-
长链非编码RNA(long non-coding RNA,lncRNA)是一类长度超过200个核苷酸的RNA分子,虽然大多数lncRNA的功能尚不清楚,但近年来已有越来越多的lncRNA的功能得到鉴定[1]。最近的研究结果表明,lncRNA具有稳定的二级结构,可作为多种肿瘤的外周生物标志物[2]。它们在很大程度上参与细胞在转录、转录后和表观遗传水平上对基因表达的调节[3],并能通过不同机制调节细胞功能[4],例如:调节染色质动态,基因表达,细胞的生长、分化和发育[5-6]。lncRNA的表达及突变能促进肿瘤的发生和转移,由于其在多种组织中的全基因组表达模式和组织表达特异性,lncRNA有望成为新的肿瘤生物标志物和治疗靶点[7]。同源异型盒基因转录的反义基因间RNA(homeobox gene anti-sense intergenic RNA,HOTAIR)是研究最深入的lncRNA之一,其来源于染色体12q13.13上同源异型盒基因 C11和同源异型盒基因 C12之间的同源异型盒基因 C反义链的转录,是第一个被发现具有反式调节功能的lncRNA[8-9]。一些研究结果表明,HOTAIR在不同类型的肿瘤中表达失调,例如:乳腺癌、非小细胞肺癌、肝细胞癌、胃肠道癌和结直肠癌等[4, 7]。并且,HOTAIR参与了多种与肿瘤发生相关的过程,例如:影响细胞的迁移、增殖、凋亡、侵袭、攻击和转移[10]。鉴于这些重要功能,HOTAIR被用作人类各种肿瘤的潜在生物标志物[11]。
反义寡核苷酸(antisense oligonucleotides,ASON)是人工合成的长度为16~22个碱基的单链DNA类似物,通过沃森-克里克碱基配对与靶基因结合,可以导致核酸内切酶介导的转录本被敲除,以致靶基因沉默[12-13]。ASON可以高效、特异、不可逆地与靶基因互补结合,但标记后的探针能否进入胶质瘤细胞与HOTAIR特异性结合并影响细胞功能尚不明确。因此,本研究通过制备放射性核素标记的靶向lncRNA HOTAIR的ASON探针99Tcm-HYNIC-ASON[HYNIC为联肼尼克酰胺(hydrazino nicotinamide)],在细胞水平上研究其对人脑胶质瘤U87细胞增殖和迁移能力的影响,为HOTAIR在胶质瘤中的应用提供良好的理论支持。
-
长度为19个碱基的ASON探针购自上海生工生物工程股份有限公司,并进行化学合成和修饰。人脑胶质瘤U87细胞购自中国科学院上海细胞库;99Tcm-高锝酸盐溶液购自北京原子高科技术股份有限公司;HYNIC购自美国TriLink公司;交联葡聚糖凝胶(Sephadex) G25购自美国GE公司;脂质体Lipofectamine 2000 购自美国Invitrogen公司;Tricine购自美国Sigma-Aldrich公司;DMEM培养基、胎牛血清购自西安励合生物有限公司;细胞计数试剂盒8(cell counting kit-8,CCK-8)购自美国APExBIO公司。超滤管购自德国Sartorius公司;MR-96A酶标仪购自美国 Thermo公司;GC-1200型 γ放射免疫计数器购自中国计量科学研究院;1645050 PowerPac Basic基础电泳仪购自美国Bio-Rad公司。
-
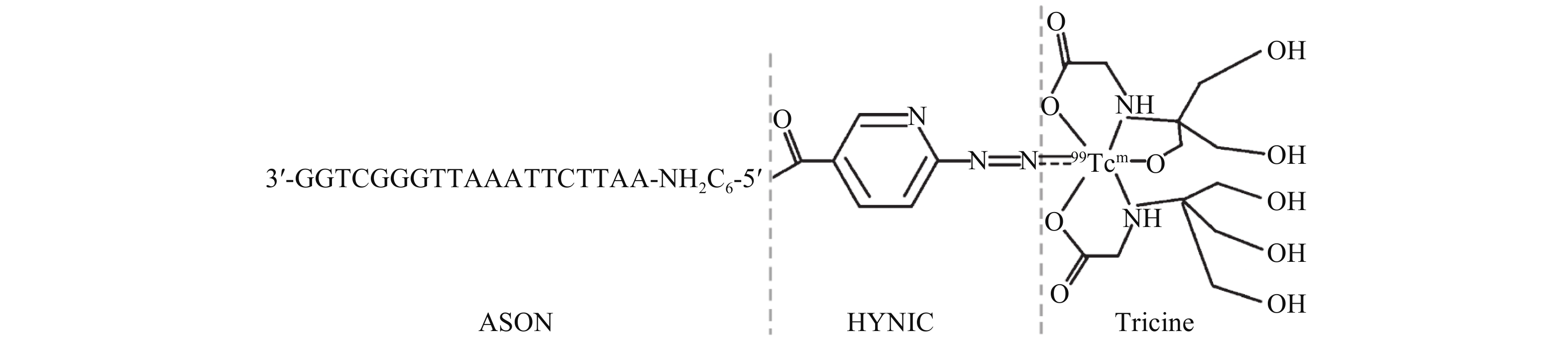
设计与HOTAIR片段完全互补的ASON序列:5′-AATTCTTAAATTGGGCTGG-3′,序列两端分别进行2′-O-甲基化修饰,两端各有2个碱基进行硫代修饰以提高稳定性。5′端连接NH2C6,即合成结构式为 3′-ASON-NH2C6-5′,具体见图1,HYNIC与Tricine共同偶联99Tcm。
-
取1 mg HYNIC溶于 100 μL N,N-二甲基甲酰胺(DMF)溶液,浓度10 mg/mL。将0.2 mg ASON溶于50 μL缓冲液(2 mol/L NaCl、0.5 mol/L NaHCO3、2 mmol/L 乙二胺四乙酸)中。将HYNIC和ASON以摩尔比为25∶1的比例混合,避光反应1~2 h。反应结束后加入60%甲醇至总体积为500 μL,置于超滤管中以15 000×g离心10 min(保证离心后体积<50 μL)。随后向离心后反应物中依次加入100 μL Tricine (100 mg/mL)、20 μL新鲜淋取的99Tcm-高锝酸盐溶液(111 MBq),最后加入4 μL新鲜配置的SnCl2·2H2O(1 mg/mL)溶液,混匀,避光反应60 min。反应完毕后用Sephadex G25分离纯化,每管收集5滴(约0.6 mL)洗脱液,分别测定每管的放射性活度和核酸浓度,并绘制曲线。取高峰管以备后续实验。
-
将99Tcm-HYNIC-ASON分别于室温(25℃)、37℃条件下在生理盐水和新鲜人血清中孵育(探针与血清或生理盐水的体积比为1∶1),于0、2、4、6、8、12 h时采用快速薄层层析(instant thin layer chromatography,ITLC)法测定放射化学纯度。
-
配置1%的琼脂糖凝胶,依次加入稀释的ASON样品、99Tcm-高锝酸盐溶液、纯化前的99Tcm-HYNIC-ASON、纯化后的99Tcm-HYNIC-ASON。采用电压120 V,电泳20 min,于紫外光下观察条带。
-
将人脑胶质瘤U87细胞培养在含15%胎牛血清、1%青霉素和链霉素混合液的高糖DMEM培养基中,放置于37℃、5% CO2的培养箱中培养24 h,待细胞密度达到90%进行传代。
配制转染混合液A:取10 μg纯化后的99Tcm-HYNIC-ASON加入500 μL无血清、无青霉素和链霉素混合液的DMEM培养基;配制转染混合液B:取25 μL Lipofectamine 2000加入475 μL DMEM培养基中,温和震荡后室温放置5 min。随后将混合液A、B混匀,室温放置20 min得到脂质体包裹的99Tcm-HYNIC-ASON(即Lipo-99Tcm-HYNIC-ASON)。将人脑胶质瘤U87细胞按每孔1.2×106个接种于六孔板中,培养24 h后每孔加入100 μL Lipo-99Tcm-HYNIC-ASON,于37℃培养箱中培养6 h,随后吸弃培养基,换为含15%胎牛血清的DMEM培养基,培养24~48 h完成转染。
-
将处于对数生长期的人脑胶质瘤U87细胞按每孔1×105个接种于12孔板中。在无青霉素和链霉素混合液、含10% 胎牛血清的DMEM培养基中培养过夜。细胞摄取实验分为2组:Lipo-99Tcm-HYNIC-ASON组(转染组)和99Tcm-HYNIC-ASON组(未转染组)。转染组每孔加入200 μL DMEM培养基、500 ng 99Tcm-HYNIC-ASON(37 kBq)和3 μL Lipofectamine 2000,混匀完成转染;未转染组每孔加入200 μL DMEM培养基和500 ng 99Tcm-HYNIC-ASON(37 kBq),混匀。将2组细胞放置于37℃培养箱中培养,分别于加样后1、2、3、4、5、6 h时收集一孔培养液于EP管中,100 μL PBS冲洗3遍,收集到同一EP管中,标记为Cout;用含乙二胺四乙酸的胰酶消化细胞,每孔用100 μL PBS冲洗3遍,收集到同一EP管中,标记为Cin。采用γ放射免疫计数器检测Cin及Cout的放射性计数,计算各个时间点细胞对探针的摄取率,细胞摄取率=Cin的放射性计数/(Cin的放射性计数+Cout的放射性计数)×100%。
-
收集对数生长期的人脑胶质瘤U87细胞接种于96孔板中,每组设5个复孔,待细胞密度达到60%,分为3组:Lipo-99Tcm-HYNIC-ASON组(转染组)、99Tcm-HYNIC-ASON组(未转染组)、99Tcm-Control组(对照组),并分别处理细胞(转染组6 h时换一次液)。根据CCK-8试剂盒说明书操作,将细胞培养1~5 d,每天于同一时间点加入CCK-8。为避免误差,每孔分别加入无血清、无青霉素和链霉素混合液的DMEM培养基稀释的CCK-8 110 μL(无血清、无青霉素和链霉素混合液的DMEM 培养基100 μL、CCK-8 10 μL),于37℃、5% CO2培养箱中孵育2.5 h后,用酶标仪检测450 nm处的吸光度(A450值)。
-
按“1.8”小节中分组将细胞按每孔1.2×106个接种于6孔板中并分别进行处理,待细胞密度达到90%时,吸弃孔内培养基,用200 μL枪头垂直在6孔板中央划线,PBS冲洗2遍洗去被划下的细胞,加入无血清培养基继续培养。记录培养细胞0、24 h时的划痕面积,计算细胞间隙融合率,细胞间隙融合率=(0 h划痕面积−24 h划痕面积)/0 h划痕面积×100%。
-
应用SPSS 22.0软件进行统计学分析。计量资料符合正态分布以
$\bar x $ ±s表示,2组间比较采用Student t检验(方差齐性),多组间比较采用单因素方差分析。P<0.05为差异有统计学意义。 -
由图2可见,纯化前(图2B)与纯化后(图2C)的99Tcm-HYNIC-ASON的放射性滞留在同一位置。ITLC法测得探针的标记率为(90.0±5.6)%。
-
分离纯化99Tcm-HYNIC-ASON共收集到洗脱液15管,纯化物的放射性活度及核酸浓度见图3。由图3B可见,未结合的ASON片段经1~5管过滤掉;5~10管为99Tcm-HYNIC-ASON,与图3A的放射性活度峰值相符。
-
由图4A可见,99Tcm-HYNIC-ASON探针12 h的放射化学纯度达80%以上,在生理盐水及新鲜人血清中的放射化学纯度无明显差别;由图4B可见,99Tcm-HYNIC-ASON探针在纯化前后都显示亮的条带,这说明探针具有完整性并且没有发生明显降解和脱靶。
-
由图5可见,在1 h后的任一时间点,转染组对Lipo-99Tcm-HYNIC-ASON的细胞摄取率均高于未转染组对99Tcm-HYNIC-ASON的细胞摄取率,且差异均有统计学意义(均P<0.01)。转染后5 h,Lipo-99Tcm-HYNIC-ASON在人脑胶质瘤U87细胞中的摄取率最大(0.70%),与未转染组(0.16%)相比,差异有统计学意义(t=17.81,P<0.01)。 而未转染组细胞最大摄取率出现在6 h时,仅为0.20%。
-
由图6可见,与未转染组和对照组相比,在转染后1、2、3、4、5 d,转染组吸光度均较低,这说明Lipofectamine 2000可以有效地使探针进入细胞并抑制细胞的增殖能力,在各个时间点,转染组与未转染组相比,吸光度的差异均有统计学意义(均P<0.05)。
-
由图7可见,转染Lipo-99Tcm-HYNIC-ASON 24 h后细胞迁移能力下降,这说明Lipofectamine 2000转染可以有效地使探针进入细胞并抑制细胞的迁移。3组细胞间隙融合率的差异有统计学意义(F=331.8,P<0.01),与未转染组相比,转染组细胞间隙融合率明显降低,且差异有统计学意义(60.0%对23.6%,t=51.54, P<0.01)。
-
鉴于HOTAIR在胶质瘤及其他恶性肿瘤中的特异性高表达,利用放射性核素标记与HOTAIR特异性结合的ASON探针对胶质瘤的早期发现具有重要意义。本研究成功地利用HYNIC螯合99Tcm标记靶向HOTAIR的ASON。本研究制备探针的标记率高且12 h内血清稳定性较好,经凝胶电泳鉴定没有发生明显的降解;细胞实验结果显示,经脂质体转染的探针可以更有效地进入细胞,并抑制人脑胶质瘤U87细胞的增殖和迁移。
在标记实验中,首先,由于HYNIC只有1个或2个99Tcm的结合位点,因此选用Tricine为协同配体,2个分子提供的位点可以与99Tcm形成稳定的复合物,获得较高的标记率。其次,为保护ASON免受 RNA酶的降解,在ASON探针两端各3个碱基分别进行甲基化及硫代修饰,以增加其体内外稳定性。再次,与其他文献[14-16]报道的标记方法相比,我们采用自配的缓冲液(2 mol/L NaCl、0.5 mol/L NaHCO3、2 mmol/L 乙二胺四乙酸)代替碳酸氢盐缓冲液,同时提高HYNIC与ASON的摩尔比为25∶1,并用超滤管过滤未结合的ASON片段。结果显示,99Tcm-HYNIC-ASON的标记率为(90.0±5.6)%,并且没有发生明显的降解。然而,ITLC法结果显示,99Tcm-高锝酸盐溶液中大部分99Tcm被氧化为胶体99Tcm,不能被SnCl2·2H2O还原,反应物经Sephadex G25分离纯化后,大部分未结合的ASON片段提前滤出,因此新鲜淋取的99Tcm对标记率有较大影响。
影响ASON探针结合的主要因素之一是放射性标记的ASON探针在细胞内的摄取率低[17]。脂质体可以通过静电相互作用将带负电荷的ASON包裹起来,这样亲脂性的ASON就可以很容易地穿过细胞膜。在细胞内部,脂质体破坏了内小体的捕获,使得ASON能够逃逸到细胞质中,与其目标mRNA结合[18-19]。此外,脂质体还能增强探针的核定位[20]。我们选择Lipofectamine 2000作为载体,在细胞摄取实验中,Lipo-99Tcm-HYNIC-ASON的摄取率由0.02%上升至0.70%,而未转染组6 h内最大摄取率仅为0.20%。另外,细胞划痕实验结果显示,转染组24 h细胞间隙融合率明显低于未转染组。CCK-8实验结果也显示,与未转染组相比,转染组细胞增殖能力明显下降。以上结果证明,脂质体能有效包裹探针穿过细胞膜进入细胞并抑制细胞的增殖及迁移能力。
本研究存在一些不足之处:(1)99Tcm与ASON片段的结合率还有待提高,目前可以采用的方法是选用新鲜淋取的99Tcm-高锝酸盐溶液和新鲜配置的SnCl2·2H2O溶液并尽快将二者混合;(2)探针的放射化学纯度相对偏低,可以进一步改进标记方法,例如通过改变99Tcm-高锝酸盐溶液体积、SnCl2·2H2O浓度以及反应时间等进行改善[21]。
综上所述,99Tcm标记的靶向HOTAIR的ASON探针99Tcm-HYNIC-ASON具有较高的标记率和体外稳定性,经脂质体转染后可以进入人脑胶质瘤U87细胞并发挥抑制作用,为U87荷瘤裸鼠的肿瘤反义显像的应用提供了良好的实验基础。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 任炯羽负责实验的实施、论文的撰写与修订;张熹元、冯薛烟负责实验的实施;张福禄负责方法的建立;赵倩负责方法的建立、论文的审阅。
99Tcm标记lncRNA HOTAIR反义寡核苷酸探针的制备及其对人脑胶质瘤U87细胞活性的影响
Preparation of 99Tcm labeled lncRNA HOTAIR antisense probe and its effect on the activity of human glioma U87 cells
-
摘要:
目的 制备新型的靶向长链非编码RNA(lncRNA)同源异型盒基因转录的反义基因间RNA(HOTAIR)的反义寡核苷酸(ASON)探针99Tcm-HYNIC-ASON(HYNIC为联肼尼克酰胺),探讨其对人脑胶质瘤U87细胞增殖和迁移能力的影响。 方法 设计并通过化学修饰合成HOTAIR的ASON,使用双功能螯合剂HYNIC偶联99Tcm并进行纯化。采用快速薄层层析(ITLC)法和琼脂糖凝胶电泳分别检测探针的标记率、放射化学纯度、体外稳定性及完整性。细胞摄取实验分为2组:Lipo-99Tcm-HYNIC-ASON组(转染组)和99Tcm-HYNIC-ASON组(未转染组),通过脂质体转染探针,测定人脑胶质瘤U87细胞对探针的摄取率;细胞计数试剂盒8(CCK-8)实验和细胞划痕实验分为3组:Lipo-99Tcm-HYNIC-ASON组(转染组)、99Tcm-HYNIC-ASON组(未转染组)、99Tcm-Control组(对照组),分别检测转染探针后细胞增殖和迁移能力的变化。2组间比较采用Student t检验,多组间比较采用单因素方差分析。 结果 99Tcm-HYNIC-ASON的标记率为(90.0±5.6)%。琼脂糖凝胶电泳结果显示,99Tcm与探针成功标记并且没有明显的降解,探针孵育12 h的放射化学纯度>80%。细胞摄取实验结果显示,转染后5 h,探针Lipo-99Tcm-HYNIC-ASON在人脑胶质瘤U87细胞中的摄取率最大(0.70%),与未转染组(0.16%)相比,差异有统计学意义(t=17.81,P<0.01)。CCK-8实验结果显示,转染探针Lipo-99Tcm-HYNIC-ASON能抑制人脑胶质瘤U87细胞的增殖能力,与未转染组相比,在各个时间点(1、2、3、4、5 d)的差异均有统计学意义(t=2.336~30.230, 均P<0.05)。细胞划痕实验结果显示,转染探针Lipo-99Tcm-HYNIC-ASON能抑制人脑胶质瘤U87细胞的迁移,3组细胞间隙融合率的差异有统计学意义(F=331.8,P<0.01),与未转染组相比,转染组细胞间隙融合率明显降低,且差异有统计学意义(60.0%对23.6%, t=51.54,P<0.01)。 结论 成功合成了靶向人脑胶质瘤lncRNA HOTAIR的探针99Tcm-HYNIC-ASON,该探针具有良好的体外稳定性和靶向结合能力,能够抑制人脑胶质瘤U87细胞的增殖和迁移。 Abstract:Objective To prepare a novel antisense oligonucleotides (ASON) probe, namely, 99Tcm-HYNIC-ASON (hydrazine nicotinamide (HYNIC)), targeting long non-coding RNA (lncRNA) homeobox gene anti-sense intergenic RNA (HOTAIR); and explore its effect on the proliferation and migration of human glioma U87 cells. Methods HOTAIR ASON was designed and synthesized by chemical modification, and the bifunctional chelating agent (HYNIC) was coupled with 99Tcm. Sephadex G25 was selected for separation and purification. The labeling rate, radiochemical purity, in vitro stability, and integrity of the probe were detected by instant thin-layer chromatography and agarose gel electrophoresis. Human glioma U87 cells were cultured for experimental use. The cell uptake assay was divided into two groups: Lipo-99Tcm-HYNIC-ASON (transfection group) and 99Tcm-HYNIC-ASON (non-transfection group). The probe was transfected by liposome to determine the probe uptake rate of human glioma U87 tumor cells. The cell counting kit-8 (CCK-8) assay and cell scratch assay were divided into three groups, namely, Lipo-99Tcm-HYNIC-ASON (transfection group), 99Tcm-HYNIC-ASON (non-transfection group), and 99Tcm-Control (control group), to detect the changes of cell proliferation and migration after transfection of probe. Student t-test was used for comparison between two groups, and one-way analysis of variance was used for multi-group comparison. Results The labeling rate of 99Tcm-HYNIC-ASON was (90.0±5.6)%. Gel electrophoresis results confirmed that 99Tcm and the probe were successfully labeled without evident degradation; the probe showed good stability and radiochemical purity >80% after being incubated for 12 h. The results of cell uptake assay showed that 5 h after liposome transfection, the maximum uptake rate of probe Lipo-99Tcm-HYNIC-ASON in human glioma U87 cells was 0.70%, which was significantly higher than that in the non-transfection group (0.16%; t=17.81, P<0.01). The results of CCK-8 assay showed that the transfection probe (Lipo-99Tcm-HYNIC-ASON) could inhibit the proliferation of human glioma U87 cells, and a significant difference was observed compared with the non-transfection group at 1, 2, 3, 4, 5 d (t=2.336–30.230, all P<0.05). The results of cell scratch assay showed that the transfection probe (Lipo-99Tcm-HYNIC-ASON) could inhibit the migration of human glioma U87 cells, and a significant difference was found in the intercellular fusion rate among the three groups (F=331.8, P<0.01). Compared with the non-transfection group (60.0%), the intercellular fusion rate in the transfection group was significantly lower (23.6%), and the difference was statistically significant (t=51.54, P<0.01). Conclusions The ASON probe targeting human glioma lncRNA HOTAIR has been successfully synthesized. The probe has good stability and targeted binding ability in vitro, which can inhibit the proliferation and migration of human glioma U87 cells. -
-
[1] Botti G, Scognamiglio G, Aquino G, et al. LncRNA HOTAIR in tumor microenvironment: what role?[J/OL]. Int J Mol Sci, 2019, 20(9): 2279[2020-11-10]. https://www.mdpi.com/1422-0067/20/9/2279. DOI: 10.3390/ijms20092279. [2] Tan SK, Pastori C, Penas C, et al. Serum long noncoding RNA HOTAIR as a novel diagnostic and prognostic biomarker in glioblastoma multiforme[J/OL]. Mol Cancer, 2018, 17(1): 74[2020-11-10]. https://molecular-cancer.biomedcentral.com/articles/10.1186/s12943-018-0822-0. DOI: 10.1186/s12943-018-0822-0. [3] Toy HI, Okmen D, Kontou PI, et al. HOTAIR as a prognostic predictor for diverse human cancers: a meta- and bioinformatics analysis[J/OL]. Cancers (Basel), 2019, 11(6): 778[2020-11-10]. https://www.mdpi.com/2072-6694/11/6/778. DOI: 10.3390/cancers11060778. [4] Hajjari M, Rahnama S. Association between snps of long non-coding rna HOTAIR and risk of different cancers[J/OL]. Front Genet, 2019, 10: 113[2020-11-10]. https://www.frontiersin.org/articles/10.3389/fgene.2019.00113/full. DOI: 10.3389/fgene.2019.00113. [5] Bhan A, Mandal SS. LncRNA HOTAIR: a master regulator of chromatin dynamics and cancer[J]. Biochim Biophys Acta, 2015, 1856(1): 151−164. DOI: 10.1016/j.bbcan.2015.07.001. [6] 张书琴, 崔明, 肖惠文, 等. 姜黄素对非小细胞肺癌细胞A549和H460放射敏感性的影响[J]. 国际放射医学核医学杂志, 2020, 44(3): 164−173. DOI: 10.3760/cma.j.cn121381−201912006−00005.
Zhang SQ, Cui M, Xiao HW, et al. Effects of curcumin on the radiosensitivity of non-small cell lung cancer A549 and H460 cells[J]. Int J Radiat Med Nucl Med, 2020, 44(3): 164−173. DOI: 10.3760/cma.j.cn121381−201912006−00005.[7] Bhan A, Soleimani M, Mandal SS. Long noncoding RNA and cancer: a new paradigm[J]. Cancer Res, 2017, 77(15): 3965−3981. DOI: 10.1158/0008-5472.CAN-16-2634. [8] Zhan S, Wang K, Xiang Q, et al. lncRNA HOTAIR upregulates autophagy to promote apoptosis and senescence of nucleus pulposus cells[J]. J Cell Physiol, 2020, 235(3): 2195−2208. DOI: 10.1002/jcp.29129. [9] Sun G, Wang YY, Zhang JX, et al. MiR-15b/HOTAIR/p53 form a regulatory loop that affects the growth of glioma cells[J]. J Cell Biochem, 2018, 119(6): 4540−4547. DOI: 10.1002/jcb.26591. [10] Yuan C, Ning Y, Pan Y. Emerging roles of HOTAIR in human cancer[J]. J Cell Biochem, 2020, 121(5/6): 3235−3247. DOI: 10.1002/jcb.29591. [11] Hajjari M, Salavaty A. HOTAIR: an oncogenic long non-coding RNA in different cancers[J]. Cancer Biol Med, 2015, 12(1): 1−9. DOI: 10.7497/j.issn.2095-3941.2015.0006. [12] Li MW, Ding X, Zhang YA, et al. Antisense oligonucleotides targeting lncRNA AC104041.1 induces antitumor activity through Wnt2B/β-catenin pathway in head and neck squamous cell carcinomas[J/OL]. Cell Death Dis, 2020, 11(8): 672[2020-11-10]. https://www.nature.com/articles/s41419-020-02820-3. DOI: 10.1038/s41419-020-02820-3. [13] Bushart D, Zalon AJ, Zhang H, et al. Antisense oligonucleotide therapy targeted against ATXN3 improves potassium channel-mediated purkinje neuron dysfunction in spinocerebellar ataxia type 3[J]. Cerebellum, 2021, 20(1): 41−53. DOI: 10.1007/s12311-020-01179-7. [14] Fu P, Shen BZ, Zhao CJ, et al. Molecular imaging of MDM2 messenger RNA with 99mTc-labeled antisense oligonucleotides in experimental human breast cancer xenografts[J]. J Nucl Med, 2010, 51(11): 1805−1812. DOI: 10.2967/jnumed.110.077982. [15] Fu P, Tian L, Cao XL, et al. Imaging CXCR4 expression with 99mTc-radiolabeled small-interference rna in experimental human breast cancer xenografts[J]. Mol Imaging Biol, 2016, 18(3): 353−359. DOI: 10.1007/s11307-015-0899-4. [16] Liu N, Ding HL, Vanderheyden JL, et al. Radiolabeling small RNA with technetium-99m for visualizing cellular delivery and mouse biodistribution[J]. Nucl Med Biol, 2007, 34(4): 399−404. DOI: 10.1016/j.nucmedbio.2007.02.006. [17] Kang L, Wang RF, Yan P, et al. Noninvasive visualization of RNA delivery with 99mTc-radiolabeled small-interference RNA in tumor xenografts[J]. J Nucl Med, 2010, 51(6): 978−986. DOI: 10.2967/jnumed.109.069906. [18] Zhou Q, Fu Z. In vitro and in vivo study of a novel liposome-mediated dual drug delivery for synergistic lung cancer therapy via oral administration[J]. Onco Targets Ther, 2020, 13: 12695−12703. DOI: 10.2147/OTT.S276837. [19] Almeida B, Nag OK, Rogers KE, et al. Recent progress in bioconjugation strategies for liposome-mediated drug delivery[J/OL]. Molecules. 2020, 25(23): 5672 [2020-11-10]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7730700. DOI: 10.3390/molecules25235672. [20] Zhang M, Wang Q, Wan KW, et al. Liposome mediated-CYP1A1 gene silencing nanomedicine prepared using lipid film-coated proliposomes as a potential treatment strategy of lung cancer[J]. Int J Pharm, 2019, 566: 185−193. DOI: 10.1016/j.ijpharm.2019.04.078. [21] Kang L, Xu XJ, Ma C, et al. Optimized preparation of a 99mTc-radiolabeled probe for tracing microRNA[J]. Cell Biochem Biophys, 2015, 71(2): 905−912. DOI: 10.1007/s12013-014-0281-1. -






 下载:
下载: