-
人工放射性核素99Tc、131I、60Co和天然放射性核素235U、232Th在疾病治疗[1]、辐射灭菌[2]、清洁能源的开发[3]等健康和能源领域得到了有效应用。核医学治疗过程中产生的含放射性核素的废液、患者排泄物、用药后的呕吐物及洗涤液等,一般通过沉淀凝集、离子交换等方法对其进行减容、固化后,按固体放射性废物处理,操作繁琐、成本较高。核事故以及核能再处理时放射性核素铀不可避免地释放到水体和土壤等自然环境中,对人体的细胞、神经、骨骼、免疫系统、生殖系统产生辐射损伤[4]和化学毒性伤害[5],危害环境安全并威胁人类健康。因此,对核能利用过程中产生的废水中的放射性核素进行有效去除,在降低核医学治疗后的处理成本、保护环境和人类健康等方面具有重要意义。
吸附法与其他水体中放射性核素的去除方法(如化学沉淀法[6]、离子交换法和膜分离法[7])相比,具有操作简单、易大规模应用和二次污染小等优势,有着较好的应用前景。纳米材料因其独特的尺寸效应和大量表面基团在吸附法去除水体中的放射性核素领域备受关注,氧化石墨烯和碳纳米管等碳纳米材料、纳米二氧化钛等钛基纳米材料以及氧化铁纳米颗粒和纳米0价铁等铁基纳米材料作为吸附剂被广泛用于去除水体中放射性核素的研究。相比其他两类纳米材料,铁基纳米吸附材料易于从溶液中磁性分离,且兼具纳米材料优异的表面效应、孔隙效应和易修饰性,能够克服传统吸附剂难以分离的劣势。自20世纪80年代以来,研究者对纳米材料的研究有了长足的进步,在去除水体中放射性核素领域具有巨大的应用潜能[8]。过去数十年来,基于不同特性的碳纳米材料、钛基纳米材料和铁基纳米材料应用于去除水体中放射性核素的研究成为热点。
-
由碳原子组成的直径<100 nm的碳纳米材料,有着高比表面积、高表面反应活性和较多功能基团。其中具有代表性的一种是石墨烯的衍生物——氧化石墨烯,其保持了石墨烯优异的表面性能及单层结构:引入含氧基团能够提高稳定性,增加表面反应活性位点,便于进行功能化修饰[9];通过引入含氧基团经络合作用或离子交换作用实现对放射性核素的吸附[10]。氨基[11]、聚丙烯酰胺[12]和聚苯胺[13]等功能化基团的引入可以减少氧化石墨烯的团聚现象,增强吸附能力。另一种具有代表性的是由一层或多层石墨片卷曲成管状结构且两端具有半圆球状封端结构的碳纳米管[14],其具有独特的管状结构、比表面积大、多层孔隙及碳原子不饱和性的特点,因而极易进行功能化修饰[15]。通过氧化作用除去杂质[16],引入羧甲基纤维素等功能化材料可提高碳纳米管的表面积,增强其在水中的分散性[17]。通过引入含氧基团、含氮基团和含磷基团,可以增强其对放射性核素的表面络合能力或配位结合能力[18]。
-
钛基纳米材料由于具有优良的离子交换能力、较大的比表面积、孔径及层状结构[19]而被广泛用于去除水体中放射性核素的研究,其通过表面络合作用吸附放射性核素后,将核素转变为较稳定的晶相结构,牢固地存在于吸附材料上[20]。其中具有代表性的一类是纳米二氧化钛,其通过内层络合作用对放射性核素进行吸附。通过含磷基团、富里酸和腐殖酸进行功能化修饰[21],或利用二氧化钛本身的光敏性能结合其他吸附材料,能够有效地提高纳米二氧化钛对放射性核素的吸附能力[22]。
-
铁基纳米材料具有较多的功能基团、较大的比表面积和便于磁性分离的特点,被广泛用于去除水体中放射性核素的研究。其中具有代表性的是氧化铁纳米颗粒,通过引入巯基和氨基等功能性基团,结合金属-有机骨架化合物、明胶等吸附材料,能够赋予其更强的分散稳定性和吸附性能。
碳纳米材料和钛基纳米材料在吸附放射性核素方面有一定的结构优势,但是难以从溶液中分离的缺点在一定程度上限制了其应用。相比之下,同样具有优异分散性、易功能化修饰且易于从水体中磁性分离的铁基纳米吸附材料受到越来越多的关注。氧化铁纳米颗粒、纳米0价铁和多金属纳米铁等磁性铁基纳米材料因其自身的还原性或化学稳定性,在去除水体中放射性核素领域具有很大的应用潜力。
-
通过磁改性或具有核-壳结构的Fe2O3和Fe3O4等磁性纳米材料,能够改善纳米吸附材料在水中难以分离的劣势;通过功能化修饰,可以改善其在水中的分散性,实现对放射性核素更有效地吸附。下面将具体综述磁性纳米材料在去除水体中放射性核素的研究进展,并根据组成及结构类型将其分为3类:磁改性纳米材料、核-壳结构的磁性纳米材料和其他磁性金属纳米材料。
-
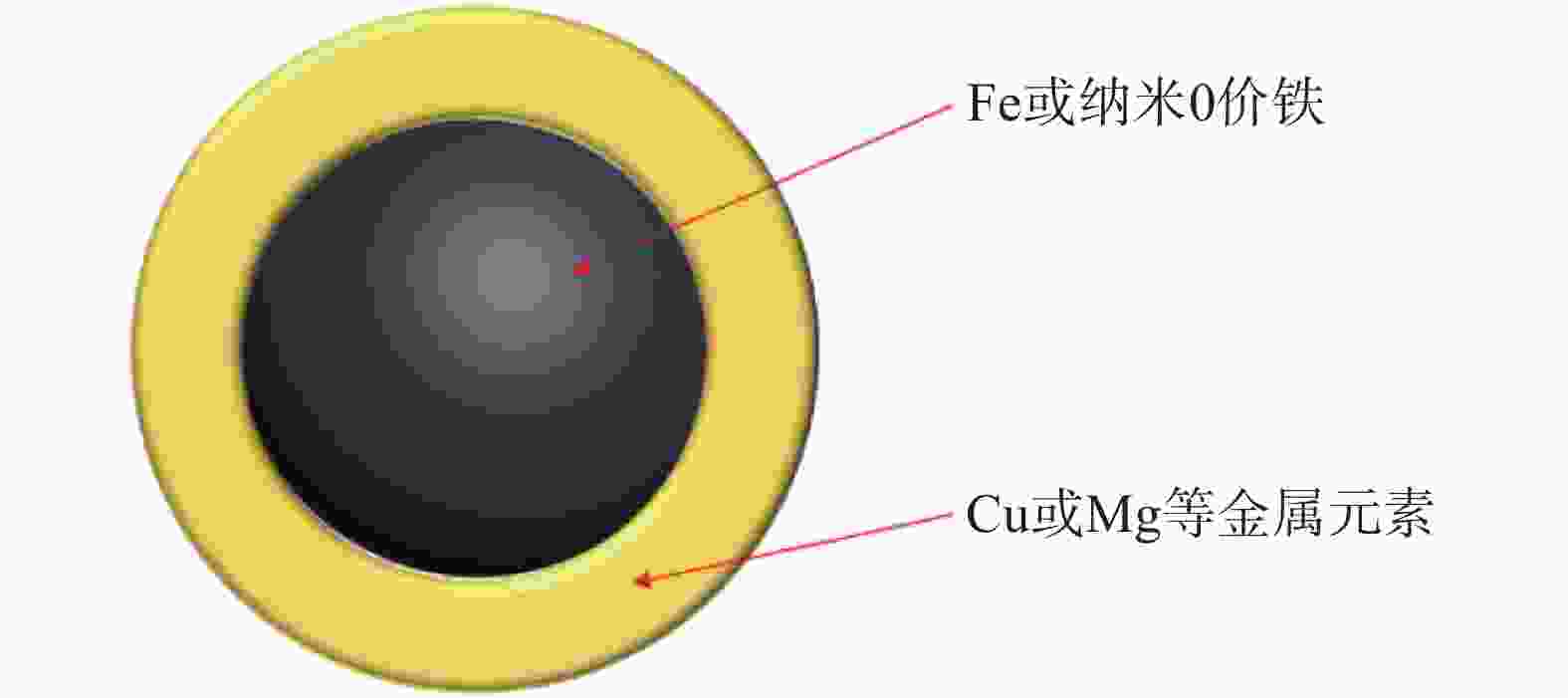
制备磁性吸附材料最便捷的思路便是将吸附材料进行磁改性,使其具有优异的磁分离性,经典的磁改性纳米吸附材料结构示意图如图1所示。Shao等[23]采用原位共沉淀法制备了葫芦[6]脲/氧化石墨烯磁性纳米复合材料,将氧化石墨烯(GO)纳米片作为固定瓜环葫芦脲(CB[6])和Fe3O4的基质用于吸附水体中的U(Ⅵ),具有良好的磁分离效率、稳定性和可回收性。Kadam等[24]制备了果胶稳定的磁性氧化石墨烯普鲁士蓝纳米复合材料,用于去除水体中的Cs(Ⅰ),果胶能够增强Go片的分离和磁铁矿在Go表面的分布,使材料具有更高的吸附容量。
图 1 磁改性纳米吸附材料代表性结构示意图
Figure 1. Schematic diagram of representative structure of magnetically modified adsorption materials
对吸附性优异的碳纳米管、金属-有机骨架化合物进行磁改性,是制备性能稳定的磁性纳米吸附材料的有效策略。Liu等[25]将氨基引入包覆有Fe3O4纳米颗粒的碳纳米管和聚磷腈骨架中制备了纳米复合材料NH2-PZS/CNT/Fe3O4,用于去除水体中的U(Ⅵ),其吸附热力学为自发吸热过程,对U(Ⅵ)具有高选择性,通过Langmuir等温模型计算出最大吸附容量为250 mg/g。Naeimi和Faghihian[26]采用六氰合铁酸钾镍对金属-有机骨架材料进行磁改性,制备了六氰合铁酸钾镍功能化的金属-有机骨架磁性纳米复合材料,用于去除水体中的Cs(Ⅰ),其吸附过程为自发吸热的单层化学吸附,最大吸附容量达到153 mg/g,此材料具有良好的可循环利用性。
利用吸附材料的还原性,能够改变放射性核素的价态,提高去除效率。Lv等[27]通过煅烧黄松和铁前体制备了纳米0价铁/磁性碳复合材料nZVI/MC,用于去除水体中的U(Ⅵ),通过将U(Ⅵ)还原为U(Ⅳ)并捕获在多孔的生物碳基质中,实现了对U(Ⅵ)的去除,具有良好的可重复利用性。此外,Hu等[28]通过快速热解负载Fe(Ⅱ)的水生植物生物质,制备了稳定在生物碳上的磁性Fe3O4纳米粒子,用于去除水体中的U(Ⅵ),这种磁性生物碳纳米材料通过内层络合作用吸附U(Ⅵ)并将其还原共沉淀为U(Ⅳ),使生物废料转化为有应用价值的吸附剂成为可能。
对于真实环境中放射性核素的去除,水体中较高的阳离子浓度以及强辐射性可能导致吸附材料的不稳定。Lujanienė 等[29]制备了一种粒子直径<10 nm的磁性Go复合材料,通过物理吸附去除水体中的Cs(Ⅰ),该复合材料的最大吸附容量达到362 mg/g,基本不受高浓度K+和Na+的影响,对天然海水中Cs(Ⅰ)的去除效率接近100%;Zhao等[30]将胺化的Fe3O4嵌入铌(Nb)取代的结晶硅钛酸盐(Nb-CST)中,制备了磁性Nb取代的结晶硅钛酸盐Mag-Nb-CST,用于去除水体中的Sr(Ⅱ)与Cs(Ⅰ),该材料具有抗辐射性,在真实海水中对Cs(Ⅰ)的去除率能达到94.19% 。这两项研究为制备真实环境中性能稳定的磁性纳米吸附材料提供了思路。
-
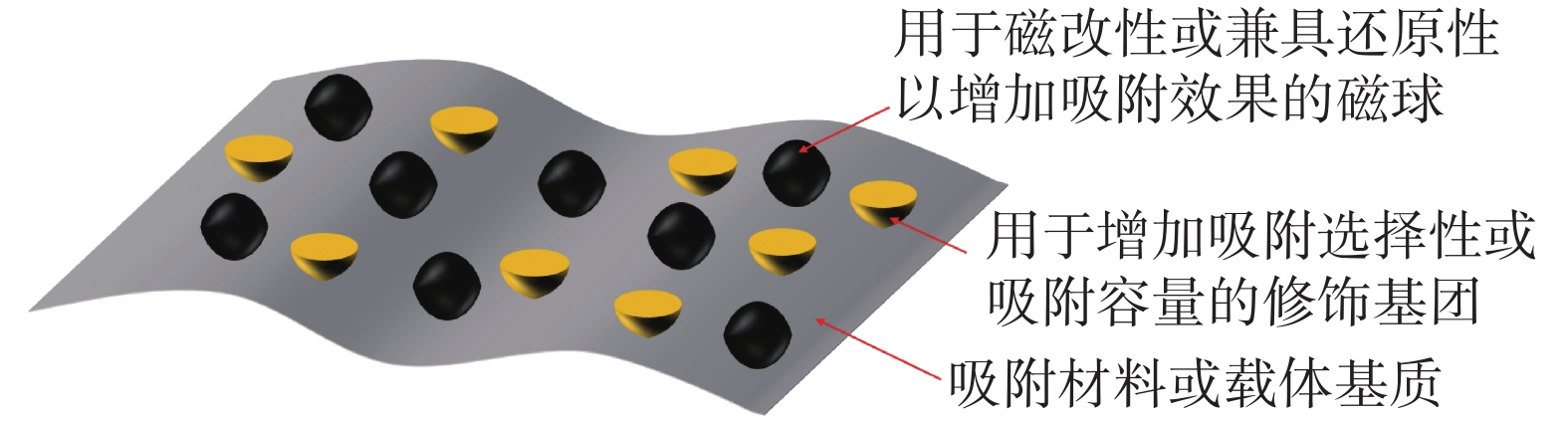
除了对吸附材料进行磁改性的策略外,通过有机聚合物或金属配位聚合物直接包覆磁性纳米材料,形成具有核-壳结构的吸附材料,也是制备磁性纳米吸附材料的可行之策,经典的核-壳结构纳米吸附材料设计如图2所示。Chen等[31]制备了一种多壳空心球形Fe3O4@MnOx复合纳米材料,去除水体中的U(Ⅵ)和Eu(Ⅲ)。其吸附热力学为吸热过程,对U(Ⅵ)和Eu( Ⅲ )的最大吸附容量分别达到106.72、 138.13 mg/g,在腐殖酸存在的条件下,材料显示出了更强的吸附能力。放射性废水一般具有强酸性,为了改善材料的稳定性,Tang等[32]采用共沉淀法制备了明胶涂层包覆的Fe3O4@Gelatin磁性复合材料,通过内层络合作用去除水体中的U(Ⅵ),为自发吸热的化学吸附过程,Fe3O4表面的明胶涂层有效地提高了材料在水溶液中的稳定性。Xu等[33]通过简便的方法制备了具有核-壳结构的磁性复合材料Fe3O4/WO3,多孔交换剂三氧化钨(WO3)均匀地分布在Fe3O4表面,用于去除水体中的Sr(Ⅱ)和Cs(Ⅰ),根据Langmuir等温模型计算出Fe3O4@WO3对Sr(Ⅱ)和Cs(Ⅰ)的理论最大吸附容量分别为44.178、53.175 mg/g,该材料也具有在酸性溶液中稳定性高的特点,为制备在真实环境中耐酸的吸附材料提供了参考。
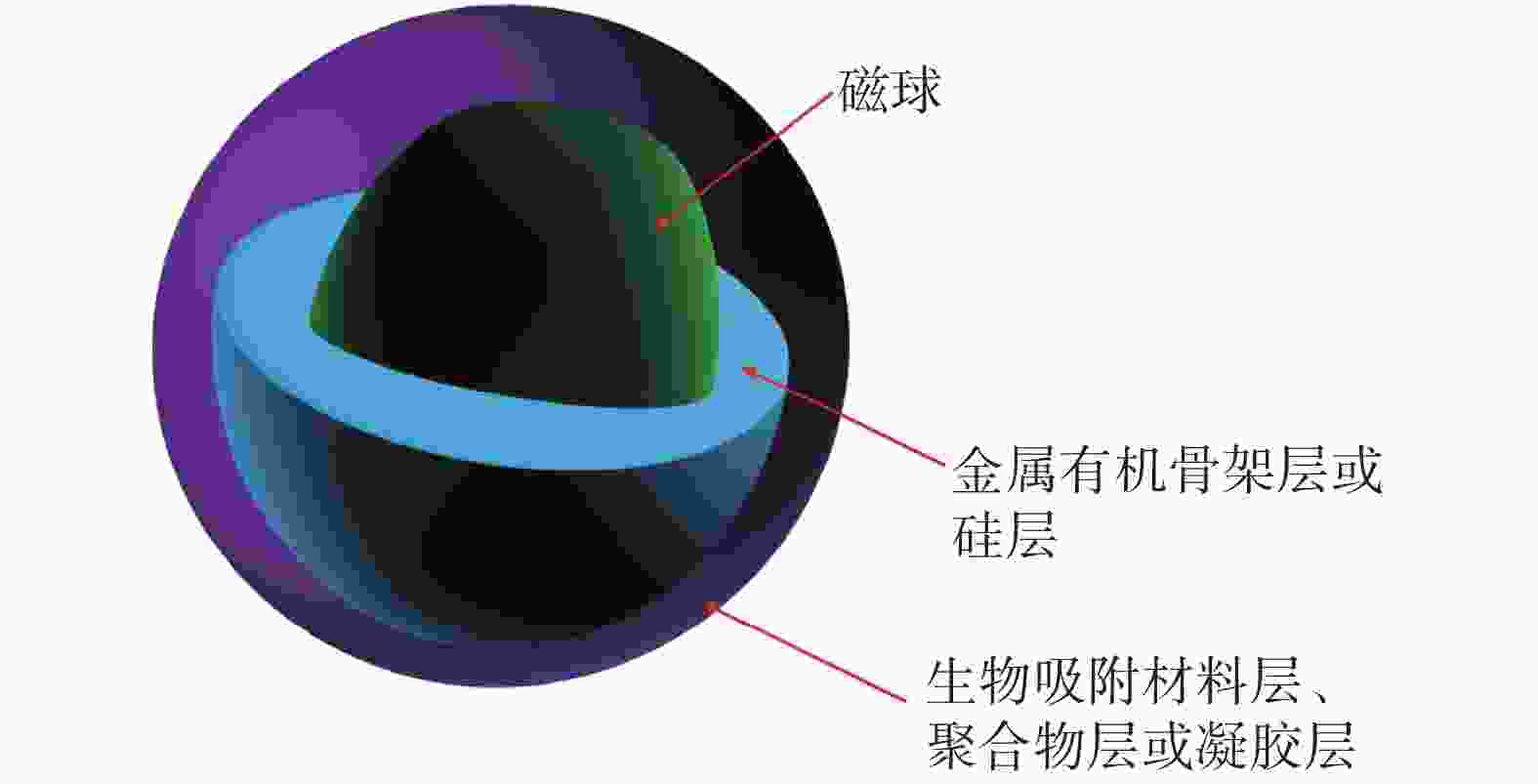
图 2 核-壳结构纳米吸附材料代表性结构示意图
Figure 2. Schematic diagram of representative structure of core-shell adsorption materials
对于真实环境中放射性核素去除的研究,Yang等[34]通过将聚乙烯亚胺(PEI)包覆在Fe3O4纳米粒子组成的磁性纳米团簇上,制备了铜氰化高铁氰化钠(NaCuHCF)功能化的磁性纳米复合材料NaCuHCF-PEI-MNC,用于去除水体中的Cs(Ⅰ),通过离子交换实现了对Cs(Ⅰ)的吸附,并进行了放射性条件下真实海水中去除Cs(Ⅰ)性能的测试,结果显示,NaCuHCF-PEI-MNC最大吸附容量达到166.67 mg/g,去除率达到99.73%。Li等[35]将偶氮胂Ⅲ修饰到Fe3O4@C核-壳结构纳米颗粒的表面,制备了Fe3O4@C@ASA,用于去除水体中的痕量U(Ⅵ),在真实的盐湖水中对U(Ⅵ)的去除率达到99%,并且不受共存离子的影响。
对磁性纳米颗粒进行功能化修饰是目前较受关注的纳米材料的设计策略。将聚多巴胺、明胶等聚合物分子修饰在磁性纳米颗粒表面,能够有效改善材料在水体中的分散性和稳定性;MOF材料[36]、活性炭和生物吸附材料[37]等的巧妙结合赋予材料更强的选择和吸附性能。由此可以预见,功能化修饰磁性纳米颗粒用于去除水体中的放射性核素极具开发前景和应用潜力。
-
通过结合铁的前驱体或其他金属离子,可能获得具有更加优异结构和吸附性能的纳米材料,图3为经典的其他磁性金属纳米材料的结构示意图。Duan等[38]通过煅烧Ni0.6Fe2.4C2O4·2H2O前驱体成功获得了对U(Ⅵ)具有良好吸附性能的多孔磁性Ni0.6Fe2.4O4微粒,该金属纳米材料表现出典型的多孔性质,孔径分布均匀,通过静电作用和内层络合作用吸附铀离子,其吸附过程为自发吸热过程,最大吸附容量为189.04 mg/g。Ivanets等[39]通过甘氨酸硝酸盐燃烧法制备了一种对水体中Co(Ⅱ)有较强吸附力的介孔结构磁性纳米离子铁氧体镁MgFe2O4,具有较好的可重复利用性。Shubair等[40]制备了纳米级0价铁和双金属纳米Fe/Cu颗粒两种磁性纳米材料用于去除水体中的Cs(Ⅰ),吸附热力学为自发放热过程,对Cs(Ⅰ)去除率均达到99%。开发多金属磁性纳米吸附材料为探索具有优异多孔结构且能够通过多种机制进行吸附的材料提供了可能。
-
磁性纳米材料具有的结构优势使其易于实现功能化改造,便于磁性分离回收,不会对环境造成二次污染,因此在去除水体中的放射性核素领域有着较好的研究前景。但目前其研究尚处于基础阶段且多针对单一[25]或同系放射性核素[30],应用于真实环境的报道较少[35]。无机离子及有机物共存条件下会对吸附时的离子传输效率或活性位点产生较大影响[29],强辐射条件可能导致纳米材料结构塌陷[34],因此,复杂条件下对水体中放射性核素去除的有效策略有待进一步探索。基于以上的回顾,以下几个方面的研究热点是值得关注的:(1)生物基质转化为吸附材料可以实现资源再利用,且易扩大生产,对其进行磁改性或将其修饰在磁性纳米材料的表面或许是实现产业化大规模生产值得探索的方向;(2)针对真实环境中存在的强辐射和复杂共存离子,对其吸附的机制进行深入研究,结合已成熟应用的吸附剂开发具有辐射稳定性、优异选择性和抗离子强度影响的磁性吸附材料具有极大的应用前景;(3)磁性纳米吸附材料具有快速磁分离回收的优点,将其用于辐射防护洗消剂的开发可能具有良好的研究前景。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 原野负责文献的搜集整理、综述的撰写;毕常芬、李祎亮负责综述的选题、修订与审阅。
去除水体中放射性核素的磁性纳米材料的研究进展
Research progress of magnetic nanomaterials for removing radionuclides from water
-
摘要: 核医学实验、核医学治疗及核事故产生的放射性核素不可避免地释放到水体、土壤等自然环境中,对环境造成极大破坏,对人体的遗传、免疫、骨骼、神经系统等产生辐射损伤和化学毒性,严重危害人类健康。磁性纳米吸附材料具有优异的表面性质和磁响应性,被广泛用于去除水体中放射性核素的研究。笔者对磁性纳米材料去除水体中放射性核素的研究进展进行综述。Abstract: Radionuclides produced by nuclear medical experiments, nuclear medicine treatment and nuclear accidents are inevitably released into the natural environment such as water and soil, causing great damage to the environment and the genetic, immune, skeletal and nervous systems of the human body. With excellent surface properties and magnetic responsiveness, magnetic nano-adsorption materials are widely used in the removal of radionuclides from water. In this paper, the research progress of the magnetic nanomaterials for removing radionuclides from water is reviewed.
-
Key words:
- Radionuclide /
- Magnetic nanomaterials /
- Water /
- Removal
-
-
[1] Kim MH, Kim SG, Kim CG, et al. A novel Tc-99m and fluorescence labeled peptide as a multimodal imaging agent for targeting angiogenesis in a murine hindlimb ischemia model[J]. Appl Radiat Isot, 2017, 121: 22−27. DOI: 10.1016/j.apradiso.2016.12.026. [2] Guzman ZMD, Cervancia CR, Dimasuay KGB, et al. Radiation inactivation of Paenibacillus larvae and sterilization of American Foul Brood (AFB) infected hives using 60Co gamma rays[J]. Appl Radiat Isot, 2011, 69(10): 1374−1379. DOI: 10.1016/j.apradiso.2011.05.032. [3] Durpel LVD, Guesdon B, Lecomte M. Industrial prospects for the optimized use of U, Pu and Th for sustainable nuclear energy deployment[J]. Energy Procedia, 2011, 7: 21−33. DOI: 10.1016/j.egypro.2011.06.004. [4] Meylan S, Incerti S, Karamitros M, et al. Simulation of early DNA damage after the irradiation of a fibroblast cell nucleus using Geant4-DNA[J/OL]. Sci Rep, 2017, 7(1): 11923[2020-03-10]. http://www.nature.com/srep/index.html. DOI: 10.1038/s41598-017-11851-4. [5] Burkart W, Danesi PR, Hendry JH. Properties, use and health effects of depleted uranium[J]. Int Congress Series, 2005, 1276: 133−136. DOI: 10.1016/j.ics.2004.09.047. [6] Atabek R, Bouniol P, Vitorge P, et al. Cement use for radioactive waste embedding and disposal purposes[J]. Cem Concr Res, 1992, 22(2/3): 419−429. DOI: 10.1016/0008-8846(92)90084-9. [7] Ambashta RD, Sillanpää MET. Membrane purification in radioactive waste management: a short review[J]. J Environ Radioact, 2012, 105: 76−84. DOI: 10.1016/j.jenvrad.2011.12.002. [8] Daer S, Kharraz J, Giwa A, et al. Recent applications of nanomaterials in water desalination: A critical review and future opportunities[J]. Desalination, 2015, 367: 37−48. DOI: 10.1016/j.desal.2015.03.030. [9] Stankovich S, Piner RD, Chen XQ, et al. Stable aqueous dispersions of graphitic nanoplatelets via the reduction of exfoliated graphite oxide in the presence of poly(sodium 4-styrenesulfonate)[J]. J Mater Chem, 2006, 16(2): 155−158. DOI: 10.1039/b512799h. [10] Jin ZX, Sheng J, Sun YB. Characterization of radioactive cobalt on graphene oxide by macroscopic and spectroscopic techniques[J]. J Radioanal Nucl Chem, 2014, 299(3): 1979−1986. DOI: 10.1007/s10967-019-06538-1. [11] Fang F, Kong L, Huang J, et al. Removal of cobalt ions from aqueous solution by an amination graphene oxide nanocomposite[J]. J Hazard Mater, 2014, 270: 1−10. DOI: 10.1016/j.jhazmat.2014.01.031. [12] Song WC, Wang XX, Wang Q, et al. Plasma-induced grafting of polyacrylamide on graphene oxide nanosheets for simultaneous removal of radionuclides[J]. Phys Chem Chem Phys, 2015, 17(1): 398−406. DOI: 10.1039/c4cp04289a. [13] Sun YB, Shao DD, Chen CL, et al. Highly efficient enrichment of radionuclides on graphene oxide-supported polyaniline[J]. Environ Sci Technol, 2013, 47(17): 9904−9910. DOI: 10.1021/es401174n. [14] Lijima S. Helical microtubules of graphitic carbon[J]. Nature, 1991, 354: 56−58. DOI: 10.1038/354056a0. [15] Belloni F, Kutahyali C, Rondinella VV, et al. Can carbon nanotubes play a role in the field of nuclear waste management?[J]. Environ Sci Technol, 2009, 43(5): 1250−1255. DOI: 10.1021/es802764g. [16] Wang XK, Chen CL, Hu WP, et al. Sorption of 243Am(Ⅲ) to multiwall carbon nanotubes[J]. Environ Sci Technol, 2005, 39(8): 2856−2860. DOI: 10.1021/es048287d. [17] Shao DD, Jiang ZQ, Wang XK, et al. Plasma induced grafting carboxymethyl cellulose on multiwalled carbon nanotubes for the removal of UO22+ from aqueous solution[J]. J Phys Chem B, 2009, 113(4): 860−864. DOI: 10.1021/jp8091094. [18] Shim JW, Park SJ, Ryu SK. Effect of modification with HNO3 and NaOH on metal adsorption by Pitch-based activated carbon fibers[J]. Carbon, 2001, 39(11): 1635−1642. DOI: 10.1016/S0008-6223(00)00290-6. [19] Yang DJ, Zheng ZF, Liu HW, et al. Layered Titanate Nanofibers as Efficient Adsorbents for Removal of Toxic Radioactive and Heavy Metal Ions from Water[J]. J Phys Chem C, 2008, 112(42): 16275−16280. DOI: 10.1021/jp803826g. [20] Yang DJ, Zheng ZF, Yuan Y, et al. Sorption induced structural deformation of sodium hexa-titanate nanofibers and their ability to selectively trap radioactive Ra(Ⅱ) ions from water[J]. Phys Chem Chem Phys, 2010, 12(6): 1271−1277. DOI: 10.1039/b911085b. [21] Tan XL, Wang XK, Fang M, et al. Sorption and desorption of Th(Ⅳ) on nanoparticles of anatase studied by batch and spectroscopy methods[J]. Colloids Surf A physicochem Eng Asp, 2007, 296(1−3): 109−116. DOI: 10.1016/j.colsurfa.2006.09.032. [22] Liu SS, Wang N, Zhang YC, et al. Efficient removal of radioactive iodide ions from water by three-dimensional Ag2O-Ag/TiO2 composites under visible light irradiation[J]. J Hazard Mater, 2015, 284: 171−181. DOI: 10.1016/j.jhazmat.2014.10.054. [23] Shao L, Wang XF, Ren YM, et al. Facile fabrication of magnetic cucurbit[6]uril/graphene oxide composite and application for uranium removal[J]. Chem Eng J, 2016, 286: 311−319. DOI: 10.1016/j.cej.2015.10.062. [24] Kadam AA, Jang J, Lee DS. Facile synthesis of pectin-stabilized magnetic graphene oxide Prussian blue nanocomposites for selective cesium removal from aqueous solution[J]. Bioresour Technol, 2016, 216: 391−398. DOI: 10.1016/j.biortech.2016.05.103. [25] Liu Y, Zhao ZP, Yuan DZ, et al. Introduction of amino groups into polyphosphazene framework supported on CNT and coated Fe3O4 nanoparticles for enhanced selective U(Ⅵ) adsorption[J]. Appl Surf Sci, 2019, 466: 893−902. DOI: 10.1016/j.apsusc.2018.10.097. [26] Naeimi S, Faghihian H. Performance of novel adsorbent prepared by magnetic metal-organic framework (MOF) modified by potassium nickel hexacyanoferrate for removal of Cs+ from aqueous solution[J]. Sep Purif Technol, 2017, 175: 255−265. DOI: 10.1016/j.seppur.2016.11.028. [27] Lv ZM, Yang SM, Chen L, et al. Nanoscale zero-valent iron/magnetite carbon composites for highly efficient immobilization of U(Ⅵ)[J]. J Environ Sci(China), 2019, 76: 377−387. DOI: 10.1016/j.jes.2018.06.001. [28] Hu QY, Zhu YL, Hu BW, et al. Mechanistic insights into sequestration of U(Ⅵ) toward magnetic biochar: Batch, XPS and EXAFS techniques[J]. J Environ Sci(China), 2018, 70: 217−225. DOI: 10.1016/j.jes.2018.01.013. [29] Lujanienė G, Šemčuk S, Lečinskytė A, et al. Magnetic graphene oxide based nano-composites for removal of radionuclides and metals from contaminated solutions[J]. J Environ Radioactiv, 2017, 166(Pt 1): 166−174. DOI: 10.1016/j.jenvrad.2016.02.014. [30] Zhao XD, Meng QH, Chen G, et al. An acid-resistant magnetic Nb-substituted crystalline silicotitanate for selective separation of strontium and/or cesium ions from aqueous solution[J]. Chem Eng J, 2018, 352: 133−142. DOI: 10.1016/j.cej.2018.06.175. [31] Chen ZS, Jian W, Pu ZX, et al. Synthesis of magnetic Fe3O4/CFA composites for the efficient removal of U(Ⅵ) from wastewater[J]. Chem Eng J, 2017, 320: 448−457. DOI: 10.1016/j.cej.2017.03.074. [32] Tang PF, Shen J, Hu ZD, et al. High-efficient scavenging of U(Ⅵ) by magnetic Fe3O4@Gelatin composite[J]. J Mol Liq, 2016, 221: 497−506. DOI: 10.1016/j.molliq.2016.06.008. [33] Xu B, Zhu YK, Liu HB, et al. The kinetic and thermodynamic adsorption of Eu(Ⅲ) on synthetic maghemite[J]. J Mol Liq, 2016, 221: 171−178. DOI: 10.1016/j.molliq.2016.05.055. [34] Yang HM, Hwang KS, Park CW, et al. Sodium-copper hexacyanoferrate-functionalized magnetic nanoclusters for the highly efficient magnetic removal of radioactive caesium from seawater[J]. Water Res, 2017, 125: 81−90. DOI: 10.1016/j.watres.2017.08.037. [35] Li P, Wang JJ, Wang XL, et al. Arsenazo-functionalized magnetic carbon composite for uranium(Ⅵ) removal from aqueous solution[J]. J Mol Liq, 2018, 269: 441−449. DOI: 10.1016/j.molliq.2018.08.073. [36] Yuan GY, Zhao CS, Tu H, et al. Removal of Co(Ⅱ) from aqueous solution with Zr-based magnetic metal-organic framework composite[J]. Inorg Chim Acta, 2018, 483: 488−495. DOI: 10.1016/j.ica.2018.08.057. [37] Yin YN, Wang JL, Yang XY, et al. Removal of strontium ions by immobilized saccharomyces cerevisiae in magnetic chitosan microspheres[J]. Nucl Eng Technol, 2017, 49(1): 172−177. DOI: 10.1016/j.net.2016.09.002. [38] Duan SX, Liu X, Wang YN, et al. Highly efficient entrapment of U(Ⅵ) by using porous magnetic Ni0.6Fe2.4O4 micro-particles as the adsorbent[J]. J Taiwan Inst Chem E, 2016, 65: 367−377. DOI: 10.1016/j.jtice.2016.05.041. [39] Ivanets AI, Srivastava V, Roshchina MY, et al. Magnesium ferrite nanoparticles as a magnetic sorbent for the removal of Mn2+, Co2+, Ni2+ and Cu2+ from aqueous solution[J]. Ceram Int, 2018, 44(8): 9097−9104. DOI: 10.1016/j.ceramint.2018.02.117. [40] Shubair T, Eljamal O, Khalil AME, et al. Novel application of nanoscale zero valent iron and bimetallic nano-Fe/Cu particles for the treatment of cesium contaminated water[J]. J Environ Chem Eng, 2018, 6(4): 4253−4264. DOI: 10.1016/j.jece.2018.06.015. -





 下载:
下载: