-
辐射适应性反应(adaptive response,AR)是靶物质(如细胞)先暴露于一个较低的辐射剂量,间隔一段时间后,再接受较大剂量(效应剂量)照射,靶细胞对大剂量照射引起的生物效应产生抵抗[1]。低剂量辐射诱导的AR为当前放射生物学研究的热点。微RNA(microRNAs,miRNA)作为内源性非编码小RNA分子,广泛参与调节细胞分化、增殖、凋亡及细胞免疫应答等重要生物过程[2]。有研究者发现,部分miRNA通过调控DNA双链断裂修复,抑制肿瘤细胞增殖和转移,增强肿瘤细胞的辐射敏感性[3-4];或通过调节细胞凋亡相关信号通路上下游分子影响电离辐射诱导的细胞凋亡,发挥生物功能[5-6]。尽管已有研究结果表明,miRNA可以调节电离辐射生物效应,其在不同细胞中的表达水平与电离辐射敏感性有关,但目前尚未见辐射诱导AR相关的miRNA研究报道。辐射诱导AR相关miRNA研究及其调控机制的阐明,对辐射防护有一定理论参考价值,而且筛选到的miRNA还可能作为生物标志物为临床肿瘤放射治疗方案的制定提供参考依据。本研究探索X射线辐射诱导非小细胞肺癌(non-small-cell lung cancer,NSCLC)A549细胞凋亡的AR,并通过基因芯片筛选相关的miRNA,运用生物信息学分析miRNA调控的靶基因及信号通路,初步探索辐射AR的可能机制。
-
NSCLC A549细胞购自中国医学科学院基础医学研究所细胞中心,培养代次为16~27代。DMEM培养基、胎牛血清、0.25%胰酶均购自美国GIBCO公司。5×106个/瓶A549细胞培养于含10%胎牛血清、10%青链霉素的(北京索莱宝科技有限公司)DMEM完全培养基中,培养条件为37℃、5% CO2,培养24 h后进行照射。
-
使用X射线辐照仪[美国Precision X-ray(PXi)公司,型号X-RAY 225]进行细胞照射,剂量率分别为0.38 Gy/min(低剂量50、200 mGy照射时源距96 cm)和0.96 Gy/min(效应剂量20 Gy照射时源距60 cm)。将A549细胞分为6组,包括50 mGy+20 Gy、200 mGy+20 Gy、20 Gy、50 mGy、200 mGy照射组及对照组(0 Gy),前2组细胞分别用50、200 mGy初始剂量进行照射,培养6 h后用20 Gy的效应剂量进行照射,同时,20 Gy、50 mGy、200 mGy照射组分别用20 Gy、50 mGy、200 mGy进行照射,对照组不照射。
-
采用碘化丙啶(PI)/Annexin-V双染法(美国BD公司)进行细胞凋亡检测。不同剂量照射A549细胞后培养24 h,用0.25%胰酶进行消化,210×g离心5 min后,用PBS重悬细胞,洗2次,用100 µL 1×Annexin V缓冲液(美国BD公司)重悬细胞。按照实验设计加入PI、Annexin-V染料,4℃染色30 min。染色后加入PBS,4℃、210×g离心5 min,洗2次,细胞用400目尼龙膜过滤,用流式细胞仪(美国BD公司Accuri C6 Plus型)进行细胞凋亡的检测。
-
用TRIzol试剂(北京索莱宝科技有限公司)裂解照射后的A549细胞,按照试剂盒的说明提取细胞总RNA。每个样本提取10 μg总RNA,利用NanoDrop(美国LabTech公司)进行RNA质检。质检合格的样品用于后续miRNA测序分析。使用Small RNA Sample Pre Kit(美国Illumina公司)构建文库,利用miRNA的3′及5′端特殊结构,直接将RNA两端加上接头,反转录合成cDNA。进行PCR扩增,电泳分离目标DNA片段,切胶回收得到cDNA文库。质检合格后的文库按照有效浓度进行HiSeq/MiSeq测序。本研究miRNA测序及前期数据分析均由天津诺禾致源公司完成。
-
对各样品中miRNA进行表达量的统计,并用TPM(transcript per million)进行表达量归一化处理[7]。聚类图由cluster 3.0软件生成,由总信号密度大于1000的miRNA生成。用火山图推断差异miRNA的整体分布情况,从差异倍数和校正后的显著水平(q<0.05)进行评估,筛选差异miRNA[8-9],筛选条件为q<0.01、log2(差异倍数)>1。采用miRanda、RNAhybrid软件对各比较组之间差异表达miRNA靶基因进行预测,并对miRNA靶基因进行基因本体(gene ontology,GO)功能注释分析。GO富集分析方法为GO seq,其能准确地计算出GO条目被候选靶基因富集的概率[10]。京都基因与基因组百科全书(Kyoto encyclopedia of genes and genomes,KEGG)通路显著性富集分析是以KEGG通路为单位,应用超几何检验,与整个基因组背景相比,找出在候选靶基因中显著性富集的通路[11]。对候选靶基因中显著性富集的信号通路采用Benjamini&Hochberg(BH)的方法对P值进行校正(P<0.05为差异有统计学意义,即显著性富集)。
-
为了验证miRNA测序结果,随机挑选10个50 mGy+20 Gy照射组和200 mGy+20 Gy照射组差异表达趋势相同的miRNA,采用实时荧光定量PCR(quantitative real-time PCR,qRT-PCR)验证其表达水平。使用SuperTaq聚合酶(美国应用生物系统公司)和MicroRNAs qPCR SYBR Green试剂盒(上海生工生物工程有限公司)进行检测。使用miRNA特异性引物从总RNA中扩增获取cDNA,其引物见表1。使用qRT-PCR仪(美国应用生物系统公司 7500fast型)进行分析。Ct值被定义为荧光通过固定阈值的分数循环数。U6作为内参基因,各组间的miRNA相对表达水平用2−△△Ct计算[12]。
引物名称 引物序列(5′→3′) miR-92a-3p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACACAGGC miR-92a-3p-F GCGTATTGCACTTGTCCCG miR-501-3p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAGAATC miR-501-3p-F GAATGCACCCGGGCAAG miR-582-5p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAGTAAC miR-582-5p-F CGCGTTACAGTTGTTCAACCA miR-1260b-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACATGGTG miR-1260b-F CGCGATCCCACCACTGC miR-138-5p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCGGCCT miR-138-5p-F GCGAGCTGGTGTTGTGAATC miR-3662-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCATCAG miR-3662-F CGCGGAAAATGATGAGTAGTGA miR-185-3p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACGACCAG miR-185-3p-F CGAGGGGCTGGCTTTCCT miR-1307-5p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAGCCGG miR-1307-5p-F CGTCGACCGGACCTCGA miR-182-3p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACTAGTTG miR-182-3p-F GCGCGTGGTTCTAGACTTGC miR-1908-5p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACGACCAA miR-1908-5p-F TAATTTCGGCGGGGACGGCGA all-reverse AGTGCAGGGTCCGAGGTATT U6-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAAAATA U6-F GCGCGTCGTGAAGCGTTC U6-R GTGCAGGGTCCGAGGT 注:RNA为核糖核酸;PCR为聚合酶链式反应 表 1 微RNA的实时荧光定量PCR引物序列
Table 1. The primers for quantitative real-time PCR analysis of the microRNAs
-
应用SPSS 20.0软件进行统计学分析。符合正态分布的数据以
$\bar x $ ±s表示,采用Welch t检验进行2组间的比较。P<0.05为差异有统计学意义。 -
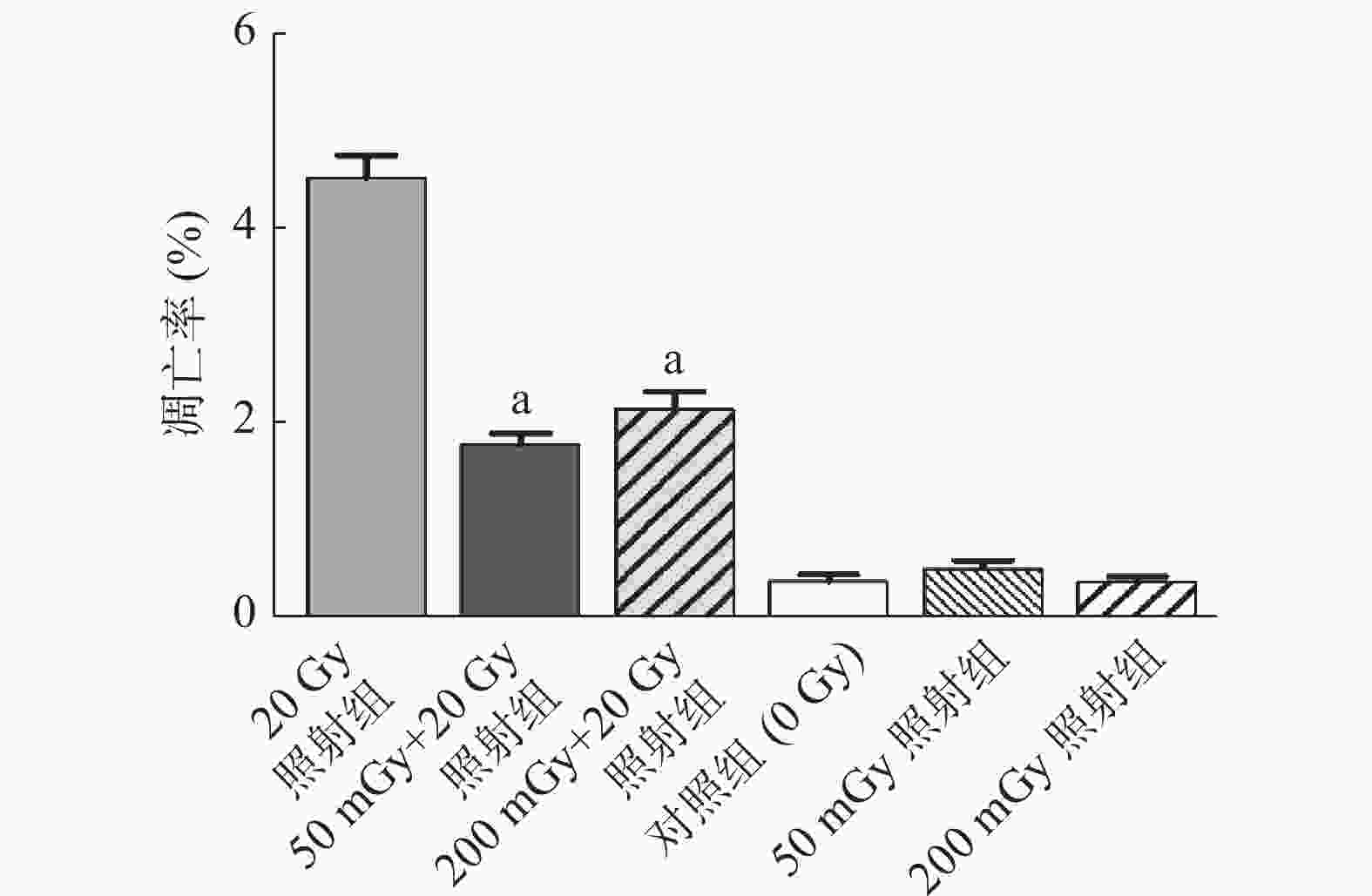
50 mGy+20 Gy照射组和200 mGy+20 Gy照射组的A549细胞早期凋亡率分别为(1.81±0.11)%和(2.17±0.19)%,低于20 Gy照射组的(4.54±0.23)%,且差异均有统计学意义(均P<0.01);50 mGy照射组、200 mGy照射组的细胞凋亡率与对照组比较,差异均无统计学意义(t=1.165、0.078,均P>0.05)(图1)。以上结果表明,50 mGy和200 mGy照射剂量的X射线均能诱导A549细胞凋亡的AR。
-
本研究中50 mGy+20 Gy、200 mGy+20 Gy、20 Gy照射组鉴定到的成熟miRNA的个数分别为649、681、905个,差异表达miRNA聚类分析热图见图2。与20 Gy照射组相比,50 mGy+20 Gy照射组上调miRNA有7个、下调miRNA有36个;200 mGy+20 Gy照射组上调miRNA有23个、下调miRNA有40个(图3)。与20 Gy照射组相比,50 mGy+20 Gy照射组、200 mGy+20 Gy照射组共同差异表达的miRNA有1个上调、15个下调(表2)。
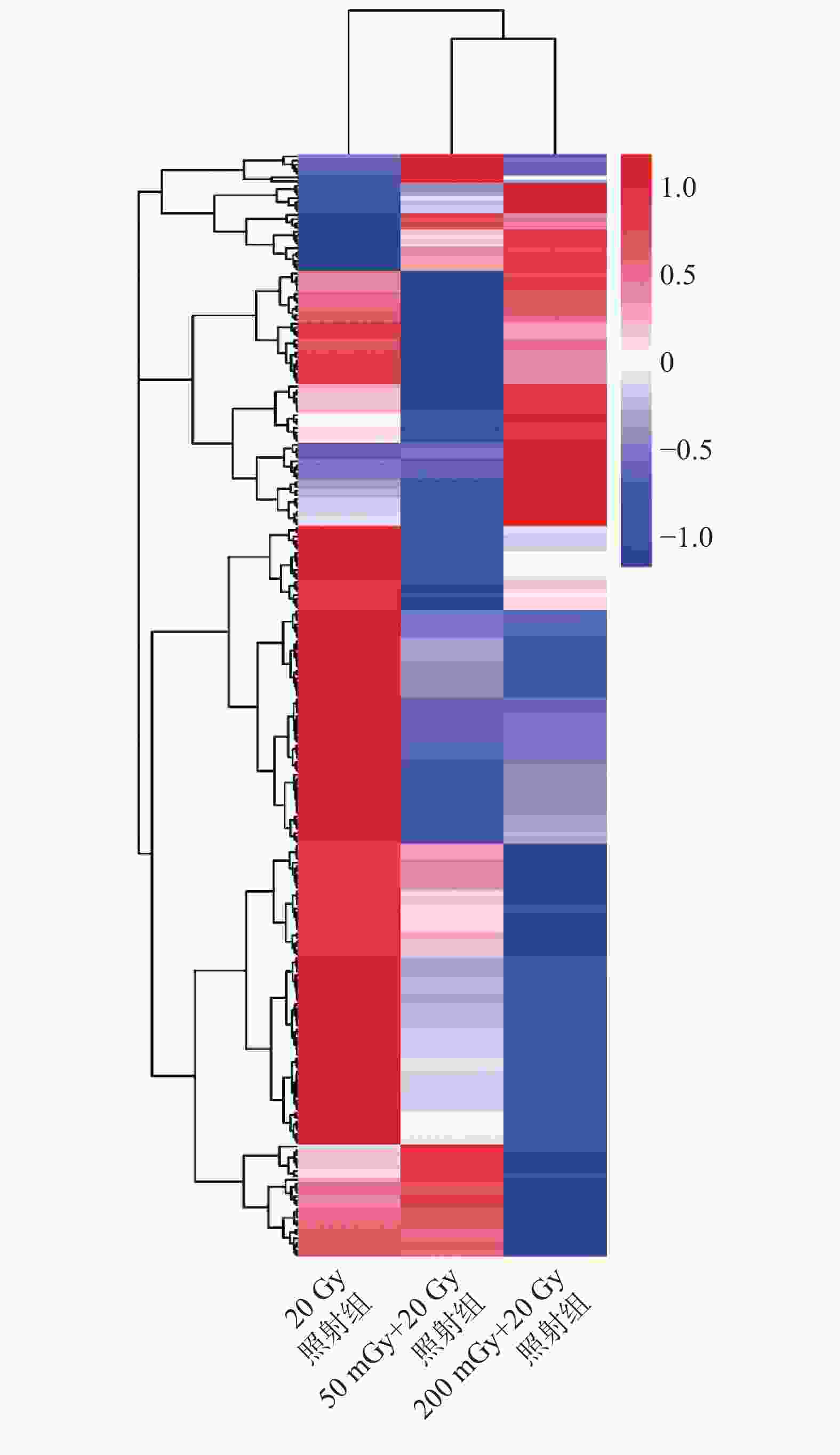
图 2 X射线照射后3组非小细胞肺癌A549细胞差异表达miRNA的聚类分析热图
Figure 2. The heatmap of unsupervised hierarchical clustering of microRNAs expression in three groups of non-small-cell lung cancer A549 cells after X-ray irradiation
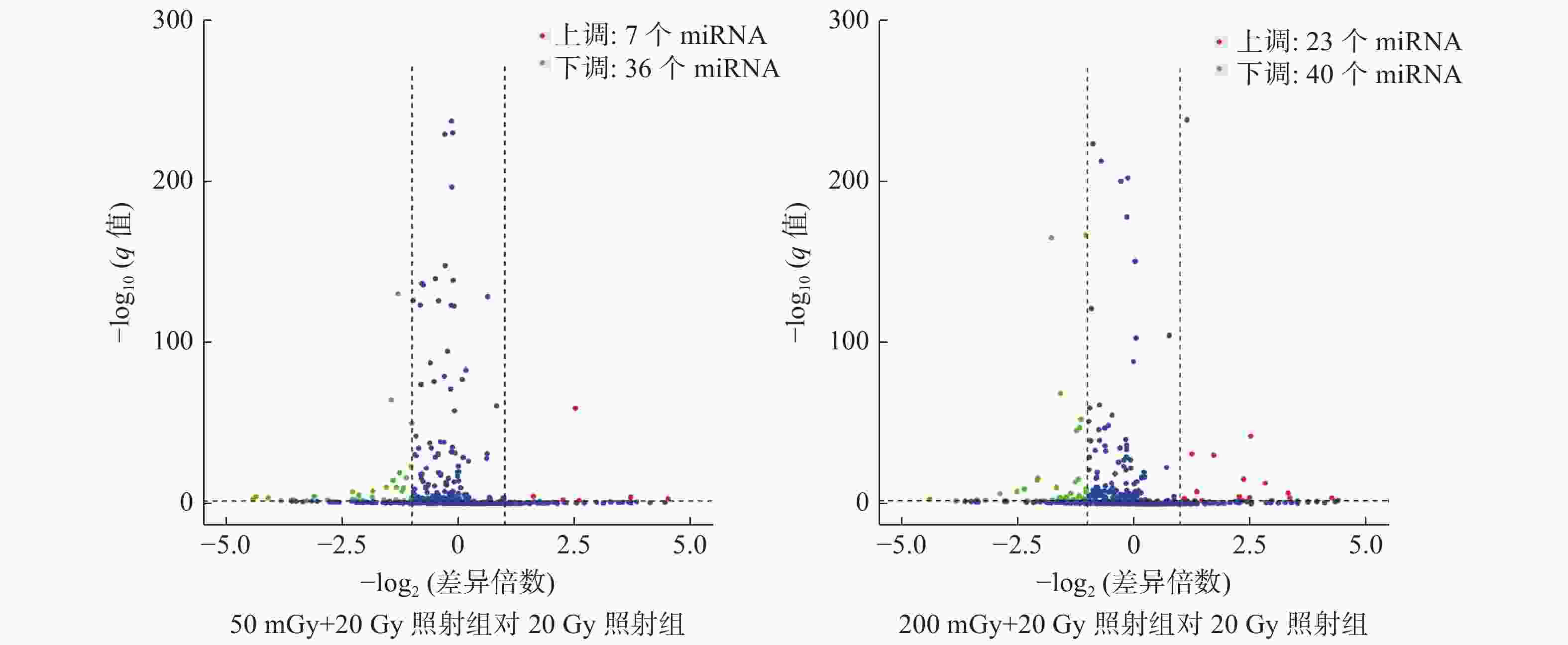
图 3 X射线照射后3组非小细胞肺癌A549细胞的差异miRNA火山图
Figure 3. The volcano plot of microRNAs expression in three groups of non-small-cell lung cancer A549 cells after X-ray irradiation
miRNA名称 位置(染色体上) 20 Gy照射组(A) 50 mGy+20 Gy照射组(B) 200 mGy+20 Gy照射组(C) log2(B/A) log2(C/A) miR-92a-3p 13q31.3 7712.85 3157.42 3396.89 −1.29 −1.19 miR-193b-3p 16p13.12 1291.15 523.84 379.80 −1.30 −1.77 miR-484 16p13.11 572.06 210.49 256.39 −1.44 −1.16 miR-1307-5p 10q24.33 9.66 0 1.06 −3.60 −3.19 miR-182-3p 7q32.2 7.11 0 0 −3.83 −3.83 miR-127-3p 14q32.2 652.59 326.11 297.88 −1.00 −1.14 miR-1303 5q33.2 106.58 47.04 25.53 −1.18 −2.07 miR-138-5p 3p21.22/16p13.3 54.29 15.15 10.64 −1.84 −2.36 miR-185-3p 22q11.21 42.63 8.77 7.45 −2.28 −2.52 miR-654-5p 14q32.31 32.25 15.95 10.64 −1.02 −1.61 miR-3662 6q23.3 2.00 26.31 15.96 3.71 2.99 miR-378d 4p16.2/8q22.1 28.79 7.97 11.70 −1.85 −1.30 miR-1908-5p 11q12.2 10.57 2.39 0 −2.14 −4.41 miR-582-5p 5q12.1 24.23 9.57 8.51 −1.34 −1.52 miR-501-3p Xp11.23 98.38 39.07 23.40 −1.33 −2.08 miR-1260b 11q21 83.99 28.70 26.60 −1.55 −1.66 注:A、B、C均为miRNA相对表达水平。miRNA为微RNA 表 2 X射线照射后非小细胞肺癌A549细胞的16个差异表达的miRNA
Table 2. Sixteen microRNAs differentially expressed in non-small cell lung cancer A549 cells after X-ray irradiation
-
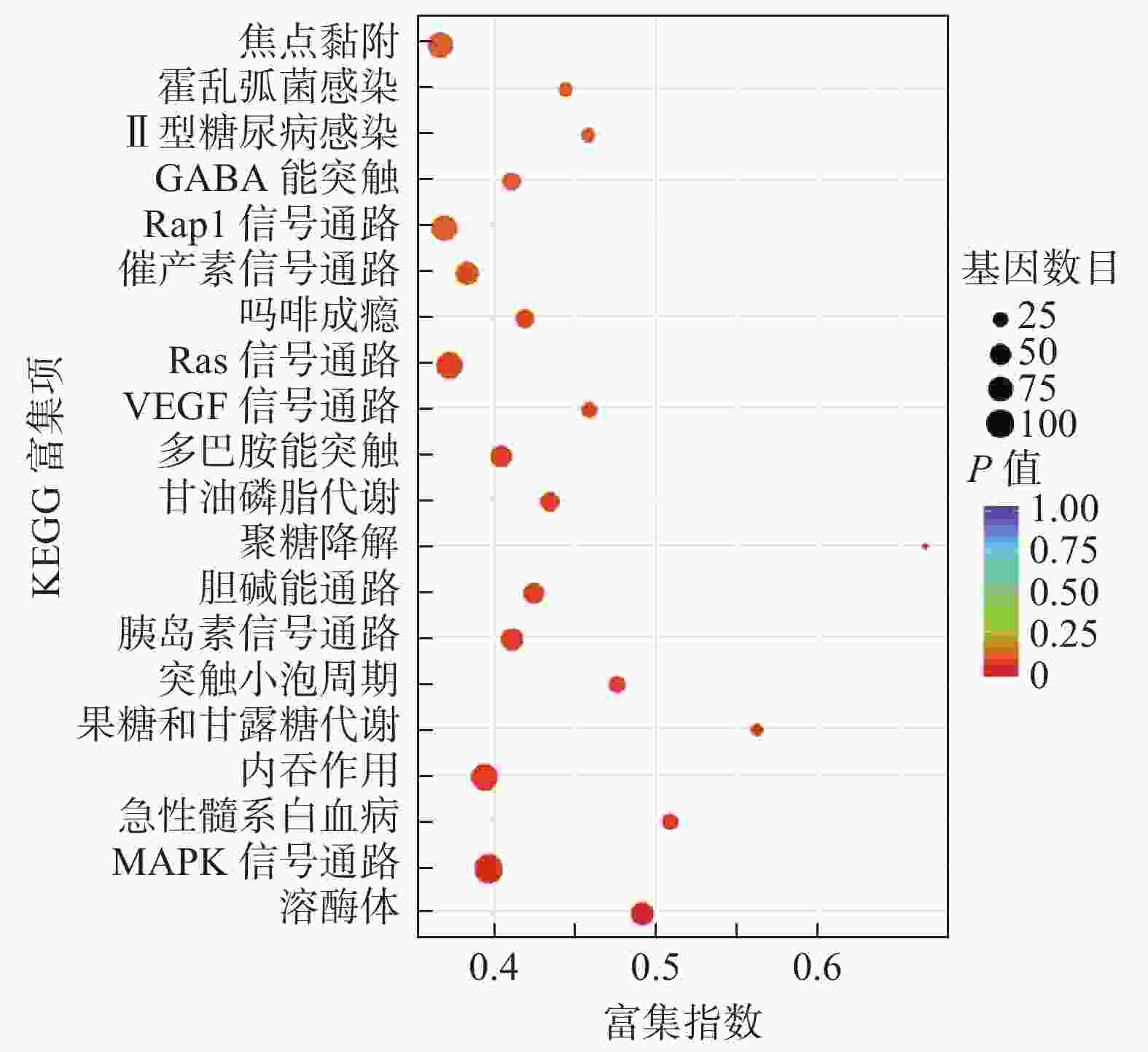
miRNA靶基因进行GO功能注释分析结果发现,共同差异表达miRNA调控靶基因注释功能最多的是生物过程,其次为细胞组分及分子功能(表3)。对miRNA调控的靶基因进行KEGG信号通路分析,结果发现,共同差异表达miRNA调控的靶基因显著富集于溶酶体、丝裂原活化蛋白激酶(mitogen-activated protein kinase,MAPK)信号通路、内吞作用、血管内皮生长因子信号通路、Ras信号通路和Rap1信号通路等(图4)。
GO类别 GO富集描述 基因数 P值 生物过程 细胞通讯调节(regulation of cell communication) 1166 <0.01 代谢过程的正调节(positive regulation of metabolic process) 1271 <0.01 信号的调节(regulation of signaling) 1147 <0.01 代谢过程(metabolic process) 3472 <0.01 单体过程(single-organism process) 4178 <0.01 细胞组分 细胞质(cytoplasm) 3474 <0.01 细胞内(intracellular) 4285 <0.01 细胞内组分(intracellular part) 4183 <0.01 细胞质组分(cytoplasmic part) 2954 <0.01 细胞器(organelle) 3860 <0.01 分子功能 蛋白质结合(protein binding) 3480 <0.01 结合(binding) 4305 <0.01 酶结合(enzyme binding) 672 <0.01 催化活性(catalytic activity) 1811 <0.01 序列特异性DNA结合(sequence-specific DNA binding) 369 <0.01 注:GO为基因本体 表 3 X射线照射后非小细胞肺癌A549细胞差异表达微RNA调控靶基因的GO富集分析(前15位)
Table 3. Gene ontology enrichment analysis of target genes regulated by differentially expressed microRNAs in non-small cell lung cancer A549 cells after X-ray irradiation (TOP 15)
-
miRNA表达情况与基因芯片结果趋势一致(图5)。50 mGy+20 Gy照射组和200 mGy+20 Gy照射组共同表达上调的miR-3662与20 Gy组相比,差异均有统计学意义(t=10.55、5.97,均P<0.01)。芯片测序结果中下调的9个miRNA,包括miR-185-3p、miR-1908-5p、miR-1307-5p、miR-182-3p、miR-92a-3p、miR-582-5p、miR-501-3p、miR-138-5P及miR-1260b,在50 mGy+20 Gy照射组均下调,且与20 Gy照射组比较,差异均有统计学意义(t=15.64~39.83,均P<0.01)。200 mGy+20 Gy照射组miR-138-5P、miR-1260b的表达水平与20 Gy照射组相当,其他7个miRNA表达水平均下调,且差异均有统计学意义(t=4.92~26.81,均P<0.05)。
-
随着放射生物学研究领域的发展,低剂量电离辐射诱导的AR越来越受到重视,并成为研究热点。目前,在动物活体及体外细胞实验均观察到低剂量辐射诱导的细胞凋亡及细胞周期的AR[13-14]。低剂量电离辐射诱导细胞凋亡的AR涉及的分子生物机制十分复杂,因此,深入探讨低剂量电离辐射诱导细胞凋亡的AR的规律和机制很有必要。本研究基于小RNA测序技术筛选与X射线辐射诱导细胞凋亡AR相关的miRNA,初步探索辐射诱导细胞凋亡的AR的可能机制。
据报道,行75 mGy X射线(12.5 mGy/min)照射后,间隔3~24 h,再进行1.5 Gy(287 mGy/min)照射,可诱导体外EL-4淋巴瘤细胞凋亡的AR[14]。Chen等[15]研究发现,50 mGy α粒子照射后可诱导NSCLC A549细胞对后续750 mGy α粒子照射产生放射抗性,即辐射AR。以往的研究结果显示,NSCLC A549经8 Gy X射线照射后,细胞凋亡率为4%~5%,为辐射不敏感细胞[16]。在本研究中,20 Gy X射线照射后A549细胞凋亡率为5%左右。本研究中初始剂量照射剂量率为0.38 Gy/min,且200 mGy照射剂量也处于目前低剂量辐射定义的临界点[17]。这提示低剂量辐射诱导A549细胞凋亡AR与照射剂量及剂量率可能均有关。
miRNA参与多种细胞分子功能的调节,细胞在接受电离辐射后,细胞内及血液中miRNA表达水平均发生变化,这提示miRNA在辐射生物效应的产生中发挥了重要调节作用,可能作为辐射生物效应产生的生物标志物[3-6]。本研究通过小RNA测序在50 mGy+20 Gy照射组和200 mGy+20 Gy照射组筛选到16个共同差异表达的miRNA,且qRT-PCR对部分miRNA的表达水平进行了验证。有研究结果表明,miR-3662在肺腺癌中的表达水平升高,并通过调控细胞外信号调节激酶(ERK)和c-Jun氨基末端激酶(JNK)信号通路抑制细胞增殖和诱导细胞周期阻滞[18]。另有研究结果显示,miR-3662靶向于在健康肺组织中高表达的基因PTAR1、NPR3和SEPT10。miR-3662下调则促进细胞增殖、分化,进而发展为肺腺癌,因此在肺腺癌的诊断中有重要意义[19]。而本研究中miR-3662是在2组产生凋亡AR的细胞中唯一共同上调表达的miRNA,这提示miR-3662可能在X射线辐射诱导A549细胞凋亡的AR中发挥了重要作用。
2组产生AR的细胞中共同下调的miRNA也在肺癌的发生、发展、转移中都发挥了重要的调控作用。例如,miR-182在NSCLC中与FBXW7、FBXW11的表达水平呈负相关,其过表达可促进肿瘤细胞增殖、集落形成、细胞周期进展,同时可抑制肿瘤细胞凋亡[20]。另有研究结果表明,过表达miR-182可通过调控RECK基因的表达,增强NSCLC A549和肺腺癌H1299细胞的增殖、侵袭和迁移能力,从而促进NSCLC的进展[21]。本研究中miR-182在辐射诱导细胞凋亡AR的NSCLC A549细胞中下调,这提示其可能在辐射诱导细胞凋亡的AR机制中发挥了负向调节作用。
为进一步研究共同差异表达的miRNA是否参与了辐射诱导细胞凋亡的AR的发生,我们对这些miRNA调控的靶基因进行功能注释及信号通路分析。结果显示,靶基因主要富集在溶酶体、MAPK信号通路、内吞作用、血管内皮生长因子信号通路、Ras信号通路及Rap1信号通路等。MAPK及其家族蛋白质,包括胞外调节蛋白激酶、应激活化蛋白激酶等,广泛存在于哺乳动物细胞并参与细胞分化、增殖和凋亡等过程[22-23]。多项研究结果表明,靶向Ras-MAPK信号通路的miRNA在肿瘤发生中发挥重要作用。如miR-382、miR-34a及miR-124能通过调节MAPK信号通路,抑制细胞黏附和细胞周期,并同时活化免疫系统相关通路[24-25]。Mutlu等[26]发现,miR-564通过ERK1/2 磷酸化调节MAPK信号通路,使细胞阻滞在G1期。有研究者发现,突变Ras等位基因的表达可以改变肿瘤细胞的敏感性,并在暴露于电离辐射后增加其存活率。在一些Ras活性和野生型P53细胞系中,抑制MEK可能通过P53诱导而致放射增敏[27]。而单次高剂量辐射,可通过调节Ras/Raf/MEK/ERK信号通路诱导细胞周期停滞和凋亡来介导其抗肿瘤作用[28]。本研究中miRNA调控的靶基因显著富集的MAPK、Ras信号通路等有可能在低剂量辐射诱导细胞凋亡的AR产生中有重要作用,有待在接下来的研究中进一步阐明。
在本研究中,我们发现低剂量X射线能诱导NSCLC A549细胞凋亡的AR。并通过小RNA测序筛选到一组差异表达的miRNA,其靶基因显著富集至MAPK、RAS等信号通路。这提示筛选出来的miRNA可能在低剂量辐射诱导的细胞凋亡AR机制中发挥重要作用,可能成为辐射效应的重要生物靶标,作为生物标志物应用于临床肿瘤放射治疗。本研究不仅对放射生物学的研究具有理论意义,而且对辐射防护和临床医学肿瘤放射治疗具有一定实用价值。未来,我们将对筛选到的miRNA在辐射诱导的AR中的调控机制进行深入研究,并在动物活体水平上进行验证。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 余小玲负责实验方法的建立和实施、数据的分析、文章的撰写;荣利、方芳负责实验的实施;丁晓文、王洪胜负责数据的分析;谢婷婷负责实验样本的准备、数据的整理;牛东升、王小春、李珏负责课题的设计和指导、论文的修订。
X射线诱导非小细胞肺癌A549细胞凋亡的适应性反应及相关miRNA筛选的研究
Adaptive response of NSCLC A549 cells induced by X-ray radiation and screening of related miRNAs
-
摘要:
目的 研究X射线辐射诱导非小细胞肺癌(NSCLC)A549细胞凋亡的适应性反应,并筛选适应性反应相关的微RNA(miRNA)。 方法 将NSCLC A549细胞分为6组,包括 50 mGy+20 Gy、200 mGy+20 Gy、20 Gy、50 mGy、200 mGy照射组及对照组(0 Gy),前2组细胞分别用50、200 mGy初始剂量进行照射,培养6 h后用20 Gy的效应剂量进行照射,20 Gy、50 mGy、200 mGy照射组同时进行照射。培养24 h后使用流式细胞仪检测细胞凋亡情况。利用小RNA测序技术筛选差异表达miRNA,并对其靶基因进行基因本体(GO)及京都基因与基因组百科全书(KEGG)通路的功能富集分析。采用实时荧光定量PCR(qRT-PCR)对部分差异表达miRNA进行验证。2组间数据的比较采用Welch t检验。 结果 50 mGy+20 Gy照射组和200 mGy+20 Gy照射组的A549细胞早期凋亡率分别为(1.81±0.11)%和(2.17±0.19)%,低于20 Gy照射组的(4.54±0.23)%,且差异均有统计学意义(t=10.680、8.006,均P<0.01)。与20 Gy照射组相比,50 mGy+20 Gy照射组和200 mGy+20 Gy照射组共同差异表达趋势miRNA有1个上调(miR-3662)、15个下调(miR-185-3p、miR-1908-5p、miR-1307-5p、miR-182-3p、miR-92a-3p、miR-582-5p、miR-501-3p、miR-138-5P、miR-1260b、miR-484、miR-378d、miR-193b-3P、miR-127-3p、miR-1303及miR-654-5p)。GO富集分析结果显示,差异表达miRNA调控靶基因功能显著富集于细胞通讯调节、代谢过程的正向调节、代谢信号的调节、酶结合及催化活性等过程。KEGG富集分析结果显示,靶基因相关信号通路显著富集于溶酶体、丝裂原活化蛋白激酶、Ras和内吞作用等信号通路。qRT-PCR检测结果显示,miRNA表达情况与基因芯片结果趋势一致(10个miRNA表达水平得到验证)。 结论 X射线50、200 mGy照射剂量均能诱导NSCLC A549细胞凋亡的适应性反应,并筛选到一组共同差异表达的miRNA,可能在X射线辐射诱导细胞凋亡的适应性反应中发挥了重要作用,有可能成为调节电离辐射生物效应的潜在靶点。 Abstract:Objective To study the adaptive response of the X-ray radiation to apoptosis of non-small cell lung cancer (NSCLC) A549 cell, and to screen microRNAs (miRNAs) related to the adaptive effect of radiation. Methods NSCLC A549 cells were irradiated in 6 groups, namely, the50 mGy+20 Gy, 200 mGy+20 Gy, 20 Gy, 50 mGy, and 200 mGy irradiation groups and the 0 Gy as control group. The cells in the first two groups were irradiated with initial doses of 50 mGy and 200 mGy respectively, and were then irradiated with an effective dose of 20 Gy after culturs for 6 hours. The 20 Gy, 50 mGy, and 200 mGy groups were irradiated at the same time. The apoptosis of A549 cellswas detected by flow cytometry after 24 hours of culturing. Small RNA sequencing was performing to detect the expression of miRNAs in different irradiation groups, and quantitative real-time PCR (qRT-PCR) was used to verify some differentially expressed miRNAs. The target genes of differentially expressed miRNAs were predicted and then analyzed via gene ontology (GO) and Kyoto encyclopedia of genes and genomes (KEGG) analyses. The data between two groups were compared by performing Welch's t-test. Results The cell apoptosis rates of A549 cells in the 50 mGy+20 Gy and 200 mGy+20 Gy groups were (1.81±0.11)% and (2.17±0.19)% respectively, which were significantly lower than (4.54±0.23)% in the 20 Gy group (t=10.680, 8.006; all P<0.01). Meanwhile, the small RNA sequencing results show that compared with the 20 Gy irradiation group, 1 miRNA (miR-3662) was up-regulated and 15 miRNAs (miR-185-3p, miR-1908-5p, miR-1307-5p, miR-182-3p, miR-92a-3p, miR-582-5p, miR-501-3p, miR-138-5P, miR-1260b, miR-484, miR-378d, miR-193b-3P, miR-127-3p, miR-1303 and miR-654-5p) were down-regulated in the 50 mGy+20 Gy and 200 mGy+20 Gy irradiation groups. GO enrichment analysis reveal that the functions of target genes were significantly enriched in cell communication regulation, positive regulation of the metabolic process, regulation of the metabolic signal, enzyme binding and catalytic activity, whereas the KEGG analysis showed that the target genes related signal pathways were significantly enriched in lysosome, mitogen-activated protein kinase, Ras, endocytosis and other signal pathways. qRT-PCR was used to validate the expression of miRNAs, and the expression levels of 10 miRNAs were consistent with the sequencing results. Conclusions The X-ray of 50 mGy and 200 mGy radiation dose can induce an adaptive response in the cell apoptosis of A549 cells. A group of differently expressed miRNAs detected in 50 mGy+20 Gy and 200 mGy+20 Gy groups may play important roles in the adaptive response of low dose radiation and may become potential targets to regulate have effects of radiation. -
Key words:
- Carcinoma, non-small-cell lung /
- X-rays /
- Apoptosis /
- MicroRNAs /
- Adaptive response
-
表 1 微RNA的实时荧光定量PCR引物序列
Table 1. The primers for quantitative real-time PCR analysis of the microRNAs
引物名称 引物序列(5′→3′) miR-92a-3p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACACAGGC miR-92a-3p-F GCGTATTGCACTTGTCCCG miR-501-3p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAGAATC miR-501-3p-F GAATGCACCCGGGCAAG miR-582-5p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAGTAAC miR-582-5p-F CGCGTTACAGTTGTTCAACCA miR-1260b-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACATGGTG miR-1260b-F CGCGATCCCACCACTGC miR-138-5p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCGGCCT miR-138-5p-F GCGAGCTGGTGTTGTGAATC miR-3662-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACCATCAG miR-3662-F CGCGGAAAATGATGAGTAGTGA miR-185-3p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACGACCAG miR-185-3p-F CGAGGGGCTGGCTTTCCT miR-1307-5p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAGCCGG miR-1307-5p-F CGTCGACCGGACCTCGA miR-182-3p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACTAGTTG miR-182-3p-F GCGCGTGGTTCTAGACTTGC miR-1908-5p-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACGACCAA miR-1908-5p-F TAATTTCGGCGGGGACGGCGA all-reverse AGTGCAGGGTCCGAGGTATT U6-RT GTCGTATCCAGTGCAGGGTCCGAGGTATTCGCACTGGATACGACAAAATA U6-F GCGCGTCGTGAAGCGTTC U6-R GTGCAGGGTCCGAGGT 注:RNA为核糖核酸;PCR为聚合酶链式反应 表 2 X射线照射后非小细胞肺癌A549细胞的16个差异表达的miRNA
Table 2. Sixteen microRNAs differentially expressed in non-small cell lung cancer A549 cells after X-ray irradiation
miRNA名称 位置(染色体上) 20 Gy照射组(A) 50 mGy+20 Gy照射组(B) 200 mGy+20 Gy照射组(C) log2(B/A) log2(C/A) miR-92a-3p 13q31.3 7712.85 3157.42 3396.89 −1.29 −1.19 miR-193b-3p 16p13.12 1291.15 523.84 379.80 −1.30 −1.77 miR-484 16p13.11 572.06 210.49 256.39 −1.44 −1.16 miR-1307-5p 10q24.33 9.66 0 1.06 −3.60 −3.19 miR-182-3p 7q32.2 7.11 0 0 −3.83 −3.83 miR-127-3p 14q32.2 652.59 326.11 297.88 −1.00 −1.14 miR-1303 5q33.2 106.58 47.04 25.53 −1.18 −2.07 miR-138-5p 3p21.22/16p13.3 54.29 15.15 10.64 −1.84 −2.36 miR-185-3p 22q11.21 42.63 8.77 7.45 −2.28 −2.52 miR-654-5p 14q32.31 32.25 15.95 10.64 −1.02 −1.61 miR-3662 6q23.3 2.00 26.31 15.96 3.71 2.99 miR-378d 4p16.2/8q22.1 28.79 7.97 11.70 −1.85 −1.30 miR-1908-5p 11q12.2 10.57 2.39 0 −2.14 −4.41 miR-582-5p 5q12.1 24.23 9.57 8.51 −1.34 −1.52 miR-501-3p Xp11.23 98.38 39.07 23.40 −1.33 −2.08 miR-1260b 11q21 83.99 28.70 26.60 −1.55 −1.66 注:A、B、C均为miRNA相对表达水平。miRNA为微RNA 表 3 X射线照射后非小细胞肺癌A549细胞差异表达微RNA调控靶基因的GO富集分析(前15位)
Table 3. Gene ontology enrichment analysis of target genes regulated by differentially expressed microRNAs in non-small cell lung cancer A549 cells after X-ray irradiation (TOP 15)
GO类别 GO富集描述 基因数 P值 生物过程 细胞通讯调节(regulation of cell communication) 1166 <0.01 代谢过程的正调节(positive regulation of metabolic process) 1271 <0.01 信号的调节(regulation of signaling) 1147 <0.01 代谢过程(metabolic process) 3472 <0.01 单体过程(single-organism process) 4178 <0.01 细胞组分 细胞质(cytoplasm) 3474 <0.01 细胞内(intracellular) 4285 <0.01 细胞内组分(intracellular part) 4183 <0.01 细胞质组分(cytoplasmic part) 2954 <0.01 细胞器(organelle) 3860 <0.01 分子功能 蛋白质结合(protein binding) 3480 <0.01 结合(binding) 4305 <0.01 酶结合(enzyme binding) 672 <0.01 催化活性(catalytic activity) 1811 <0.01 序列特异性DNA结合(sequence-specific DNA binding) 369 <0.01 注:GO为基因本体 -
[1] Yu HS, Song AQ, Fei CH, et al. Effects of low dose radiation on tumor apoptosis, cell cycle and apoptosis-related protein Bcl-2 in tumor-bearing mice[J]. Chin-Ger J Clin Oncol, 2005, 4(2): 89−92. DOI: 10.1007/S10330-004-0276-2. [2] Wu CL, Tian B, Qu XH, et al. MicroRNAs play a role in chondrogenesis and osteoarthritis (Review)[J]. Int J Mol Med, 2014, 34(1): 13−23. DOI: 10.3892/ijmm.2014.1743. [3] Piotto C, Biscontin A, Millino C, et al. Functional validation of miRNAs targeting genes of DNA double-strand break repair to radiosensitize non-small lung cancer cells[J]. Biochim Biophys Acta (BBA)-Gene Regul Mech, 2018, 1861(12): 1102−1118. DOI: 10.1016/j.bbagrm.2018.10.010. [4] Wang LJ, Li NN, Xu SJ, et al. A new and important relationship between miRNA-147a and PDPK1 in radiotherapy[J]. J Cell Biochem, 2018, 119(4): 3519−3527. DOI: 10.1002/jcb.26523. [5] Chu YC, Hu XL, Wang GF, et al. Downregulation of miR-136 promotes the progression of osteosarcoma and is associated with the prognosis of patients with osteosarcoma[J]. Oncol Lett, 2019, 17(6): 5210−5218. DOI: 10.3892/ol.2019.10203. [6] Lan FM, Yue X, Ren G, et al. miR-15a/16 enhances radiation sensitivity of non-small cell lung cancer cells by targeting the TLR1/NF-κB signaling pathway[J]. Int J Radiat Oncol Biol Phys, 2015, 91(1): 73−81. DOI: 10.1016/j.ijrobp.2014.09.021. [7] Zhou L, Chen JH, Li ZZ, et al. Integrated profiling of microRNAs and mRNAs: microRNAs located on Xq27.3 associate with clear cell renal cell carcinoma[J/OL]. PLoS One, 2010, 5(12): e15224[2020-11-29]. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0015224. DOI: 10.1371/journal.pone.0015224. [8] Love MI, Huber W, Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2[J/OL]. Genome Biol, 2014, 15(12): 550[2020-11-29]. https://genomebiology.biomedcentral.com/articles/10.1186/s13059-014-0550-8. DOI: 10.1186/s13059-014-0550-8. [9] Zhang Z, Wu H, Zhou H, et al. Identification of potential key genes and high-frequency mutant genes in prostate cancer by using RNA-Seq data[J]. Oncol Lett, 2018, 15(4): 4550−4556. DOI: 10.3892/ol.2018.7846. [10] Wen M, Shen Y, Shi SH, et al. miREvo: an integrative microRNA evolutionary analysis platform for next-generation sequencing experiments[J/OL]. BMC Bioinformatics, 2012, 13(1): 140[2020-11-29]. https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-13-140. DOI: 10.1186/1471-2105-13-140. [11] Kanehisa M, Araki M, Goto S, et al. KEGG for linking genomes to life and the environment[J]. Nucleic Acids Res, 2008, 36(Datebase issue): D480−D484. DOI: 10.1093/nar/gkm882. [12] Terradas M, Martín M, Repullès J, et al. Distinct sets of lncRNAs are differentially modulated after exposure to high and low doses of X rays[J]. Radiat Res, 2016, 186(6): 549−558. DOI: 10.1667/rr14377.1. [13] Osipov A N, Buleeva G, Arkhangelskaya E, et al. In vivo γ-irradiation low dose threshold for suppression of DNA double strand breaks below the spontaneous level in mouse blood and spleen cells[J]. Mutat Res/Genet Toxicol Environ Mutagen, 2013, 756(1/2): 141−145. DOI: 10.1016/j.mrgentox.2013.04.016. [14] 刘淑春, 赵文举, 吕喆, 等. 低剂量辐射诱导EL-4淋巴瘤细胞凋亡及细胞周期进程适应性反应的剂量率效应[J]. 吉林大学学报(医学版), 2008, 34(1): 24−27. DOI: 10.3969/j.issn.1671-587X.2008.01.006.
Liu SC, Zhao WJ, Lyu Z, et al. Dose-rate effect of adaptive response of apoptosis and cell cycle progression induced by low-dose ionizing radiation in EL-4 lymphoma cells in vitro[J]. J Jilin Univ: Med Ed, 2008, 34(1): 24−27. DOI: 10.3969/j.issn.1671-587X.2008.01.006.[15] Chen N, Wu LJ, Yuan H, et al. ROS/autophagy/Nrf2 pathway mediated low-dose radiation induced radio-resistance in human lung adenocarcinoma A549 cell[J/OL]. Int J Biol Sci, 2015, 11(7): 833−844[2020-11-29]. https://www.ijbs.com/v11p0833.htm. DOI: 10.7150/ijbs.10564. [16] Terashima S, Hosokawa Y, Tsuruga E, et al. Impact of time interval and dose rate on cell survival following low-dose fractionated exposures[J]. J Radiat Res, 2017, 58(6): 782−790. DOI: 10.1093/jrr/rrx025. [17] Wakeford R, Tawn EJ. The meaning of low dose and low dose-rate[J]. J Radiol Prot, 2010, 30(1): 1−3. DOI: 10.1088/0952-4746/30/1/E02. [18] Ye J, Xiao X, Han Y, et al. MiR-3662 suppresses cell growth, invasion and glucose metabolism by targeting HK2 in hepatocellular carcinoma cells[J]. Neoplasma, 2020, 67(4): 773−781. DOI: 10.4149/neo_2020_190730n689. [19] Powrózek T, Krawczyk P, Kowalski DM, et al. Plasma circulating microRNA-944 and microRNA-3662 as potential histologic type-specific early lung cancer biomarkers[J]. Transl Res, 2015, 166(4): 315−323. DOI: 10.1016/j.trsl.2015.05.009. [20] Chang H, Liu YH, Wang LL, et al. MiR-182 promotes cell proliferation by suppressing FBXW7 and FBXW11 in non-small cell lung cancer[J/OL]. Am J Transl Res, 2018, 10(4): 1131−1142[2020-11-29]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5934572. [21] 陈光朋, 付万垒, 杨坤富, 等. miR-182调控RECK信号通路在非小细胞肺癌进展中的作用[J]. 解放军医学杂志, 2015, 40(4): 309−314. DOI: 10.11855/j.issn.0577-7402.2015.04.11.
Chen GP, Fu WL, Yang KF, et al. Effect of miR-182 in non small cell lung cancer via regulating the RECK signaling pathway[J]. Med J Chin PLA, 2015, 40(4): 309−314. DOI: 10.11855/j.issn.0577-7402.2015.04.11.[22] Masliah-Planchon J, Garinet S, Pasmant E. RAS-MAPK pathway epigenetic activation in cancer: miRNAs in action[J/OL]. Oncotarget, 2016, 7(25): 38892−38907[2020-11-29]. https://www.oncotarget.com/article/6476/text. DOI: 10.18632/oncotarget.6476. [23] Drosten M, Barbacid M. Targeting the MAPK pathway in KRAS-Driven tumors[J]. Cancer Cell, 2020, 37(4): 543−550. DOI: 10.1016/j.ccell.2020.03.013. [24] Huang T, Yin L, Wu J, et al. MicroRNA-19b-3p regulates nasopharyngeal carcinoma radiosensitivity by targeting TNFAIP3/NF-κB axis[J/OL]. J Exp Clin Cancer Res, 2016, 35(1): 188[2020-11-29]. https://jeccr.biomedcentral.com/articles/10.1186/s13046-016-0465-1. DOI: 10.1186/s13046-016-0465-1. [25] Dent P, Yacoub A, Fisher PB, et al. MAPK pathways in radiation responses[J]. Oncogene, 2003, 22(37): 5885−5896. DOI: 10.1038/sj.onc.1206701. [26] Mutlu M, Saatci Ö, Ansari SA, et al. miR-564 acts as a dual inhibitor of PI3K and MAPK signaling networks and inhibits proliferation and invasion in breast cancer[J/OL]. Sci Rep, 2016, 6: 32541[2020-11-29]. https://www.nature.com/articles/srep32541. DOI: 10.1038/srep32541. [27] Hagan M, Wang L, Hanley JR, et al. Ionizing radiation-induced mitogen-activated protein (MAP) kinase activation in DU145 prostate carcinoma cells: MAP kinase inhibition enhances radiation-induced cell killing and G2/M-phase arrest[J]. Radiat Res, 2000, 153(4): 371−383. DOI: 10.1667/0033-7587(2000)153[0371:irimap]2.0.co;2. [28] Tian XQ, Geng J, Zheng Q, et al. Single high dose irradiation induces cell cycle arrest and apoptosis in human hepatocellular carcinoma cells through the Ras/Raf/MEK/ERK pathways[J]. Int J Radiat Biol, 2020, 96(6): 740−747. DOI: 10.1080/09553002.2020.1694188. -





 下载:
下载: