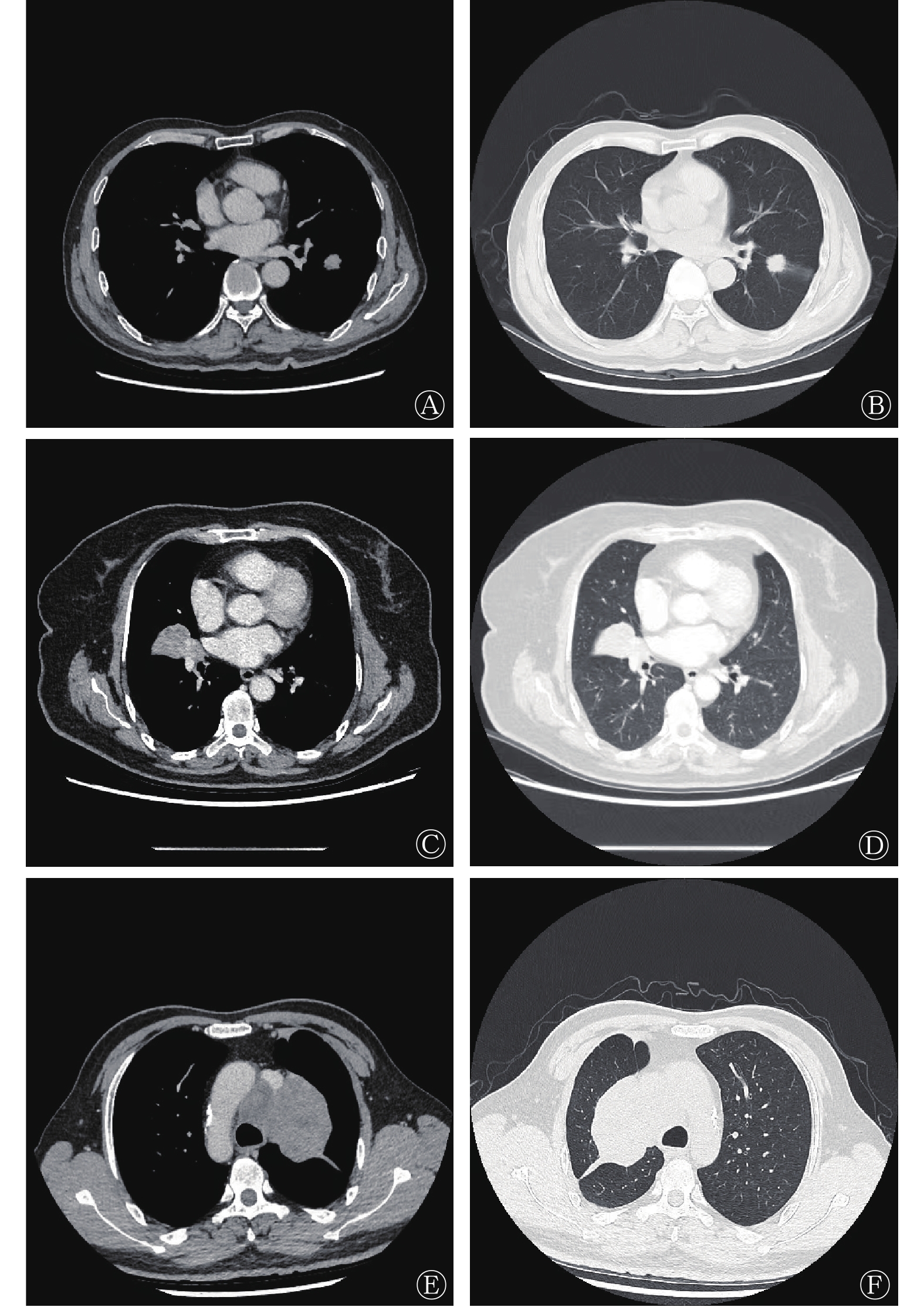
由表2可知,腺癌患者的血清CEA、CA125水平均高于鳞癌和小细胞癌患者,且差异有统计学意义(t=12.712~4.326,均P<0.001)。鳞癌患者的血清CEA、CA125水平与小细胞癌患者的相比,差异均无统计学意义(t=1.342、1.256,均P>0.05)。腺癌与鳞癌患者的NSE和CYFRA21-1水平均低于小细胞癌患者,且差异有统计学意义(t=10.342~11.534,均P<0.001)。腺癌、鳞癌和小细胞癌患者的典型病例HRCT图像如图1所示。
病理类型 CEA(ng/ml) NSE(ng/ml) CA125(U/ml) CYFRA21-1(ng/ml) SCCA(μg/L) 腺癌(n=90) 128.24±34.46ab 23.24±3.54b 147.36±24.55ab 7.05±2.61b 5.14±2.25 鳞癌(n=62) 34.82±12.89 20.13±3.11b 107.53±18.24 12.57±2.49b 10.24±3.08 小细胞癌(n=12) 36.06±14.24 70.81±20.36 109.65±13.20 45.72±12.36 4.89±2.13 注:a表示与鳞癌患者相比,差异均有统计学意义(t=12.712、4.326,均P<0.001);b表示与小细胞癌患者相比,差异均有统计学意义(t=6.304~15.134,均P<0.001)。CEA为癌胚抗原;NSE为神经元特异性烯醇化酶;CA125为糖类抗原125; CYFRA21-1为细胞角蛋白19片段抗原21-1;SCCA为鳞状细胞癌抗原 表 2 不同病理类型早期肺癌患者的血清肿瘤标志物水平比较(
$ \bar{x}\pm s $ Table 2. Comparison of serum tumor markers in patients with early lung cancer of different pathological types (
$ \bar{x}\pm s $ -
由表3可知,HRCT+5种肿瘤标志物联合检测对肺癌的诊断灵敏度为95.62%,特异度为96.43%,准确率为98.65%,均高于任意一种单独诊断,且差异有统计学意义(
χ2=55.823、48.652、62.718,均P<0.001)。 检测方法 灵敏度 特异度 准确率 CEA 42.56 88.65 64.00 NSE 43.41 91.78 67.05 CYFRA21-1 46.52 80.46 63.21 CA125 45.21 86.84 65.23 SCCA 45.02 86.95 64.00 HRCT 76.50 79.28 78.46 5种肿瘤标志物联合检测 68.26 74.84 72.32 HRCT+5种肿瘤标志物联合检测 95.62 96.43 98.65 注:CEA为癌胚抗原;NSE为神经元特异性烯醇化酶;CYFRA21-1为细胞角蛋白19片段抗原21-1;CA125为糖类抗原125; SCCA为鳞状细胞癌抗原;HRCT为高分辨率计算机体层摄影术 表 3 不同检测方法及其联合应用对早期肺癌的诊断效能(%)
Table 3. Diagnostic efficacy of different examination methods and their combination in early lung cancer (%)
-
多数肺癌患者在患病早期无临床表现,一经确诊已达中晚期,不能行根治性手术或是术后复发率高,预后不佳[7-8]。提高肺癌的早期诊断准确率,并制定合适的治疗方案对改善患者预后至关重要。目前组织病理学检查依然是确诊肺癌的“金标准”,但是其有创且不易重复操作[9]。因此,要想提高患者及时就诊的意识,应尽可能提供一种有效、无创且方便的方法来提高早期肺癌诊断的准确率,进而及时发现、尽早治疗、改善患者预后。
有研究者认为,血清肿瘤标志物的检测是诊断肿瘤的重要方法之一[10]。目前,最常用的筛查肺癌的肿瘤标志物有NSE、CEA、CYFRA21-1、CA125、SCCA等。CEA作为广谱肿瘤标志物,在多种肿瘤中的水平均有升高,有研究者报道,非小细胞肺癌患者的血清CEA水平与其表皮生长因子受体基因突变率呈正相关[9]。NSE是酶类标志物的一种,是神经母细胞瘤的特异性标志物,其在患有神经内分泌疾病的患者的血清中的含量较高,在腺癌患者中也有表达。CYFRA21-1是一种主要用于检测肺癌的肿瘤标志物,主要存在于上皮起源的肿瘤细胞的包浆中,有研究结果显示,其对非小细胞肺癌具有重要的诊断价值,尤其是鳞癌[10]。SCCA作为鳞癌的特异性肿瘤标志物,在肺、子宫、子宫颈、头颈等鳞状上皮细胞癌中均能异常表达,但也有研究结果显示,其在肝硬化、肺炎等病变中的水平也明显增高[11]。在本研究中,肺癌组患者血清NSE、CEA、CA125、CYFRA21-1、SCCA水平均高于对照组,且差异有统计学意义(均P<0.001),可见这5种血清肿瘤标志物能够用于肺癌的初步诊断。本研究还发现,不同病理类型的肺癌患者间,部分血清肿瘤标志物水平之间的差异无统计学意义,这说明肿瘤标志物的水平可能与肺癌发生的部位无关。但是,陈鹏等[12]研究发现,血清肿瘤标志物水平与不同病理类型有关,小细胞癌的血清NSE水平高于腺癌和鳞癌,腺癌患者的血清CEA水平高于鳞癌和小细胞癌,鳞癌患者的血清SCCA水平高于腺癌和小细胞癌,且差异有统计学意义(均P<0.05),这说明不同指标对不同病理类型的肺癌有特异性。虽然血清肿瘤标志物在癌症的诊断及预后评估中有一定价值,但目前研究者普遍认为其易受其他多种因素的影响,导致出现假阳性或假阴性结果,即使是多种血清肿瘤标志物联合检测,其结果仍不够理想[13]。随着科学技术的发展,影像手段在肺癌的诊断上也逐渐普及。在肺癌的诊断方面,CT发挥着重要作用,但是,CT具有增强扫描时间难以把握、扫描范围受限、特异度较低等缺点,因此,现在大多数研究者建议将影像方法与血清肿瘤标志物联合,以提高对肺癌诊断的准确率[14-15]。
在本研究中,我们分析了HRCT+5种血清肿瘤标志物联合检测诊断肺癌的效能,结果显示,联合诊断的灵敏度、特异度和准确率均高于各项单独检测,且差异有统计学意义(均P<0.001),这说明HRCT+5种血清肿瘤标志物联合检测可有效提高对肺癌的诊断灵敏度及准确率。这与Li等[16]的研究结果一致。分析原因后,我们认为这与以下原因有关:首先,血清肿瘤标志物水平检测的侧重点不同,各有所长,联合诊断可以提高诊断的准确率;其次,CT检查能明确肿瘤与周围组织的关系,HRCT更能显示病变的细微结构,使诊断证据更充足,可为临床诊断提供影像基础。但是本研究为回顾性分析,样本量较小,结果容易产生偏倚,有待后续大样本量数据来验证。
综上,在早期肺癌患者中,血清肿瘤标志物NSE、CEA、CA125、CYFRA21-1、SCCA均呈高表达状态,且联合HRCT可有效提高对肺癌诊断的准确率,有助于指导临床医师制定合适的治疗方案,继而改善患者预后。
利益冲突 所有作者声明无利益冲突
作者贡献声明 周楠负责命题的提出、研究的设计、论文的撰写;李囡馨、朱华晨负责现场试验的开展、数据的统计分析;岳英明负责数据的收集、整理与分析
高分辨率CT联合多种血清肿瘤标志物对不同病理类型早期肺癌的诊断价值
Diagnostic value of high resolution CT scan combined with various serum tumor markers in different pathological types of early lung cancer
-
摘要:
目的 探讨高分辨率CT(HRCT)联合多种血清肿瘤标志物对不同病理类型早期肺癌的诊断价值。 方法 回顾性分析2019年8月至2021年10月首都医科大学附属北京天坛医院收治的164例早期肺癌患者(肺癌组)的临床资料,同时选取同期参加体检的78名健康者作为对照组。肺癌组男性94例、女性70例,年龄(52.4±3.6)岁;其中,腺癌90例、鳞癌62例、小细胞癌12例。所有患者均经组织病理学检查结果证实。比较2组间和不同病理类型患者间血清神经元特异性烯醇化酶(NSE)、癌胚抗原(CEA)、鳞状细胞癌抗原(SCCA)、细胞角蛋白19片段抗原21-1(CYFRA21-1)、糖类抗原125(CA125)水平;以组织病理学检查结果为确诊肺癌的“金标准”,分别计算HRCT、5种肿瘤标志物、HRCT+5种肿瘤标志物诊断肺癌的灵敏度、特异度和准确率。计量资料的比较采用两独立样本t检验,计数资料的比较采用χ2检验。 结果 肺癌组的各项血清肿瘤标志物水平均高于对照组,且差异有统计学意义(t=8.107~30.460,均P<0.001)。腺癌患者的血清CEA、CA125水平均高于鳞癌和小细胞癌患者,且差异有统计学意义(t=12.712~4.326,均P<0.001)。鳞癌患者的血清CEA、CA125水平与小细胞癌患者的相比,差异均无统计学意义(t=1.342、1.256,均P>0.05)。腺癌与鳞癌患者的NSE和CYFRA21-1水平均低于小细胞癌患者,且差异有统计学意义(t=10.342~11.534,均P<0.001)。HRCT+5种肿瘤标志物联合检测对肺癌的诊断灵敏度为95.62%,特异度为96.43%,准确率为98.65%,均高于任意一种单独诊断(χ2=55.823、48.652、62.718,均P<0.001)。 结论 HRCT联合多种血清肿瘤标志物检测有助于提高对早期肺癌的诊断特异度、灵敏度和准确率,对肺癌的诊断具有较高的临床价值。 -
关键词:
- 体层摄影术,X线计算机 /
- 肺肿瘤 /
- 生物标记,肿瘤
Abstract:Objective To investigate the diagnostic value of high resolution CT (HRCT) scan combined with various serum tumor markers in different pathological types of early lung cancer. Methods The clinical data of 164 patients with early lung cancer (lung cancer group) admitted to Beijing Tiantan Hospital, Capital Medical University from August 2019 to October 2021 were analyzed retrospectively, and 78 healthy people who participated in physical examination at the same time were selected as the control group. There were 94 males and 70 females in the lung cancer group, aged (52.4±3.6) years. Among them, 90 cases were adenocarcinoma, 62 cases were squamous cell carcinoma, and 12 cases were small cell carcinoma. All the patients were confirmed by histopathological examination. The serum levels of neuron-specific enolase (NSE), carcinoembryonic antigen (CEA), cyto-keratin 19 fragment antigen 21-1, squamous cell carcinoma antigen and carbohydrate antigen 125 (CA125) were compared between the two groups and different pathological types of patients. The sensitivity, specificity and accuracy of HRCT scan, five tumor markers and HRCT scan + five tumor markers in the diagnosis of lung cancer were calculated based on the results of histopathological examination as the gold standard. The measurement data were compared by two independent samples t-test, and the counting data were compared by Chi-square test. Results The levels of serum tumor markers in lung cancer group were higher than those in control group (t=8.107–30.460; all P<0.001). The serum CEA and CA125 levels in patients with adenocarcinoma were higher than those in squamous cell carcinoma and small cell carcinoma (t=12.712–4.326; all P<0.001). No significant difference was found in serum CEA and CA125 levels between patients with squamous cell carcinoma and those with small cell carcinoma (t=1.342, 1.256; both P>0.05). The NSE level in patients with adenocarcinoma and squamous cell carcinoma was lower than that in patients with small cell carcinoma (t=10.342–11.534; all P<0.001). The sensitivity, specificity and accuracy of HRCT scann + five tumor markers in the diagnosis of lung cancer were 95.62%, 96.43% and 98.65% respectively, which were higher than any single diagnosis (χ2=55.823, 48.652, 62.718; all P<0.001). Conclusion HRCT scan combined with detection of various serum tumor markers can improve the sensitivity, specificity and accuracy of early lung cancer diagnosis, and this method has high clinical value in the diagnosis of lung cancer. -
Key words:
- Tomography, X-ray computed /
- Lung neoplasms /
- Biomarkers, tumor
-
表 1 肺癌组患者与对照组健康者的血清肿瘤标志物水平比较(
$ \bar{x}\pm s $ Table 1. Comparison of serum tumor markers between lung cancer group and control group (
$ \bar{x}\pm s $ 组别 CEA(ng/ml) NSE(ng/ml) CA125(U/ml) CYFRA21-1(ng/ml) SCCA(μg/L) 肺癌组(n=164) 45.45±4.51 23.52±3.56 124.23±29.43 16.78±2.56 7.36±2.56 对照组(n=78) 2.14±0.58 10.23±1.62 20.37±1.26 1.63±2.12 1.13±0.48 t值 30.460 8.107 14.619 10.363 14.532 P值 <0.001 <0.001 <0.001 <0.001 <0.001 注:CEA为癌胚抗原;NSE为神经元特异性烯醇化酶;CA125为糖类抗原125; CYFRA21-1为细胞角蛋白19片段抗原21-1;SCCA为鳞状细胞癌抗原 表 2 不同病理类型早期肺癌患者的血清肿瘤标志物水平比较(
$ \bar{x}\pm s $ Table 2. Comparison of serum tumor markers in patients with early lung cancer of different pathological types (
$ \bar{x}\pm s $ 病理类型 CEA(ng/ml) NSE(ng/ml) CA125(U/ml) CYFRA21-1(ng/ml) SCCA(μg/L) 腺癌(n=90) 128.24±34.46ab 23.24±3.54b 147.36±24.55ab 7.05±2.61b 5.14±2.25 鳞癌(n=62) 34.82±12.89 20.13±3.11b 107.53±18.24 12.57±2.49b 10.24±3.08 小细胞癌(n=12) 36.06±14.24 70.81±20.36 109.65±13.20 45.72±12.36 4.89±2.13 注:a表示与鳞癌患者相比,差异均有统计学意义(t=12.712、4.326,均P<0.001);b表示与小细胞癌患者相比,差异均有统计学意义(t=6.304~15.134,均P<0.001)。CEA为癌胚抗原;NSE为神经元特异性烯醇化酶;CA125为糖类抗原125; CYFRA21-1为细胞角蛋白19片段抗原21-1;SCCA为鳞状细胞癌抗原 表 3 不同检测方法及其联合应用对早期肺癌的诊断效能(%)
Table 3. Diagnostic efficacy of different examination methods and their combination in early lung cancer (%)
检测方法 灵敏度 特异度 准确率 CEA 42.56 88.65 64.00 NSE 43.41 91.78 67.05 CYFRA21-1 46.52 80.46 63.21 CA125 45.21 86.84 65.23 SCCA 45.02 86.95 64.00 HRCT 76.50 79.28 78.46 5种肿瘤标志物联合检测 68.26 74.84 72.32 HRCT+5种肿瘤标志物联合检测 95.62 96.43 98.65 注:CEA为癌胚抗原;NSE为神经元特异性烯醇化酶;CYFRA21-1为细胞角蛋白19片段抗原21-1;CA125为糖类抗原125; SCCA为鳞状细胞癌抗原;HRCT为高分辨率计算机体层摄影术 -
[1] Chen WQ, Zheng RS, Baade PD, et al. Cancer statistics in China, 2015[J]. CA Cancer J Clin, 2016, 66(2): 115−132. DOI: 10.3322/caac.21338. [2] Lim J, Shin KM, Lee KS, et al. Relationship between emphysema severity and the location of lung cancer in patients with chronic obstructive lung disease[J]. AJR Am J Roentgenol, 2015, 205(3): 540−545. DOI: 10.2214/AJR.14.13992. [3] Akcay M, Etiz D, Duruer K, et al. Dosimetric comparison of single-arc/partial-arc volumetric modulated arc therapy and intensity-modulated radiotherapy for peripheral and central lung cancer[J]. J Cancer Res Ther, 2021, 17(1): 80−87. DOI: 10.4103/jcrt.JCRT_221_19. [4] Nooreldeen R, Bach H. Current and future development in lung cancer diagnosis[J/OL]. Int J Mol Sci, 2021, 22(16): 8661[2022-02-27]. https://www.mdpi.com/1422-0067/22/16/8661. DOI: 10.3390/ijms22168661. [5] Hoy H, Lynch T, Beck M. Surgical treatment of lung cancer[J]. Crit Care Nurs Clin North Am, 2019, 31(3): 303−313. DOI: 10.1016/j.cnc.2019.05.002. [6] Wu FY, Wang L, Zhou CC. Lung cancer in China: current and prospect[J]. Curr Opin Oncol, 2021, 33(1): 40−46. DOI: 10.1097/CCO.0000000000000703. [7] Cao J, Yuan P, Wang Y, et al. Survival rates after lobectomy, segmentectomy, and wedge resection for non-small cell lung cancer[J]. Ann Thorac Surg, 2018, 105(5): 1483−1491. DOI: 10.1016/j.athoracsur.2018.01.032. [8] Veronesi G, Bottoni E, Finocchiaro G, et al. When is surgery indicated for small-cell lung cancer?[J]. Lung Cancer, 2015, 90(3): 582−589. DOI: 10.1016/j.lungcan.2015.10.019. [9] Yang ZM, Ding XP, Pen L, et al. Analysis of CEA expression and EGFR mutation status in non-small cell lung cancers[J]. Asian Pac J Cancer Prev, 2014, 15(8): 3451−3455. DOI: 10.7314/apjcp.2014.15.8.3451. [10] Chen H, Jiang Y, Jia KY, et al. Prognostic significance of postoperative longitudinal change of serum carcinoembryonic antigen level in patients with stage I lung adenocarcinoma completely resected by single-port video-assisted thoracic surgery: a retrospective study[J]. Transl Lung Cancer Res, 2021, 10(10): 3983−3994. DOI: 10.21037/tlcr-21-833. [11] Qu T, Zhang JW, Xu N, et al. Diagnostic value analysis of combined detection of Trx, CYFRA21-1 and SCCA in lung cancer[J]. Oncol Lett, 2019, 17(5): 4293−4298. DOI: 10.3892/ol.2019.10073. [12] 陈鹏, 宋长祥, 陆武, 等. SPECT同机定位CT引导经皮肺穿刺活检术联合肿瘤标志物检测对周围型肺癌的诊断价值[J]. 国际放射医学核医学杂志, 2018, 42(1): 21−24, 29. DOI: 10.3760/cma.j.issn.1673-4114.2018.01.004.
Chen P, Song CX, Lu W, et al. Diagnostic value of lung biopsy through SPECT with CT-guided percutaneous paracentesis and tumor markers for peripheral lung cancer[J]. Int J Radiat Med Nucl Med, 2018, 42(1): 21−24, 29. DOI: 10.3760/cma.j.issn.1673-4114.2018.01.004.[13] Wu HM, Wang QM, Liu QH, et al. The serum tumor markers in combination for clinical diagnosis of lung cancer[J]. Clin Lab, 2020, 66(3): 269−276. DOI: 10.7754/Clin.Lab.2019.190533. [14] Shan YQ, Yin XY, Zhao N, et al. Comparison of serum tumor markers combined with dual-source CT in the diagnosis of lung cancer[J]. Minerva Med, 2021. DOI: 10.23736/S0026-4806.21.07124-X. [15] Schneider J. Early detection of lung cancers — comparison of computed tomography, cytology and fuzzy-based tumor markers panels[J]. Cancer Biomark, 2010, 6(3/4): 149−162. DOI: 10.3233/CBM-2009-0126. [16] Li SL, Ma YY, Xiong Y, et al. Five tumor-associated autoantibodies expression levels in serum predict lung cancer and associate with poor outcome[J]. Transl Cancer Res, 2019, 8(4): 1364−1373. DOI: 10.21037/tcr.2019.07.25. -






 下载:
下载: