-
卵巢癌是女性生殖器官常见的恶性肿瘤之一,其发病率居妇科恶性肿瘤的第3位,病死率更是高居妇科肿瘤首位[1-3]。尽管可采用手术及术后化疗的方式来治疗早期卵巢癌患者,但卵巢癌术后的复发率仍高达50%~75%,5年生存率仅为30%[4]。复发性卵巢癌是指经过满意的肿瘤细胞减灭术和正规足量的化疗达到临床完全缓解、停药6个月后临床上再次出现肿瘤复发的征象。复发性卵巢癌患者的病情多已进展至晚期,无法治愈,并会对患者的生活质量及生命造成严重影响[5]。因此,尽早发现、诊断复发性卵巢癌并及时采取干预措施,对改善患者预后、延长生存时间具有重要意义。复发性卵巢癌的预后影响因素很多,包括病理类型、术前临床分期、术后再分期、血清肿瘤学标志物水平、术后残留癌灶大小和术后化疗的规范性等。
目前临床判断复发性卵巢癌的方法包括肿瘤标志物[糖类抗原125(cdarbohydrate antigen 125,CA125)和人附睾蛋白4(human epididymis protein 4,HE4)等]水平的检测、体格检查以及影像学检查,其中血清CA125在多种恶性肿瘤中高表达,缺乏特异性,采用血清CA125诊断复发性卵巢癌,结果存在约20%的假阴性[6]。CA125与HE4的联合检测大大提高了复发性卵巢癌诊断的特异度和灵敏度[7],但不能提供病变解剖形态学信息。影像学检查是诊断复发性卵巢癌不可或缺的手段,目前最常用的是CT检查。然而,CT检查在判断复发性卵巢癌腹膜增厚的良恶性方面有一定的限制。PET/CT将PET与CT完美融为一体,PET可提供病灶详尽的功能与代谢等分子信息,CT可提供病灶的精确解剖定位。与常规CT相比,PET/CT明显提高了病灶的检出率和诊断的准确率,因此可更加灵敏地检出复发性卵巢癌的转移灶。此外,葡萄糖的类似物18F-FDG作为PET/CT临床最常用的示踪剂,其在肿瘤病灶或因炎症、感染所致的白细胞增多的病灶中有不同程度的浓聚,可起到鉴别诊断的作用[8]。
本研究探讨18F-FDG PET/CT显像联合CA125、HE4水平检测诊断复发性卵巢癌的效能及对腹膜转移的预后评估价值,现报道如下。
-
回顾性分析2016年1月至2021年6月在郑州大学第一附属医院核医学科接受18F-FDG PET/CT显像的89例卵巢癌术后患者的临床和影像学资料。本研究符合《赫尔辛基宣言》的原则。
纳入标准:(1)接受腹腔肿瘤减灭术并经组织病理学检查确诊为卵巢癌;(2)术后行周期化疗后达到完全缓解;(3)达到完全缓解后,在定期随访中临床怀疑复发和(或)转移;(4)临床、病理、影像学资料完整;(5)行18F-FDG PET/CT显像检查,同期(不超过7 d)肿瘤标志物(包括CA125、HE4)水平检测结果完整。
排除标准:(1)肿瘤细胞减灭术残留肿瘤病灶>1 cm;(2)术后行周期化疗后未达完全缓解;(3)临床、病理、影像学资料不全;(4)18F-FDG PET/CT显像与血清肿瘤标志物水平检测的间隔时间超过7 d;(5)失访。
根据纳入和排除标准,本研究共选取59例复发性卵巢癌患者[复发组,中位年龄51(26~82)岁]、30例卵巢癌术后经组织病理学检查确诊为非复发性卵巢癌患者[未复发组,中位年龄55(34~79)岁]。
-
使用德国Siemens公司的Biograph TruePoint 64(52环)PET/CT扫描仪,18F-FDG由日本住友公司HM-20回旋加速器及CFN-100合成模块自动化合成(放射化学纯度≥95%)。所有患者检查前空腹6 h以上,检查前控制血糖水平低于8.0 mmol/L,按体重静脉注射18F-FDG 3.70~5.55 MBq/kg,注射后60 min开始行18F-FDG PET/CT显像,采集时间为20 min。CT扫描参数:管电压120 kV,管电流与曝光时间的乘积为120~250 mAs,层厚3 mm。PET采用三维扫描模式,头部扫描3 min/床位,体部扫描1.5 min/床位,扫描范围从颅顶至股骨上段。CT衰减校正后进行迭代法重建。重建图像在机器自带的Syngo工作站上与CT图像进行融合,经计算机重建全身横断面、冠状面及矢状面断层图像。
-
由2名具有5年以上PET/CT诊断经验的主治及以上职称的核医学医师独立阅片,阅片内容包括复发和(或)转移灶所在位置、形态、大小、与周围组织之间的关系等,意见不统一时协商达成一致。勾画PET或CT发现的可疑复发和(或)转移灶(目测病灶18F-FDG摄取程度高于周围组织者为可疑病灶),并测量其SUVmax。
其中腹膜增厚的类型分为:(1)系膜分为“污迹样”改变和结节状增厚;(2)腹膜分为肿块增厚和结节状增厚;(3)网膜分为“污迹样”改变、结节状增厚和饼状增厚。腹膜病变的位置分为腹膜种植转移易感区(包括盆腔及右侧膈下区域)和腹膜种植转移低度易感区(包括右侧结肠旁沟、左侧膈下和左侧结肠旁沟)[9]。淋巴结短径>10 mm定义为淋巴结肿大。
分别计算18F-FDG PET/CT和增强CT诊断复发性卵巢癌及术后残端复发、腹膜转移、淋巴结转移、腹腔外其他转移灶的灵敏度、特异度、准确率、阳性预测值和阴性预测值。绘制18F-FDG PET/CT、CA125、HE4单独及联合检测复发性卵巢癌的ROC曲线,并分析其独立或联合检测诊断复发性卵巢癌的灵敏度、特异度、准确率、阳性预测值和阴性预测值。
-
所有患者经治疗达到临床完全缓解后,根据相关指南[10-11]规定进行定期随访,随访时间至少1年。具体如下:术后1年内每月1次,术后第2年起每3月1次,随访内容包括临床症状(如盆腔疼痛、体重减轻)和超声、CT或MRI、PET/CT检查结果。复发性卵巢癌确诊的方法包括腹水细胞学检查、腹膜活检组织病理学检查、腹腔镜探查术、影像学检查等。随访过程中若血清肿瘤标志物CA125和(或)HE4水平呈连续上升趋势,当CA125>35 U/mL和(或)HE4>140 pmol/L时均视为阳性;随访过程中影像学检查发现病灶体积逐渐增大(1~3个月体积增大1倍),或腹盆腔腹膜较之前显著增厚,或胸腹盆腔积液量较之前显著增多,或肠梗阻表现较之前明显进展则认为复发和(或)转移;若不满足上述情况则确认为未复发和(或)转移。
-
应用SPSS 23.0软件对数据进行统计学分析。18F-FDG PET/CT、增强CT及CA125、HE4的诊断效能之间的比较均采用χ2检验。采用Cox模型进行多因素预后分析,评估多种因素(包括国际妇产科联盟临床分期、血清肿瘤标志物水平、淋巴结肿大、腹膜转移、腹外其他转移灶)对复发性卵巢癌患者预后的影响并计算95%CI。采用Kaplan-Meier生存曲线分析单个预后因素对复发性卵巢癌患者总生存期的影响。P<0.05为差异有统计学意义。
-
89例患者的一般信息、病理类型、临床分期、血清CA125水平、血清HE4水平、总生存期等临床资料的比较详见表1。
临床资料 复发组(n=59) 非复发组(n=30) 检验值 P值 年龄[M(P25~P75),岁] 51(26~82) 55(34~79) t=5.519 0.133 病理类型[例(%)] χ2=0.001 0.976 浆液性癌 51(86.44) 26(86.67) 透明细胞癌 8(13.56) 4(13.33) 术前FIGO临床分期[例(%)] χ2=10.936 0.004 Ⅰ期 0 0 Ⅱ期 20(33.90) 21(70.00) Ⅲ期 36(61.02) 9(30.00) Ⅳ期 3(5.08) 0 术后残端复发[例(%)] 26(44.07) − − − 腹膜转移[例(%)] 51(86.44) − − − 淋巴结肿大[例(%)] 36(61.01) − − − 腹腔外其他转移[例(%)] 24(40.68) − − − CA125水平中位数[M(P25~P75),U/mL] 616.70(12.0~3852.0) 28.77(4.0~91.8) t=3.657 <0.001 HE4水平中位数[M(P25~P75),pmol/mL] 333.10(49.8~1674.0) 82.73(17.5~218.1) t=4.630 <0.001 总生存期中位数[M(P25~P75),个月] 18(2~45) 31(17~57) t=4.482 <0.001 注:FIGO为国际妇产科联盟;CA125为糖类抗原125;HE4为人附睾蛋白4。−表示无此项数据 表 1 89例卵巢癌术后患者的临床资料
Table 1. Clinical data of 89 postoperative ovarian cancer patients
-
由表2可知,在89例卵巢癌(59例复发性卵巢癌、30例非复发性卵巢癌)术后患者中,18F-FDG PET/CT诊断出复发性卵巢癌55例、非复发性卵巢癌28例。增强CT诊断出复发性卵巢癌47例、非复发性卵巢癌22例。
诊断结果 随访结果 合计 阳性 阴性 18F-FDG PET/CT阳性 55 2 57 18F-FDG PET/CT阴性 4 28 32 增强CT阳性 47 8 55 增强CT阴性 12 22 34 注:FDG为氟代脱氧葡萄糖;PET/CT为正电子发射断层显像计算机体层摄影术;CT为计算机体层摄影术 表 2 89例卵巢癌术后患者的18F-FDG PET/CT和增强CT 显像的诊断结果比较(例)
Table 2. Comparison of 18F-FDG PET/CT and contrast enhanced CT in the diagnosis of 89 postoperative ovarian cancer patients (case)
由表3可知,18F-FDG PET/CT显像评估复发性卵巢癌的灵敏度、特异度、准确率、阳性预测值、阴性预测值均高于增强CT,差异均有统计学意义(均P<0.05)。
转移/复发部位 诊断方式 灵敏度 特异度 准确率 阳性预测值 阴性预测值 复发性卵巢癌(n=59) 18F-FDG PET/CT 93.22a(55/59) 93.33a(28/30) 93.26a(83/89) 96.49a(55/57) 87.50a(28/32) 增强CT 79.66(47/59) 73.33(22/30) 77.52(69/89) 85.45(47/55) 64.71(22/34) 术后残端复发(n=26) 18F-FDG PET/CT 92.31a(24/26) 81.25a(13/16) 88.10a(37/42) 88.89a(24/27) 86.67a(13/15) 增强CT 66.67(18/27) 43.75(7/16) 59.52(25/42) 66.67(18/27) 46.67(7/15) 腹膜转移(n=51) 18F-FDG PET/CT 88.00a(44/50) 80.77a(21/26) 85.53a(65/76) 89.80(44/49) 77.78a(21/27) 增强CT 71.43(40/56) 50.00(10/20) 65.79(50/76) 80.00(40/50) 50.00(10/20) 淋巴结转移(n=36) 18F-FDG PET/CT 91.67(33/36) 85.71a(12/14) 90.00a(45/50) 94.29(33/35) 80.00a(12/15) 增强CT 80.56(29/36) 50.00(7/14) 72.00(36/50) 80.56(29/36) 50.00(7/14) 腹腔外其他转移(n=24) 18F-FDG PET/CT 91.67(22/24) 75.00(6/8) 87.50(28/32) 91.67(22/24) 75.00(6/8) 增强CT − − − − − 注:a表示与增强CT相比,差异均有统计学意义(χ2=1.847~8.868,均P<0.05)。FDG为氟代脱氧葡萄糖;PET/CT为正电子发射断层显像计算机体层摄影术;CT为计算机体层摄影术。−表示无此项数据 表 3 59例复发性卵巢癌患者不同复发或转移部位的18F-FDG PET/CT和增强CT诊断效能的比较(%)
Table 3. Comparison of the diagnostic efficacy of 18F-FDG PET/CT and contrast-enhanced CT at different recurrence or metastasis sites in 59 patients with recurrent ovarian cancer (%)
在59例复发性卵巢癌患者中,随访确诊术后残端复发患者26例、腹膜转移51例、淋巴结转移36例、腹腔外其他转移24例。其中,18F-FDG PET/CT诊断出术后残端复发患者24例、腹膜转移44例、淋巴结转移33例、腹腔外其他转移22例。增强CT诊断出术后残端复发患者18例、腹膜转移40例、淋巴结转移29例。因增强CT检查结果中关于腹腔外其他转移的资料不完整,无法评估腹腔外转移,故未纳入结果分析。
由表3可知,18F-FDG PET/CT诊断复发性卵巢癌术后残端复发患者的灵敏度、特异度、准确率、阳性预测值、阴性预测值均高于增强CT,且差异均有统计学意义(χ2=4.457、4.800、8.868、3.857、5.400,均P<0.05)。18F-FDG PET/CT诊断复发性卵巢癌腹膜转移的灵敏度、特异度、准确率、阳性预测值、阴性预测值均高于增强CT,且除阳性预测值外,其他各项之间的差异均有统计学意义(χ2=4.410、4.870、8.038、8.433,均P<0.05)。18F-FDG PET/CT诊断复发性卵巢癌淋巴结转移的灵敏度、特异度、准确率、阳性预测值、阴性预测值均高于增强CT,且除灵敏度、阳性预测值外,其他各项之间的差异均有统计学意义(χ2=4.094、5.263、2.885,均P<0.05)。
-
肿瘤标志物CA125、HE4水平检测联合18F-FDG PET/CT显像诊断复发性卵巢癌的灵敏度、特异度、准确率、阳性预测值和阴性预测值分别为98.31%、96.67%、97.75%、98.31%、96.67%,高于CA125、HE4、18F-FDG PET/CT 3种方法单独及CA125、HE4两两联合检测对于不同复发或转移部位CA125、HE4、18F-FDG PET/CT 3种方法联合检查的诊断灵敏度、特异度、准确率、阳性预测值、阴性预测值均高于CA125、HE4单独及两两联合应用,差异均有统计学意义(χ2=5.192~27.101,均P<0.05);3种方法联合检查的诊断灵敏度、特异度、准确率、阳性预测值和阴性预测值均高于18F-FDG PET/CT单独应用,但差异无统计学意义(χ2=0.351~2.094,均P>0.05)(表4)。
不同的诊断方法 灵敏度 特异度 准确率 阳性预测值 阴性预测值 CA125 术后残端复发 65.38a(17/26) 43.75a(7/16) 57.14a(24/42) 65.38a(17/26) 43.75a(7/16) 腹膜转移 72.73a(40/55) 57.14a(12/21) 68.42a(52/76) 81.63a(40/49) 44.44a(12/27) 淋巴结转移 72.22a(26/36) 42.86a(6/14) 64.00a(32/50) 76.47a(26/34) 37.50a(6/16) 腹腔外其他转移 58.33a(14/24) 37.50a(3/8) 53.13a(17/32) 73.68a(14/19) 23.08a(3/13) 复发性卵巢癌 69.49a(41/59) 76.67a(23/30) 71.91a(64/89) 85.42a(41/48) 56.10a(23/41) HE4 术后残端复发 69.23a(18/26) 50.00a(8/16) 61.90a(26/42) 69.23a(18/26) 50.00a(8/16) 腹膜转移 76.74a(39/51) 56.00a(14/25) 69.74a(53/76) 78.00a(39/50) 53.85a(14/26) 淋巴结转移 66.67a(24/36) 42.86a(6/14) 64.00a(32/50) 76.47a(26/34) 64.00a(32/50) 腹腔外其他转移 62.50a(15/24) 50.00(4/8) 59.38a(19/32) 78.95(15/19) 30.78a(4/13) 复发性卵巢癌 67.80a(40/59) 70.00a(21/30) 68.54a(61/89) 81.63a(40/49) 52.50a(21/40) 18F-FDG PET/CT 术后残端复发 92.31(24/26) 81.25(13/16) 88.10(37/42) 88.89(24/27) 86.67(13/15) 腹膜转移 88.00a(44/50) 80.77(21/26) 85.53(65/76) 89.80(44/49) 77.78(21/27) 淋巴结转移 91.67(33/36) 85.71(12/14) 90.00(45/50) 94.29(33/35) 80.00(12/15) 腹腔外其他转移 91.76(22/24) 75.00a(6/8) 87.50(28/32) 91.67(22/24) 75.00a(6/8) 复发性卵巢癌 93.22(55/59) 93.33(28/30) 93.26(83/89) 96.49(55/57) 87.50(28/32) CA125+HE4* 术后残端复发 73.08a(19/26) 56.25a(9/16) 66.67a(28/42) 73.08a(19/26) 56.25a(9/16) 腹膜转移 82.35a(42/51) 64.00a(16/25) 76.32a(58/76) 82.35a(42/51) 64.00a(16/25) 淋巴结转移 77.78a(28/36) 71.43(10/14) 76.00a(38/50) 87.50a(28/32) 55.56a(10/18) 腹腔外其他转移 70.83a(17/24) 50.00(4/8) 65.63a(21/32) 80.95a(17/21) 36.37a(4/11) 复发性卵巢癌 81.36a(48/59) 76.67a(23/30) 79.78a(71/89) 87.27a(48/55) 67.65a(23/34) CA125+HE4+18F-FDG PET/CT 术后残端复发 100.00(26/26) 93.75(15/16) 97.62(41/42) 96.30(26/27) 100.00(15/15) 腹膜转移 98.04(50/51) 96.00(24/25) 97.37(74/76) 98.04(50/51) 96.00(24/25) 淋巴结转移 97.22(35/36) 92.86(13/14) 96.00(48/50) 97.22(35/36) 92.86(13/14) 腹腔外其他转移 100.00(24/24) 87.50(7/8) 96.88(31/32) 96.00(24/25) 100.00(7/7) 复发性卵巢癌 98.31(58/59) 96.67(29/30) 97.75(87/89) 98.31(58/59) 96.67(29/30) 注:*表示当CA125>35 U/mL或HE4>140 pmol/L时视为阳性;a表示与CA125+HE4+18F-FDG PET/CT联合检查比较,差异均有统计学意义(χ2=5.192~27.101,均P<0.05)。CA125为糖类抗原125;HE4为人附睾蛋白4;PET/CT为正电子发射断层显像计算机体层摄影术 表 4 59例复发性卵巢癌患者的肿瘤标志物、18F-FDG PET/CT及其不同联合方式的诊断效能比较(%)
Table 4. Comparison of the diagnostic efficacy of tumor markers, 18F-FDG PET/CT and different combinations in 59 patients with recurrent ovarian cancer (%)
-
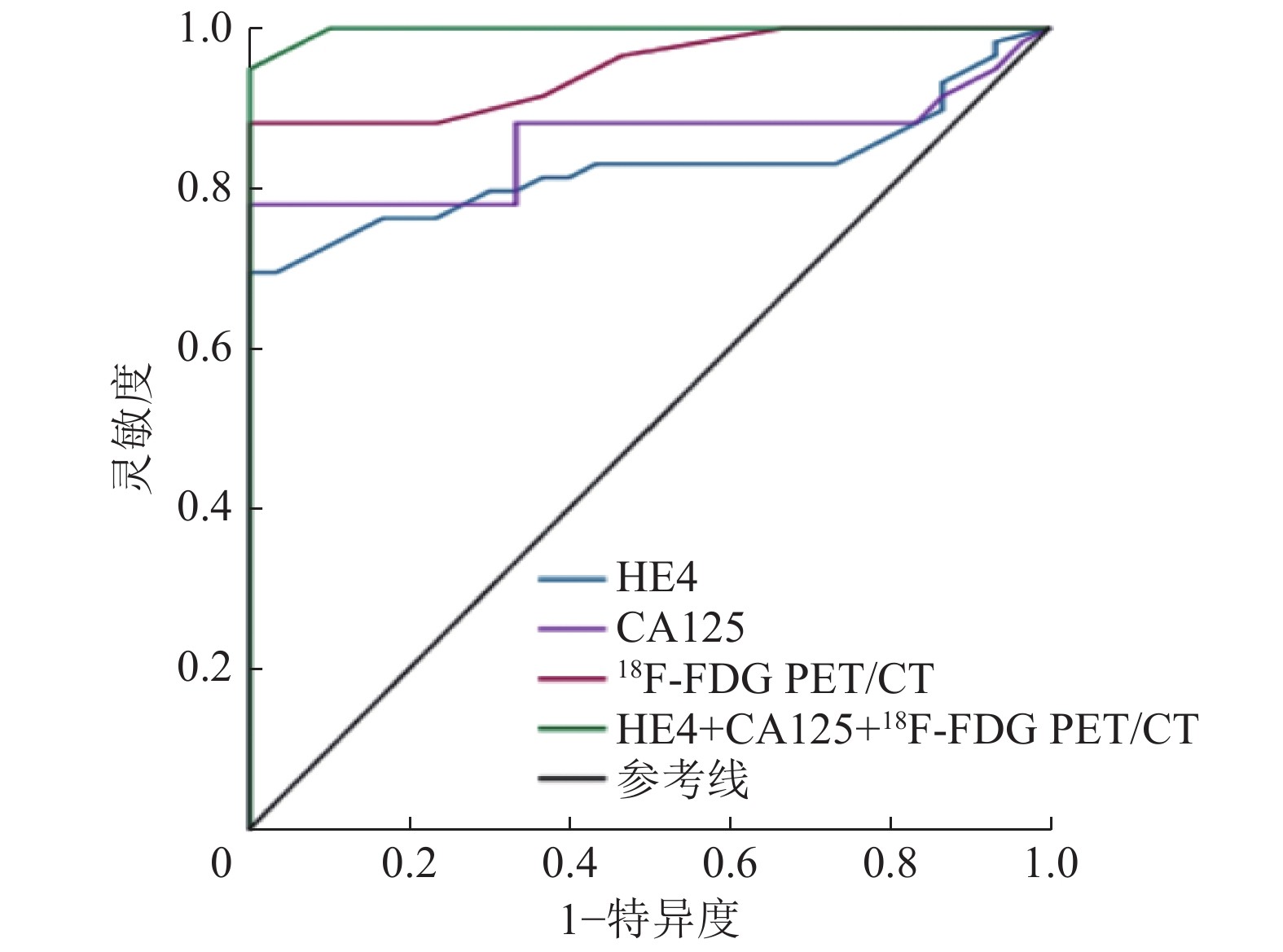
18F-FDG PET/CT代谢参数SUVmax及其联合血清CA125、HE4诊断复发性卵巢癌的ROC曲线分析结果显示,SUVmax的诊断临界值为5.60,AUC为0.949,95%CI:0.909~0.990;血清CA125水平的诊断临界值为91.80 U/mL,AUC为0.858,95%CI:0.728~0.938;HE4水平的诊断临界值为196.89 pmol/L,AUC为0.825,95%CI:0.737~0.912;HE4+CA125+18F-FDG PET/CT的曲线下面积为0.997,95%CI:0.993~1.002(图1)。
-
由表5可知,Cox模型评估国际妇产科联盟临床分期、血清CA125水平、血清HE4水平、淋巴结肿大、腹膜转移、腹腔外其他转移对复发性卵巢癌患者生存率影响的结果显示,腹膜转移是影响复发性卵巢癌患者的独立影响因素(P<0.05)。
影响因素 风险比 95%置信区间 P值 国际妇产科联盟临床分期 7.891 0.632~98.486 0.109 CA125水平 1.020 0.995~1.045 0.119 HE4水平 1.002 0.981~1.024 0.852 腹膜转移 55.688 3.784~819.477 0.003 淋巴结肿大 0.663 0.079~5.588 0.706 腹腔外其他转移 0.733 0.107~5.025 0.752 注:CA125为糖类抗原125;HE4为人附睾蛋白4 表 5 59例复发性卵巢癌患者预后影响因素加权Cox回归分析结果
Table 5. Weighted Cox regression analysis of factors affecting the prognosis of 59 patients with recurrent ovarian cancer
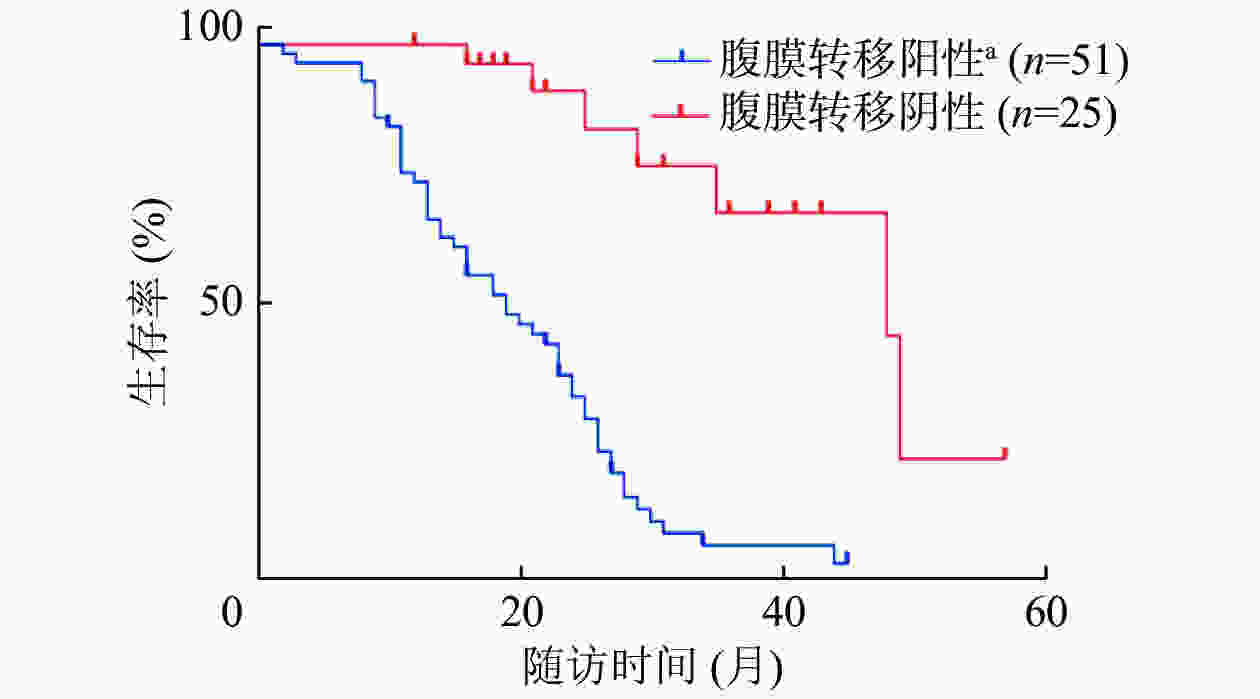
51例腹膜转移阳性与25例腹膜转移阴性的复发性卵巢癌患者的Kaplan-Meier生存曲线结果显示,腹膜转移阴性患者的总生存期明显高于腹膜转移阳性患者,且差异有统计学意义(χ2=30.320,P<0.001)(图2)。
-
在51例复发性卵巢癌腹膜转移患者中,35例复发性卵巢癌腹膜转移病灶位于腹膜种植转移易感区,16例位于腹膜种植转移低度易感区。
51例复发性卵巢癌腹膜转移患者均存在腹膜增厚,其中规则增厚18例,不规则增厚33例。腹膜不规则增厚的患者中,28例表现为结节状增厚,5例表现为肿块状增厚。腹膜不规则增厚患者中11例表现为系膜及网膜“污迹样”改变,5例表现为网膜“污迹样”改变及系膜结节状增厚。
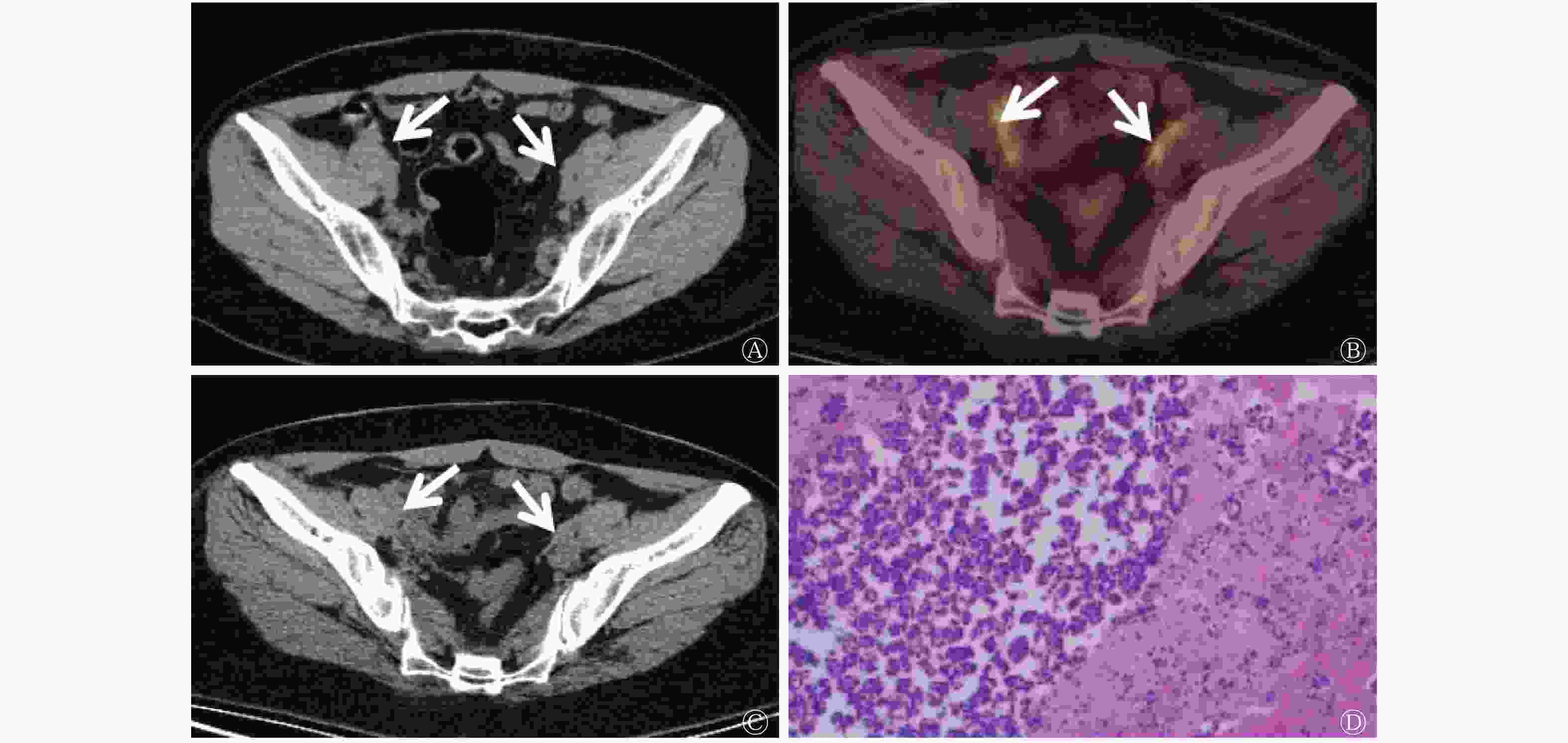
CT未显示出有明显病灶而18F-FDG PET/CT显像诊断为复发性卵巢癌、后经随访结束证实的典型病例图像见图3。
-
卵巢恶性肿瘤是女性生殖器官常见的恶性肿瘤之一,发病率仅次于子宫颈癌和子宫体癌。目前,手术仍然是治疗卵巢癌的主要方式,在经过规范的治疗后,超过80%的患者能够得到完全缓解,但在以后的2~5年里大部分患者面临复发的风险[12],并有70%会发生腹膜转移[6]。血清CA125和HE4水平是被广泛用于卵巢癌诊断和疗效监测的肿瘤标志物。CA125水平的升高最常见于上皮性卵巢肿瘤患者的血清中,在正常卵巢组织中含量较低,其诊断的灵敏度较高。有研究结果表明,血清CA125可在大多数复发性卵巢癌患者的血清中高表达,尤其是在部分常规影像学检查结果为阴性的患者中,这是卵巢癌复发的明显标志[8],且CA125水平持续升高是卵巢癌预后不良的危险因素之一[13]。本研究结果表明,CA125水平为91.80 U/mL时诊断复发性卵巢癌的灵敏度为69.49%、准确率为71.91%、阳性预测值为85.42%,这提示CA125是监测复发性卵巢癌的重要肿瘤标志物,与以上研究结果一致。
HE4常常和CA125一起作为卵巢上皮癌的监测指标。HE4虽然被称为附睾特异性蛋白,但在卵巢上皮癌患者的血清中亦被扩增[14]。本研究结果显示,当HE4≥196.89 pmol/mL时诊断复发性卵巢癌的灵敏度为67.80%、准确率为68.54%、阳性预测值为81.63%,这提示HE4是监测复发性卵巢癌的重要肿瘤标志物。另外,本研究结果提示,CA125和HE4联合应用对复发性卵巢癌的诊断效能均高于其单独检测,且差异有统计学意义。
有研究结果表明,肺癌、胃癌、结直肠癌、肝炎、肝硬化和胰腺炎等疾病均可出现血清CA125水平升高,同时其水平随着年龄、生育状况、吸烟和月经等方面的不同而有所改变。另外,卢媛媛等[15]发现,HE4在浆液性癌、子宫内膜癌、透明细胞癌和黏液性癌中的表达水平都很高。由此可见,虽然采用血清CA125和HE4水平诊断卵巢癌的灵敏度很高,但特异度较差。
影像学检查包括CT和MRI,其多是从形态学改变的角度出发,分析病灶的部位、形态、质地、血流灌注情况以及与邻近组织的关系等,可对复发性卵巢癌的病灶进行准确定位及测量,并在疗效评估等方面提供有效的参考。但是,传统影像学方法通常是在组织出现解剖结构变化之后才能显示出有效信息,对于长径较小的病灶显示困难,尤其对于一些特殊腹膜部位(道格拉斯窝、膈下和脏器表面等)是检测盲区。另外,由于腹盆腔手术操作造成解剖结构的改变,尤其是卵巢癌患者若前期接受减瘤手术已切除大网膜等结构,或术区的瘢痕组织,都会对传统影像学诊断造成干扰[16-18]。
近年来,随着分子影像学技术的快速发展,18F-FDG PET/CT显像在复发性卵巢癌诊断中的应用越来越广。PET/CT通过观察病灶对18F-FDG的代谢程度来判断病灶的良恶性,具有灵敏度高、定位准确等特点,可为早期发现肿瘤病灶、诊断疾病、临床分期和评估预后、复发、转移等方面提供可靠的影像学依据。智生芳等[19]研究结果证实,18F-FDG PET/CT诊断复发性卵巢癌的灵敏度和准确率都较高。Thrall等[6]发现,18F-FDG PET/CT对疑似复发性卵巢癌的诊断灵敏度高达90%。本研究结果显示,18F-FDG PET/CT对疑似复发性卵巢癌的诊断灵敏度为93.22%。
本研究结果显示,18F-FDG PET/CT联合血清CA125、HE4可进一步提高其对复发性卵巢癌的诊断效能。本研究在对18F-FDG PET/CT与CA125和HE4联合检测复发性卵巢癌的分析中发现,联合检测的灵敏度为98.31%、特异度为96.67%、准确率为97.75%、阳性预测值为98.31%、阴性预测值为96.67%,高于任何一项的单独应用。这与卢仁泉等[20]的研究结果一致。
腹膜种植转移是卵巢癌最常见的转移方式,也是影响患者预后的重要因素之一。本研究结果证实,腹膜转移是复发性卵巢癌的独立预后因素,且腹膜转移阴性患者的总生存期明显高于腹膜转移阳性患者,这与Hebel等[21]的研究结果相似。另外,我们发现18F-FDG PET/CT对复发性卵巢癌腹膜转移灶检出的灵敏度和准确率均优于增强CT检查。同样,多项研究结果证实,PET/CT融合图像诊断腹膜癌的灵敏度及准确率显著高于CT和PET的单独应用[13, 22-23]。Fularz等[22]发现,与CT相比,PET/CT在检测复发性卵巢癌腹膜转移灶方面具有更高的灵敏度和准确率,这与本研究的结果一致。另外,有学者证实18F-FDG PET/CT可发现长径<0.5 cm的腹膜恶性病灶[23]。本研究结果显示,腹膜病变的分布位置对判断复发性卵巢癌腹膜转移的诊断有重要参考意义。
本研究结果显示,复发性卵巢癌患者的腹膜病变可用于预测患者的总生存期,卵巢癌术后复发但没有腹膜转移的患者的总生存期明显长于有腹膜转移的患者,这与Hebel等[21]的研究结果相似。
综上所述,腹膜转移是卵巢癌患者预后的独立危险因素;PET/CT对复发性卵巢癌的诊断及对转移灶的评估优于增强CT检查;PET/CT代谢参数与血清肿瘤学标志物联合应用,可以提高对复发性卵巢癌的诊断效能。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 周莹负责研究命题的提出与设计、研究过程的实施、数据的提供与分析,论文的起草和最终版本的修订;涂宁负责研究过程的实施、数据的提供与分析、论文的起草和最终版本的修订;冯洪燕、王科、谢新立、韩星敏负责研究过程的实施和数据的提供与分析;卜丽红负责研究命题的提出与设计、论文的起草和最终版本的修订。
18F-FDG PET/CT 联合 CA125、HE4 在诊断复发性卵巢癌及其腹膜转移预后评估的价值
Value of 18F-FDG PET/CT combined with CA125 and HE4 in the diagnosis of recurrent ovarian cancer and its peritoneal metastasis
-
摘要:
目的 探讨18F-氟脱氧葡萄糖(FDG) PET/CT联合糖类抗原125(CA125)、人附睾蛋白4(HE4)水平在复发性卵巢癌的诊断和腹膜转移预后评估中的应用价值。 方法 回顾性分析2016年1月至2021年6月在郑州大学第一附属医院接受18F-FDG PET/CT显像的89例卵巢癌术后患者的影像学资料和临床资料,其中59例经组织病理学检查确诊为复发性卵巢癌患者[复发组,中位年龄51(26~82)岁]、30例卵巢癌术后经组织病理学检查确诊为良性腹膜增厚患者[未复发组,中位年龄55(34~79)岁]。所有患者均随访至少1年。分别计算18F-FDG PET/CT和CT增强诊断复发性卵巢癌及术后残端复发、腹膜转移、淋巴结转移、腹腔外其他转移灶的效能。绘制18F-FDG PET/CT、CA125、HE4及其联合检测复发性卵巢癌的受试者工作特征曲线,并分析不同检查方法诊断复发性卵巢癌的效能。诊断效能之间的比较采用χ2检验。采用Cox模型进行多因素预后分析,评估多种因素对复发性卵巢癌患者预后的影响并计算95%置信区间(CI)。采用Kaplan-Meier生存曲线分析单个预后因素对复发性卵巢癌患者总生存期的影响。 结果 18F-FDG PET/CT诊断复发性卵巢癌的灵敏度、特异度、准确率、阳性预测值、阴性预测值分别为93.22%(55/59)、 93.33%(28/30)、 93.26%(83/89)、96.49%(55/57)、 87.50%(28/32),高于增强CT[79.66%(47/59)、73.33%(22/30)、77.52%(69/89)、85.46%(47/55)、64.71%(22/34)],各项之间的差异均有统计学意义(χ2=4.193~8.828,均P<0.05),同时,18F-FDG PET/CT对术后残端复发、腹膜转移、淋巴结转移的诊断效能(除腹膜转移阳性预测值、淋巴结转移灵敏度及阳性预测值外)均高于增强CT(χ2=2.885~8.868,均P<0.05)。18F-FDG PET/CT联合CA125及HE4诊断复发性卵巢癌的灵敏度、特异度、准确率、阳性预测值、阴性预测值分别为98.31%(58/59)、 96.67%(29/30)、97.75%(87/89)、98.31%(58/59)、 96.67%(29/30),高于三者单独应用或CA125+HE4联合应用,除18F-FDG PET/CT单独应用外,其他各项之间的差异均有统计学意义(χ2=5.192~27.101,均P<0.05),最大标准化摄取值的诊断临界值为5.60,血清CA125的诊断临界值为91.80 U/mL,HE4的诊断临界值为196.89 pmol/L。腹膜转移是复发性卵巢癌的预后独立影响因素(95%CI:3.784~819.477,P=0.003),腹膜转移阴性患者的总生存期明显高于腹膜转移阳性患者(χ2=30.320,P<0.001),腹膜转移病灶多分布于腹膜种植转移易感区。 结论 18F-FDG PET/CT在复发性卵巢癌复发诊断及转移灶评估中的效能优于增强CT;其代谢参数与血清肿瘤学标志物联合应用,可以提高对复发性卵巢癌的诊断效能。 Abstract:Objective To explore the application value of 18F-fluorodeoxyglucose (FDG) PET/CT combined with carbohydrate antigen 125 (CA125) and human epididymal protein 4 (HE4) levels in the diagnosis of recurrent ovarian cancer and prognostic evaluation of peritoneal metastasis. Methods The imaging data of 89 postoperative ovarian cancer patients who underwent 18F-FDG PET/CT in the First Affiliated Hospital of Zhengzhou University from January 2016 to June 2021 were retrospectively analyzed. Pathological examinations diagnosed 59 patients with recurrent ovarian cancer (recurrence group, median age: 51(26–82) years), whereas 30 patients with ovarian cancer were diagnosed with benign peritoneal thickening by histopathological examination after surgery (non-recurrence group, median age: 55(34–79) years). All patients were followed up for at least 1 year. The efficacy of 18F-FDG PET/CT and contrast-enhanced CT in the diagnosis of recurrent ovarian cancer and postoperative stump recurrence, peritoneal metastasis, lymph node metastasis, and other metastases outside the abdominal cavity was calculated. The receiver operator characteristic curve of 18F-FDG PET/CT, CA125, HE4 alone, and combined detection of recurrent ovarian cancer was obtained, and the efficacy of independent or combined detection of recurrent ovarian cancer was analyzed. Comparison of diagnostic efficency used χ2 text. The Cox model was used for multivariate prognostic analysis to evaluate the influence of multiple factors on the prognosis of patients with recurrent ovarian cancer and calculate the 95% confidence interval (CI). Kaplan–Meier survival curve was used to analyze the influence of a single prognostic factor on the overall survival of patients with recurrent ovarian cancer. Results The sensitivity, specificity, accuracy, positive predictive value, and negative predictive value of 18F-FDG PET/CT in diagnosing recurrent ovarian cancer were 93.22%(55/59), 93.33%(28/30), 93.26%(83/89), 96.49%(55/57) and 87.50%(28/32), respectively, which were higher than those of contrast-enhanced CT (79.66%(47/59), 73.33%(22/30), 77.52%(69/89), 85.46%(47/55), and 64.71%(22/34)). The differences among all items were statistically significant (χ2=4.193–8.828, all P<0.05). In addition, the diagnostic efficacy of 18F-FDG PET/CT for postoperative stump recurrence, peritoneal metastasis, and lymph node metastasis (except the positive predictive values of peritoneal metastasis, sensitivity, and lymph node metastasis) were all higher than those of the contrast-enhanced CT (χ2=2.885–8.868, all P<0.05). The sensitivity, specificity, accuracy, positive predictive value, and negative predictive value of 18F-FDG PET/CT combined with CA125 and HE4 in the diagnosis of recurrent ovarian cancer were 98.31%(58/59), 96.67%(29/30), 97.75%(87/89), 98.31%(58/59), and 96.67%(29/30), respectively, which were higher than the three methods were applied alone or two combined applications, except for the application of 18F-FDG PET/CT alone, the differences between the other items were statistically significant (χ2=5.192–27.101, all P<0.05). The diagnostic cut-off values of standardized uptake, serum CA125, and HE4 were 5.60, 91.80 U/mL, and 196.89 pmol/L, respectively. Peritoneal metastasis is an important independent prognostic factor for recurrent ovarian cancer (95%CI: 3.784–819.477, P=0.003). The overall survival of patients with negative peritoneal metastasis was significantly higher than that of patients with positive peritoneal metastasis (χ2=30.320, P<0.001). Peritoneal metastasis lesions were mostly distributed in the susceptible area of peritoneal implantation and metastasis. Conclusions 18F-FDG PET/CT performs better than contrast-enhanced CT in recurrence diagnosis and metastasis assessment of recurrent ovarian cancer. 18F-FDG PET/CT metabolic parameters, serum tumor markers, and the combined use of drugs can improve the diagnostic efficiency for recurrent ovarian cancer. -
表 1 89例卵巢癌术后患者的临床资料
Table 1. Clinical data of 89 postoperative ovarian cancer patients
临床资料 复发组(n=59) 非复发组(n=30) 检验值 P值 年龄[M(P25~P75),岁] 51(26~82) 55(34~79) t=5.519 0.133 病理类型[例(%)] χ2=0.001 0.976 浆液性癌 51(86.44) 26(86.67) 透明细胞癌 8(13.56) 4(13.33) 术前FIGO临床分期[例(%)] χ2=10.936 0.004 Ⅰ期 0 0 Ⅱ期 20(33.90) 21(70.00) Ⅲ期 36(61.02) 9(30.00) Ⅳ期 3(5.08) 0 术后残端复发[例(%)] 26(44.07) − − − 腹膜转移[例(%)] 51(86.44) − − − 淋巴结肿大[例(%)] 36(61.01) − − − 腹腔外其他转移[例(%)] 24(40.68) − − − CA125水平中位数[M(P25~P75),U/mL] 616.70(12.0~3852.0) 28.77(4.0~91.8) t=3.657 <0.001 HE4水平中位数[M(P25~P75),pmol/mL] 333.10(49.8~1674.0) 82.73(17.5~218.1) t=4.630 <0.001 总生存期中位数[M(P25~P75),个月] 18(2~45) 31(17~57) t=4.482 <0.001 注:FIGO为国际妇产科联盟;CA125为糖类抗原125;HE4为人附睾蛋白4。−表示无此项数据 表 2 89例卵巢癌术后患者的18F-FDG PET/CT和增强CT 显像的诊断结果比较(例)
Table 2. Comparison of 18F-FDG PET/CT and contrast enhanced CT in the diagnosis of 89 postoperative ovarian cancer patients (case)
诊断结果 随访结果 合计 阳性 阴性 18F-FDG PET/CT阳性 55 2 57 18F-FDG PET/CT阴性 4 28 32 增强CT阳性 47 8 55 增强CT阴性 12 22 34 注:FDG为氟代脱氧葡萄糖;PET/CT为正电子发射断层显像计算机体层摄影术;CT为计算机体层摄影术 表 3 59例复发性卵巢癌患者不同复发或转移部位的18F-FDG PET/CT和增强CT诊断效能的比较(%)
Table 3. Comparison of the diagnostic efficacy of 18F-FDG PET/CT and contrast-enhanced CT at different recurrence or metastasis sites in 59 patients with recurrent ovarian cancer (%)
转移/复发部位 诊断方式 灵敏度 特异度 准确率 阳性预测值 阴性预测值 复发性卵巢癌(n=59) 18F-FDG PET/CT 93.22a(55/59) 93.33a(28/30) 93.26a(83/89) 96.49a(55/57) 87.50a(28/32) 增强CT 79.66(47/59) 73.33(22/30) 77.52(69/89) 85.45(47/55) 64.71(22/34) 术后残端复发(n=26) 18F-FDG PET/CT 92.31a(24/26) 81.25a(13/16) 88.10a(37/42) 88.89a(24/27) 86.67a(13/15) 增强CT 66.67(18/27) 43.75(7/16) 59.52(25/42) 66.67(18/27) 46.67(7/15) 腹膜转移(n=51) 18F-FDG PET/CT 88.00a(44/50) 80.77a(21/26) 85.53a(65/76) 89.80(44/49) 77.78a(21/27) 增强CT 71.43(40/56) 50.00(10/20) 65.79(50/76) 80.00(40/50) 50.00(10/20) 淋巴结转移(n=36) 18F-FDG PET/CT 91.67(33/36) 85.71a(12/14) 90.00a(45/50) 94.29(33/35) 80.00a(12/15) 增强CT 80.56(29/36) 50.00(7/14) 72.00(36/50) 80.56(29/36) 50.00(7/14) 腹腔外其他转移(n=24) 18F-FDG PET/CT 91.67(22/24) 75.00(6/8) 87.50(28/32) 91.67(22/24) 75.00(6/8) 增强CT − − − − − 注:a表示与增强CT相比,差异均有统计学意义(χ2=1.847~8.868,均P<0.05)。FDG为氟代脱氧葡萄糖;PET/CT为正电子发射断层显像计算机体层摄影术;CT为计算机体层摄影术。−表示无此项数据 表 4 59例复发性卵巢癌患者的肿瘤标志物、18F-FDG PET/CT及其不同联合方式的诊断效能比较(%)
Table 4. Comparison of the diagnostic efficacy of tumor markers, 18F-FDG PET/CT and different combinations in 59 patients with recurrent ovarian cancer (%)
不同的诊断方法 灵敏度 特异度 准确率 阳性预测值 阴性预测值 CA125 术后残端复发 65.38a(17/26) 43.75a(7/16) 57.14a(24/42) 65.38a(17/26) 43.75a(7/16) 腹膜转移 72.73a(40/55) 57.14a(12/21) 68.42a(52/76) 81.63a(40/49) 44.44a(12/27) 淋巴结转移 72.22a(26/36) 42.86a(6/14) 64.00a(32/50) 76.47a(26/34) 37.50a(6/16) 腹腔外其他转移 58.33a(14/24) 37.50a(3/8) 53.13a(17/32) 73.68a(14/19) 23.08a(3/13) 复发性卵巢癌 69.49a(41/59) 76.67a(23/30) 71.91a(64/89) 85.42a(41/48) 56.10a(23/41) HE4 术后残端复发 69.23a(18/26) 50.00a(8/16) 61.90a(26/42) 69.23a(18/26) 50.00a(8/16) 腹膜转移 76.74a(39/51) 56.00a(14/25) 69.74a(53/76) 78.00a(39/50) 53.85a(14/26) 淋巴结转移 66.67a(24/36) 42.86a(6/14) 64.00a(32/50) 76.47a(26/34) 64.00a(32/50) 腹腔外其他转移 62.50a(15/24) 50.00(4/8) 59.38a(19/32) 78.95(15/19) 30.78a(4/13) 复发性卵巢癌 67.80a(40/59) 70.00a(21/30) 68.54a(61/89) 81.63a(40/49) 52.50a(21/40) 18F-FDG PET/CT 术后残端复发 92.31(24/26) 81.25(13/16) 88.10(37/42) 88.89(24/27) 86.67(13/15) 腹膜转移 88.00a(44/50) 80.77(21/26) 85.53(65/76) 89.80(44/49) 77.78(21/27) 淋巴结转移 91.67(33/36) 85.71(12/14) 90.00(45/50) 94.29(33/35) 80.00(12/15) 腹腔外其他转移 91.76(22/24) 75.00a(6/8) 87.50(28/32) 91.67(22/24) 75.00a(6/8) 复发性卵巢癌 93.22(55/59) 93.33(28/30) 93.26(83/89) 96.49(55/57) 87.50(28/32) CA125+HE4* 术后残端复发 73.08a(19/26) 56.25a(9/16) 66.67a(28/42) 73.08a(19/26) 56.25a(9/16) 腹膜转移 82.35a(42/51) 64.00a(16/25) 76.32a(58/76) 82.35a(42/51) 64.00a(16/25) 淋巴结转移 77.78a(28/36) 71.43(10/14) 76.00a(38/50) 87.50a(28/32) 55.56a(10/18) 腹腔外其他转移 70.83a(17/24) 50.00(4/8) 65.63a(21/32) 80.95a(17/21) 36.37a(4/11) 复发性卵巢癌 81.36a(48/59) 76.67a(23/30) 79.78a(71/89) 87.27a(48/55) 67.65a(23/34) CA125+HE4+18F-FDG PET/CT 术后残端复发 100.00(26/26) 93.75(15/16) 97.62(41/42) 96.30(26/27) 100.00(15/15) 腹膜转移 98.04(50/51) 96.00(24/25) 97.37(74/76) 98.04(50/51) 96.00(24/25) 淋巴结转移 97.22(35/36) 92.86(13/14) 96.00(48/50) 97.22(35/36) 92.86(13/14) 腹腔外其他转移 100.00(24/24) 87.50(7/8) 96.88(31/32) 96.00(24/25) 100.00(7/7) 复发性卵巢癌 98.31(58/59) 96.67(29/30) 97.75(87/89) 98.31(58/59) 96.67(29/30) 注:*表示当CA125>35 U/mL或HE4>140 pmol/L时视为阳性;a表示与CA125+HE4+18F-FDG PET/CT联合检查比较,差异均有统计学意义(χ2=5.192~27.101,均P<0.05)。CA125为糖类抗原125;HE4为人附睾蛋白4;PET/CT为正电子发射断层显像计算机体层摄影术 表 5 59例复发性卵巢癌患者预后影响因素加权Cox回归分析结果
Table 5. Weighted Cox regression analysis of factors affecting the prognosis of 59 patients with recurrent ovarian cancer
影响因素 风险比 95%置信区间 P值 国际妇产科联盟临床分期 7.891 0.632~98.486 0.109 CA125水平 1.020 0.995~1.045 0.119 HE4水平 1.002 0.981~1.024 0.852 腹膜转移 55.688 3.784~819.477 0.003 淋巴结肿大 0.663 0.079~5.588 0.706 腹腔外其他转移 0.733 0.107~5.025 0.752 注:CA125为糖类抗原125;HE4为人附睾蛋白4 -
[1] Bray F, Ferlay J, Soerjomataram I, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in185 countries[J]. CA: Cancer J Clin, 2018, 68(6): 394−424. DOI: 10.3322/caac.21492. [2] 严浩, 何杨, 杨润峰, 等. 复发性卵巢癌的临床特点分析[J]. 中国医师杂志, 2015, 17(4): 487−490. DOI: 10.3760/cma.j.issn.1008-1372.2015.04.003.
Yan H, He Y, Yang RF, et al. Clinical features of patients with recurrent epithelial ovarian cancer[J]. J Chin Physician, 2015, 17(4): 487−490. DOI: 10.3760/cma.j.issn.1008-1372.2015.04.003.[3] Lee CK, Lord S, Grunewald T, et al. Impact of secondary cytoreductive surgery on survival in patients with platinum sensitive recurrent ovarian cancer: analysis of the CALYPSO trial[J]. Gynecol Oncol, 2015, 136(1): 18−24. DOI: 10.1016/j.ygyno.2014.09.017. [4] 李敏, 李绪清, 颜士杰, 等. 18F-脱氧葡萄糖PET/CT联合CA125在诊断卵巢癌复发转移中的应用价值 [J]. 重庆医科大学学报, 2017, 42(12): 1635−1638. DOI: 10.13406/j.cnki.cyxb.001417.
Li M, Li XQ, Yan SJ, et al. Value of 18F-fluorodeoxyglucose PET/CT combined with serum carbohydrate antigen 125 in detecting the recurrence and metastasis of postoperative ovarian cancer [J]. J Chongqing Med Univ, 2017, 42(12): 1635−1638. DOI: 10.13406/j.cnki.cyxb.001417.[5] 邵婷, 陈秀玮. 卵巢癌的病因假说及危险因素和流行病学研究进展[J/OL]. 中华临床医师杂志: 电子版, 2013, 7(19): 8894−8897[2020-08-27]. https://d.wanfangdata.com.cn/periodical/zhlcyszz201319081. DOI: 10.3877/cma.j.issn.1674-0785.2013.19.082.
Shao T, Chen XW. Etiological hypothesis, risk factors and epidemiological research progress of ovarian cancer[J/OL]. Chin J Clin (Electron Ed), 2013, 7(19): 8894−8897[2020-08-27]. https://d.wanfangdata.com.cn/periodical/zhlcyszz201319081. DOI: 10.3877/cma.j.issn.1674-0785.2013.19.082.[6] Thrall MM, DeLoia JA, Gallion H, et al. Clinical use of combined positron emission tomography and computed tomography (FDG-PET/CT) in recurrent ovarian cancer[J]. Gynecol Oncol, 2007, 105(1): 17−22. DOI: 10.1016/j.ygyno.2006.10.060. [7] 徐晓伟, 周春英, 王俊霞, 等. 血清甲胎蛋白、人附睾蛋白4及糖类抗原125水平与卵巢癌相关性分析[J]. 临床合理用药杂志, 2015, 8(4): 162, 164. DOI: 10.15887/j.cnki.13-1389/r.2015.04.098.
Xu XW, Zhou CY, Wang JX, et al. Correlation between serum alpha fetoprotein, human epididymal protein 4 and carbohydrate antigen 125 and ovarian cancer[J]. Chin J Clin Ration Drug Use, 2015, 8(4): 162, 164. DOI: 10.15887/j.cnki.13-1389/r.2015.04.098.[8] 关鑫, 刁海丹. HE4在卵巢癌术后复发中的预测价值研究[J]. 中国实用医药, 2016, 11(31): 61−62. DOI: 10.14163/j.cnki.11-5547/r.2016.31.035.
Guan X, Diao HD. Predictive value of HE4 in postoperative recurrence of ovarian cancer[J]. China Prac Med, 2016, 11(31): 61−62. DOI: 10.14163/j.cnki.11-5547/r.2016.31.035.[9] Anthony MP, Khong PL, Zhang JB. Spectrum of 18F-FDG PET/CT appearances in peritoneal disease [J]. AJR Am J Roentgenol, 2009, 193(6): W523−W529. DOI: 10.2214/AJR.09.2936. [10] 卢淮武, 霍楚莹, 林仲秋. 《2019NCCN卵巢癌包括输卵管癌及原发性腹膜癌临床实践指南(第1版)》解读[J]. 中国实用妇科与产科杂志, 2019, 35(5): 536−546. DOI: 10.19538/j.fk2019050112.
Lu HW, Huo CY, Lin ZQ. Interpretation of 2019 NCCN clinical practice guidelines to ovariam cancer including fallopian cancer and primary peritoneal cancer (1st edition)[J]. Chin J Pract Gynecol Obstet, 2019, 35(5): 536−546. DOI: 10.19538/j.fk2019050112.[11] 卢淮武, 霍楚莹, 许妙纯, 等. 《2020NCCN卵巢癌包括输卵管癌及原发性腹膜癌临床实践指南(第1版)》解读[J]. 中国实用妇科与产科杂志, 2020, 36(4): 340−348. DOI: 10.19538/j.fk2020040113.
Lu HW, Huo CY, Xu MC, et al. Interpretation of "NCCN guidelines version 1.2020 ovarian cancer/fallopian tube cancer/primary peritoneal cancer"[J]. Chin J Pract Gynecol Obstet, 2020, 36(4): 340−348. DOI: 10.19538/j.fk2020040113.[12] 罗丹, 孔为民. 复发性卵巢癌治疗的研究进展[J]. 癌症进展, 2019, 17(17): 2003−2006. DOI: 10.11877/j.issn.1672-1535.2019.17.17.06.
Luo D, Kong WM. Research progress in the treatment of recurrent ovarian cancer[J]. Oncol Prog, 2019, 17(17): 2003−2006. DOI: 10.11877/j.issn.1672-1535.2019.17.17.06.[13] 唐梅, 林丽慧, 林元, 等. 血清人附睾蛋白4与CA125在上皮性卵巢癌中的表达及其与预后的关系[J]. 解放军医学院学报, 2018, 39(1): 24−27. DOI: 10.3969/j.issn.2095-5227.2018.01.007.
Tang M, Lin LH, Lin Y, et al. Expressions of serum human epididymis protein 4 and CA125 in patients with epithelial ovarian cancer and their relationships with prognosis[J]. Acad J Chin PLA Med Sch, 2018, 39(1): 24−27. DOI: 10.3969/j.issn.2095-5227.2018.01.007.[14] 马克奇, 王丰梅. HE4、Kif2a在卵巢上皮性肿瘤中的表达与研究进展[J]. 临床医药文献电子杂志, 2019, 6(70): 194. DOI: 10.16281/j.cnki.jocml.2019.70.173.
Ma KQ, Wang FM. Expression and research progress of HE4 and Kif2a in ovarian epithelial tumors[J]. Electron J Clin Med Lit, 2019, 6(70): 194. DOI: 10.16281/j.cnki.jocml.2019.70.173.[15] 卢媛媛, 江飞云, 吴守乐. 人血清附睾蛋白与糖类抗原检测在卵巢癌诊断中的价值[J]. 华北理工大学学报(医学版), 2018, 20(3): 214−219. DOI: 10.19539/j.cnki.2095-2694.2018.03.010.
Lu YY, Jiang FY, Wu SL. Value of serum HE4 and CA125 single and combined detection in ovarian cancer diagnosis[J]. J North China Univ Sci Technol: Health Sci Ed, 2018, 20(3): 214−219. DOI: 10.19539/j.cnki.2095-2694.2018.03.010.[16] Castellucci P, Perrone AM, Picchio M, et al. Diagnostic accuracy of 18F-FDG PET/CT in characterizing ovarian lesions and staging ovarian cancer: correlation with transvaginal ultrasonography, computed tomography, and histology [J]. Nucl Med Commun, 2007, 28(8): 589−595. DOI: 10.1097/MNM.0b013e3281afa256. [17] Gu P, Pan LL, Wu SQ, et al. CA 125, PET alone, PET-CT, CT and MRI in diagnosing recurrent ovarian carcinoma: a systematic review and meta-analysis[J]. Eur J Radiol, 2009, 71(1): 164−174. DOI: 10.1016/j.ejrad.2008.02.019. [18] Yuan Y, Gu ZX, Tao XF, et al. Computer tomography, magnetic resonance imaging, and positron emission tomography or positron emission tomography/computer tomography for detection of metastatic lymph nodes in patients with ovarian cancer: a meta-analysis[J]. Eur J Radiol, 2012, 81(5): 1002−1006. DOI: 10.1016/j.ejrad.2011.01.112. [19] 智生芳, 毕伟, 黄晓红, 等. 18F-FDG PET/CT对卵巢癌患者术后复发、转移的诊断敏感性及准确性研究 [J]. 中国CT和MRI杂志, 2016, 14(2): 100−102, 111. DOI: 10.3969/j.issn.1672-5131.2016.02.032.
Zhi SF, Bi W, Huang XH, et al. Study on the sensitivity and accuracy of PET/CT 18F-FDG in the diagnosis of postoperative recurrence and metastasis in patients with ovarian cancer [J]. Chin J CT MRI, 2016, 14(2): 100−102, 111. DOI: 10.3969/j.issn.1672-5131.2016.02.032.[20] 卢仁泉, 郭林, 沈烨红. HE4在卵巢癌诊治中的临床应用评价[J]. 中国癌症杂志, 2010, 20(9): 680−685. DOI: 10.3969/j.issn.1007-3639.2010.09.008.
Lu RQ, Guo L, Shen YH. The value of human epididymis protein 4 in patients with ovarian cancer[J]. China Oncol, 2010, 20(9): 680−685. DOI: 10.3969/j.issn.1007-3639.2010.09.008.[21] Hebel CB, Behrendt FF, Heinzel A, et al. Negative 18F-2-fluorodeoxyglucose PET/CT predicts good cancer specific survival in patients with a suspicion of recurrent ovarian cancer [J]. Eur J Radiol, 2014, 83(3): 463−467. DOI: 10.1016/j.ejrad.2013.12.006. [22] Fularz M, Adamiak P, Czepczyński R, et al. Utility of PET/CT in the diagnosis of recurrent ovarian cancer depending on CA 125 serum level[J]. Nuklearmedizin, 2015, 54(4): 158−162. DOI: 10.3413/Nukmed-0709-14-11. [23] Sironi S, Messa C, Mangili G, et al. Integrated FDG PET/CT in patients with persistent ovarian cancer: correlation with histologic findings[J]. Radiology, 2004, 233(2): 433−440. DOI: 10.1148/radiol.2332031800. -





 下载:
下载: