-
结直肠癌是最常见的恶性肿瘤之一,复发和转移是其主要的病死原因。肝脏是结直肠癌血行转移的最常见部位,超过一半的患者在其疾病发展过程中会发生肝转移[1-3]。结直肠癌肝转移(colorectal cancer liver metastases,CRCLM)是影响患者预后的重要因素之一,手术是其主要的治疗手段,但只有7%~25%的CRCLM患者在确诊后可以行根治性切除手术[4-6],大部分患者由于病灶数量、大小和位置等原因无法行根治性手术,只能通过化疗、射频和靶向治疗等手段延长生存期及提高生活质量。目前,CRCLM患者治疗后的5年总生存率已提高至35%~50%或以上,而未经治疗的患者5年生存率低于5%~10%[2-4]。因此,早期准确地诊断CRCLM并明确转移灶的数量和位置等,对于肿瘤的分期、治疗方案的选择和预后的评估都具有重要的临床意义。CT、MRI和18F-FDG PET/CT等均是检测和诊断CRCLM的重要手段,近年来出现的一体化PET/MRI亦被广泛关注。目前,关于CRCLM 18F-FDG PET/MRI显像的文献报道较少,笔者回顾性分析27例结直肠癌患者同日先后行全身18F-FDG PET/CT显像与上腹部PET/MRI局部显像的相关资料,评估18F-FDG PET/MRI显像对CRCLM的诊断价值,并比较其与PET/CT的差异。
-
回顾性分析2018年9月至2019年9月于宁波明州医院PET中心行全身18F-FDG PET/CT显像和上腹部18F-FDG PET/MRI显像并疑似有CRCLM的27例患者的临床资料,其中男性19例、女性8例,年龄28~78(60.6±12.2)岁;未行手术患者11例,已行手术患者16例。纳入标准:(1)临床资料及影像学资料完整;(2)经组织病理学检查证实为结直肠癌;(3)无其他肿瘤病史或伴有其他部位的原发肿瘤;(4)经组织病理学检查或影像学随访可对肝内病灶做出最终诊断;(5)近期(<3个月)无肝脏手术、射频消融、化疗等治疗史。反之则为排除标准。所有患者均在行PET/CT和PET/MRI前知悉流程及注意事项,并签署了知情同意书。本研究获得宁波明州医院伦理学委员会的批准(批准号:明伦202005001)。
-
PET/CT显像仪为德国Siemens公司Biograph mCT扫描仪。18F-FDG由南京江原安迪科正电子研究发展有限公司提供,放射化学纯度>95%。所有患者均禁食6 h以上,空腹血糖<11.1 mmol/L。患者按体重(3.70~5.55 MBq/kg)静脉注射18F-FDG显像剂,安静平躺休息60 min后行PET/CT显像。PET/CT扫描前饮水500 mL并排尿,扫描范围从颅顶至股骨中段。CT扫描参数:管电压120 kV,管电流为自动毫安,螺距0.8,球管单圈螺旋时间为0.5 s,层厚为3.0 mm。CT重建参数:层厚3.0 mm,层间距2.0 mm。PET扫描参数:采用三维模式,共扫描7~10个床位,1.5 min/床位。PET重建参数:采用有序子集最大期望值迭代法(OSEM)重建图像,2次迭代,24个子集,高斯滤波半高宽为4.0 mm,散射校正,利用CT图像进行衰减校正。将PET和CT图像传送到Syngo MMWP工作站,进行帧对帧图像对位融合。
PET/MRI显像仪为美国GE公司Signa PET/MRI(3.0T)扫描仪。PET/MRI显像在PET/CT显像后进行,扫描范围自肝脏上缘至肾脏下缘。基于MRI的衰减校正序列与PET采集同时开始,MRI扫描序列包括:横断面T1加权成像(weighted imaging,WI)采用屏气LAVA(可变容积加速肝脏采集)-Flex序列进行扫描,横断面T2WI采用呼吸门控Propeller序列进行扫描,横断面弥散加权成像(diffusion-weighted imaging,DWI)(b值为800 s/mm2和0 s/mm2)采用呼吸门控进行扫描,冠状面T2WI采用T2单次激发快速自旋回波(SSFSE)进行扫描。PET采用时间飞行(TOF)及点扩散函数(PSF)技术,选择有序子集最大期望值迭代(OSEM)算法进行图像重建。PET三维采集与MRI检查同步进行,以确保PET和MRI数据具有最佳的时间和解剖位置配准,采集时长10 min。
分别应用显像仪系统自带的Syngo MMWP和Advantage工作站对PET/CT和PET/MRI数据进行图像后处理。
-
PET/CT图像由2位具有5年以上PET/CT诊断经验的核医学医师采用视觉分析法独立阅片,且不参考PET/MRI图像。PET/MRI图像由2位具有1年以上PET/MRI诊断经验的核医学医师采用视觉分析法独立阅片,且不参考PET/CT图像。为避免回忆偏差,两次阅片时间间隔2周以上,意见不一致时协商达成一致。PET/CT的评价标准:(1)当病灶18F-FDG代谢高于周围肝组织时,无论CT上病灶呈何种密度均视为阳性;(2)当病灶代谢与周围肝组织差异不明显时,CT上病灶呈稍低密度,且不符合囊肿和血管瘤典型特征的病灶视为阳性;(3)当病灶代谢低于周围肝组织时,无论CT上病灶呈何种密度均视为阴性。PET/MRI的评价标准:(1)当病灶18F-FDG代谢高于周围肝组织时,无论MRI上病灶呈何种信号均视为阳性;(2)当病灶代谢与周围肝组织差异不明显时,MRI上病灶呈T1WI低信号,T2WI高信号,DWI呈弥散受限,且不符合囊肿和血管瘤典型特征的病灶视为阳性;(3)当病灶代谢低于周围肝组织时,无论MRI上病灶呈何种信号均视为阴性。在CT及MRI图像上测量并记录病灶长径。每个患者最多测量10个病灶,若超过10个病灶,则选择PET/CT或PET/MRI图像上能识别的最大10个病灶。分别基于患者及病灶分析,比较2种检查方法对CRCLM的各项诊断效能。并根据CRCLM病灶的长径,分为长径≤1 cm组、1 cm<长径≤2 cm组和长径≥2 cm组,计算和比较各组的2种检查方法诊断CRCLM的灵敏度。
-
对所有符合纳入、排除标准的患者,根据其组织病理学检查、现有的检查结果以及随访数据进行综合考虑[7-10],具体的诊断标准:(1)行组织病理学检查的患者以其结果为“金标准”;(2)在PET/CT、PET/MRI以及另外一种影像学检查方法中[CT和(或)MRI]均有典型CRCLM表现者判定为恶性[9];(3)在影像学随访过程中,病灶长径增大(增大>20%[10])、增多者判定为恶性[9-10];(4)在随访期间行化疗或其他治疗的患者,病灶长径缩小(缩小>20%[10])、减少者判定为恶性;(5)在随访间期未行治疗的患者,影像学随访(>3个月)期间病灶缩小或无明显改变者判定为良性;(6)影像学随访(>3个月)期间肝内均未见任何病灶者判定为良性。本研究中纳入的患者影像学随访时间为97~386 d,均未失访。
-
应用SPSS 24.0软件对数据进行统计学分析。定性资料用率表示,采用McNemar检验比较18F-FDG PET/CT与PET/MRI诊断CRCLM的灵敏度、特异度和准确率;采用Pearson χ2检验比较2种检查方法诊断CRCLM的阳性预测值和阴性预测值。P<0.05为差异有统计学意义。
-
根据诊断标准可知,27例结直肠癌患者中,肝转移瘤患者16例,肝良性病变患者11例。16例肝转移瘤患者中,6例肝内病灶经组织病理学检查确诊,6例在18F-FDG PET/CT、PET/MRI和另外一种影像学检查[CT和(或)MRI]中均有典型肝转移瘤表现,4例在随访期间(随访时间:126~386 d)显示病灶增多或体积增大。11例肝良性病变患者在随访期间(97~308 d)肝内病灶均无明显改变(随访期间无肿瘤治疗史)。在27例结直肠癌患者中,共发现118个肝内病灶,其中肝转移瘤病灶72个,长径(2.2±1.5) cm;肝良性病灶46个,长径(0.8±0.6) cm。
-
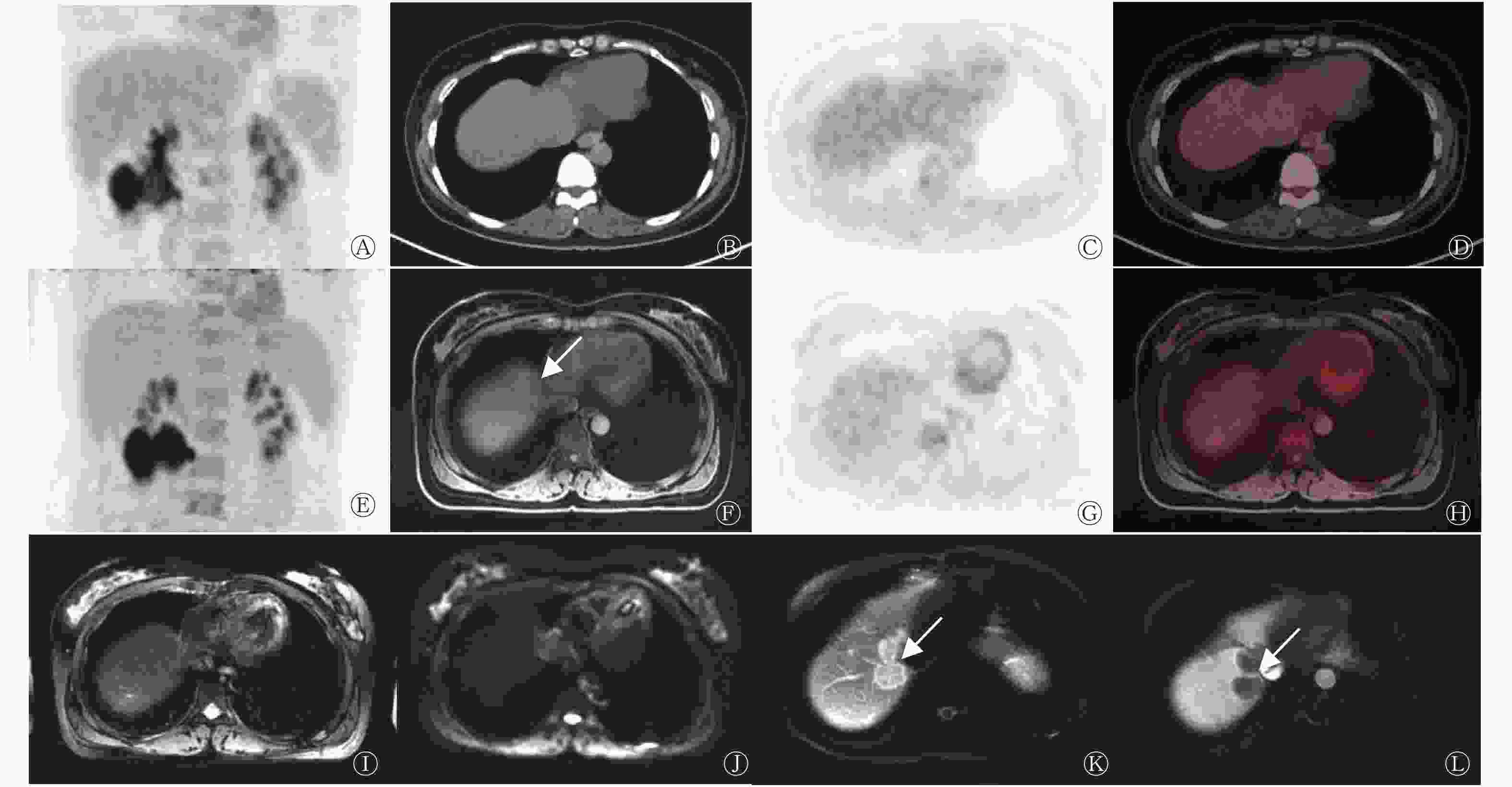
由表1可知,18F-FDG PET/MRI诊断CRCLM的灵敏度和准确率均高于PET/CT,但差异均无统计学意义(均P>0.05),二者特异度一致。16例CRCLM患者中,PET/CT假阴性3例,PET/MRI检出了其中的2例,病灶长径均<1.0 cm(图1),另外1例PET/MRI亦呈假阴性,病灶长径为1.1 cm。该假阴性患者的病灶在PET/CT中不可见,在PET/MRI中未见18F-FDG代谢增高,影像学随访结果示病灶明显增大(图2)。
影像学方法 灵敏度 特异度 准确率 阳性预测值 阴性预测值 PET/CT 81(13/16) 100(11/11) 89(24/27) 100(13/13) 79(11/14) PET/MRI 94(15/16) 100(11/11) 96(26/27) 100(15/15) 92(11/12) 检验值 − − − χ2=0.000 χ2=0.851 P值 0.500 1.000 0.500 1.000 0.598 注:FDG为氟脱氧葡萄糖;PET为正电子发射断层显像术;CT为计算机体层摄影术;MRI为磁共振成像;−为McNemar检验,无检验值 表 1 27例结直肠癌肝转移患者18F-FDG PET/CT与PET/MRI 诊断效能的比较 (%)
Table 1. Comparison of diagnostic efficacy between 18F-FDG PET/CT and PET/MRI in colorectal cancer liver meastases based on 27 patients (%)
-
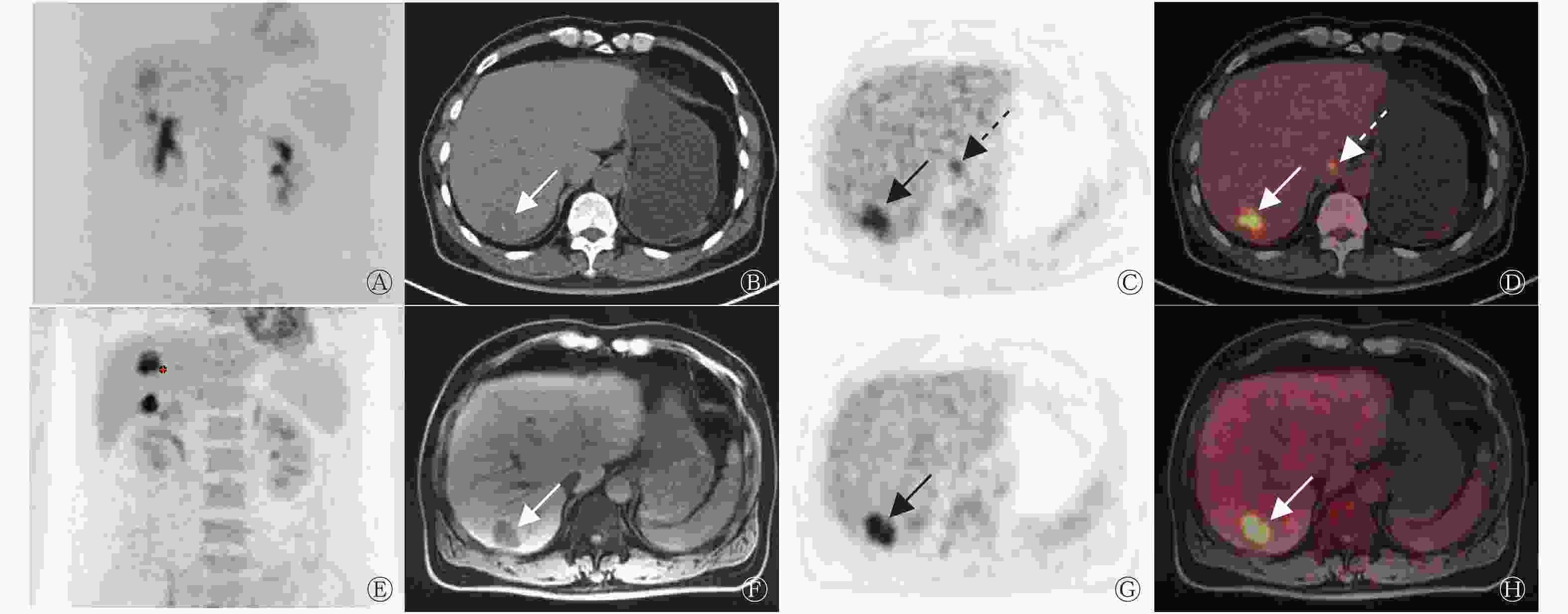
由表2可知,18F-FDG PET/MRI诊断CRCLM的灵敏度、准确率和阴性预测值均高于PET/CT,且差异有统计学意义(均P<0.05)。1例患者在行PET/CT检查时,在肝尾状叶见18F-FDG代谢增高病灶,考虑为肝转移瘤,但PET/MRI及影像学随访在肝尾状叶均未见异常病灶,考虑肝尾状叶病灶在PET/CT上呈假阳性(图3)。在16例CRCLM患者中,与PET/CT相比,44%(7/16)的患者在PET/MRI上发现更多肝转移病灶。72个肝转移病灶中,PET/CT假阴性病灶27个,其中长径≤1 cm组的病灶25个,1 cm<长径≤2 cm组的病灶2个;PET/MRI假阴性病灶11个,其中长径≤1 cm组的病灶10个,1 cm<长径≤2 cm组的病灶1个。
影像学
方法灵敏度 特异度 准确率 阳性
预测值阴性
预测值PET/CT 63(45/72) 98(45/46) 76(90/118) 98(45/46) 63(45/72) PET/MRI 85(61/72) 100(46/46) 91(107/118) 100(61/61) 81(46/57) 检验值 − − − χ2=1.339 χ2=5.072 P值 <0.001 1.000 <0.001 0.430 0.024 注:表中,FDG为氟脱氧葡萄糖;PET为正电子发射断层显像术;CT为计算机体层摄影术;MRI为磁共振成像;−为McNemar检验,无检验值 表 2 18F-FDG PET/CT与PET/MRI在27例结直肠癌患者肝 转移病灶(72个)中诊断效能的比较 (%)
Table 2. Comparison of diagnostic efficacy between 18F-FDG PET/CT and PET/MRI in colorectal cancer patients(27) liver meastases lesion(72) (%)
-
长径≤1 cm组的病灶27个,PET/MRI的诊断灵敏度63%(17/27)高于PET/CT 7%(2/27),且差异有统计学意义(McNemar检验,P<0.05);1 cm<长径≤2 cm组的病灶8个,PET/MRI的诊断灵敏度88%(7/8)高于PET/CT 75%(6/8),差异无统计学意义(McNemar检验,P>0.05);长径>2 cm组的病灶37个,2种方法的诊断灵敏度均为100%(37/37)。
-
结直肠癌是一种常见的消化道恶性肿瘤,其发病与大肠慢性炎症、大肠腺瘤、高脂肪低纤维素饮食等密切相关[11]。结直肠癌起病较为隐匿,早期多无明显临床表现,待出现明显症状时大多已发生转移。肝脏是结直肠癌最主要且最重要的远处转移器官,1/2以上的结直肠癌患者可出现肝转移,并且肝脏常常是其唯一的远处转移器官[11]。因此,及时、准确地诊断肝转移对患者的临床治疗及预后有重要的意义。
PET/MRI作为新型的影像学技术,结合了PET的功能影像信息和MRI较高的软组织分辨率及解剖定位信息,在大大降低辐射剂量的同时提高了对病灶定位及定性的准确率,在肿瘤影像的临床应用中具有良好的前景[12-13]。PET/MRI和PET/CT虽然都是功能与形态融合的显像方式,但并不是MRI简单地替代PET/CT中的CT,2种设备的PET图像在数据采集与处理、图像重建和采集时间等方面存在较大差异。
18F-FDG PET/CT与PET/MRI对CRCLM的诊断效能各文献报道不一。Maffione等[14]的一项包括18篇文献1059例患者的Meta分析结果显示,PET和PET/CT对CRCLM患者诊断的灵敏度为93%,高于本研究的结果,其原因可能是PET/MRI可检出较多的肝脏小转移灶,而这些病灶在PET/CT上多呈假阴性。Lee等[9]对55例结直肠癌患者的研究结果显示,PET/MRI对CRCLM患者诊断的灵敏度为85%,对CRCLM病灶诊断的灵敏度为88%~89%,与本研究结果较为一致。Brendle等[15]对15例结直肠癌患者的研究结果显示,PET/MRI诊断CRCLM病灶的灵敏度为71%、准确率为74%,低于本研究结果,其原因可能是在他们的研究中,黏液性肿瘤比例较多(38.2%)。Berger等[16]的研究结果显示,黏液性肿瘤对18F-FDG的摄取较低,PET只能发现59%的黏液性肿瘤,同时黏液性肿瘤与实体肿瘤相比只有轻微的弥散受限[15, 17],从而影响病灶的检出。
在有关肝转移瘤PET/MRI与PET/CT的对比研究中,Donati等[18]对37例患者的研究结果显示,PET/MRI与PET/CT诊断肝转移病灶的灵敏度分别为93%和76%,准确率分别为92%~96%和85%。Beiderwellen等[19]对32例患者的研究结果显示,PET/MRI与PET/CT诊断肝转移病灶的灵敏度分别为92.2%和67.8%、准确率分别为96.1%和82.4%,均高于本研究结果。其可能原因:(1)本研究中,小的肝转移病灶较多,其中≤1 cm的病灶占37.5%(27/72);而在Donati等[18]的研究中,≤1 cm的肝转移病灶仅占18.2%(10/55)。Hong等[20]的一项Meta分析结果显示,PET/CT与PET/MRI对长径≤1 cm的肝转移病灶的诊断灵敏度分别为29%~30%和44%~71%,均显著低于对长径>1 cm的诊断灵敏度(87%~89%和50%~98%),本研究亦得出类似的结果。(2)本研究中的PET/CT和PET/MRI均使用平扫CT和平扫MRI;而在Donati等[18]的研究中,MRI显像使用的是肝胆特异性对比增强剂(Gd-EOB-DTPA),Beiderwellen等[19]的研究使用的是增强CT。既往研究结果显示,增强CT和增强MRI(Gd-EOB-DTPA)均有助于提高对肝转移的诊断灵敏度和准确率[21-22]。(3)在Donati等[18]和Beiderwellen等[19]的研究中,原发肿瘤并非都是结直肠癌,而肝转移灶的18F-FDG摄取与原发肿瘤病理类型密切相关。
本研究及其他一些学者的研究结果均显示,PET/MRI诊断肝转移病灶的灵敏度和准确率均显著高于PET/CT,尤其是在长径≤1 cm的病灶中,并且PET/MRI在显示病灶的清晰度和诊断置信度等方面均优于PET/CT[15, 18-20]。其可能原因:(1)MRI具有较高的软组织分辨率,可综合不同序列进行分析,能较好地显示肝脏的解剖细节,小的转移灶在MRI图像上即可呈现异常信号。(2)MRI中的DWI是目前唯一能够从分子水平检测活体组织内水分子扩散运动的无创检测方法。当组织发生病变时,其细胞功能和代谢发生变化,水分子扩散运动受限,在DWI序列上呈高信号,有助于早期准确地检测出肝内小的转移病灶[22]。(3)PET/MRI为同步采集,不受呼吸运动的影响,因此提高了PET图像与MRI图像的融合配准,有助于肝顶部病灶的检出。(4)PET/MRI的PET采集时间(10 min)长于PET/CT(1.5 min),采集时间越长,图像质量越好,从而有利于肝转移灶的检出。
18F-FDG并非肿瘤特异性显像剂,当合并炎症和感染时均可出现18F-FDG代谢增高,从而造成假阳性结果。由于肝脏可生理性摄取18F-FDG,因此体积较小、摄取18F-FDG不明显的病灶易造成假阴性结果。另外,部分恶性程度较低或一些特殊病理类型的CRCLM病灶(如黏液腺癌、印戒细胞癌)对18F-FDG的摄取量有限[16, 23],亦可造成假阴性结果。本研究中,大部分假阴性病灶的长径均≤1 cm,PET/MRI显像中仅有1个假阴性病灶的长径>1 cm,18F-FDG代谢均未见增高,这可能与病灶的病理类型或乏血供有关。因此在临床工作中,不能仅依靠病灶代谢的高低来判断其性质,应结合CT和MRI图像中病灶的形态、临床特点、实验室检查和其他影像学检查结果综合考虑,以减少假阳性率和假阴性率。
本研究仍存在许多局限性:(1)本研究为回顾性研究,存在一定程度的选择偏倚,可能会遗漏一些假阴性或假阳性病例;(2)部分病例通过影像学随访而非组织病理学检查结果诊断,部分小的病灶可能通过现有的影像学检查方法无法检出,因此PET/CT和PET/MRI诊断CRCLM实际的灵敏度可能低于本研究结果;(3)病例数量有限,得出的结果可能存在偏差,需要更大样本做进一步研究;(4)没有对肝外转移情况进行比较,PET/MRI扫描局限于上腹部,重点比较了2种检查方法在肝转移中的差异,但全身显像是PET/CT检查的一个重要优势,通过PET/CT了解患者全身情况仍非常必要。
综上所述,与PET/CT相比,PET/MRI对CRCLM病灶的检出及定性具有明显优势,尤其是在长径≤1 cm的病灶中,有助于临床早期准确诊断CRCLM并更好地指导患者的临床治疗。未来仍需大样本前瞻性研究进一步证实PET/MRI的临床应用价值。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 于军负责研究命题的提出与设计、数据的收集与分析、论文的撰写;张莺、陈聪负责研究命题的提出与设计、论文的审阅;任东栋负责研究命题的设计、数据的收集与分析、论文的审阅;李洋、任春玲负责数据的获取与分析;蔡君负责图像的处理。
18F-FDG PET/CT与PET/MRI在结直肠癌肝转移诊断中的比较分析
Comparative analysis of 18F-FDG PET/CT and PET/MRI in the diagnosis of colorectal cancer liver metastases
-
摘要:
目的 比较18F-氟脱氧葡萄糖(FDG) PET/CT与PET/MRI显像对结直肠癌肝转移的诊断价值。 方法 回顾性分析2018年9月至2019年9月于宁波明州医院行全身18F-FDG PET/CT显像及上腹部18F-FDG PET/MRI显像,并疑似有结直肠癌肝转移的27例患者的临床资料,其中男性19例、女性8例,年龄28~78(60.6±12.2)岁。肝内病灶的最终诊断以组织病理学检查或影像学随访结果为参考标准,分别计算2种检查方法诊断结直肠癌肝转移瘤的灵敏度、特异度、准确率、阳性预测值和阴性预测值。根据肝转移病灶大小分长径≤1 cm、1 cm<长径≤2 cm、长径>2 cm 3组,分别计算各组的2种检查方法的灵敏度。采用McNemar检验比较2种检查方法诊断结直肠癌肝转移瘤的灵敏度、特异度和准确率;采用χ2检验比较2种检查方法诊断结直肠癌肝转移瘤的阳性预测值和阴性预测值。 结果 基于27例结直肠癌患者分析,18F-FDG PET/MRI诊断肝转移瘤的灵敏度、准确率[94%(15/16)、96%(26/27)]均高于PET/CT[81%(13/16)、89%(24/27)],但差异均无统计学意义(McNemar检验,均P>0.05)。基于病灶分析,在27例结直肠癌患者中共发现118个肝内病灶,其中肝转移瘤病灶72个,18F-FDG PET/MRI诊断结直肠癌肝转移瘤病灶的灵敏度、准确率、阴性预测值[85%(61/72)、91%(107/118)、81%(46/57)]均高于PET/CT[63%(45/72)、76%(90/118)、63%(45/72)],且差异均有统计学意义(McNemar检验,χ2=5.072,均P<0.05)。对于长径≤1 cm的肝转移病灶,18F-FDG PET/MRI的诊断灵敏度[63%(17/27)]明显高于PET/CT[7%(2/27)],且差异有统计学意义(McNemar检验,P<0.05)。 结论 与18F-FDG PET/CT相比,18F-FDG PET/MRI在结直肠癌肝转移中具有更高的诊断价值,尤其是在长径≤1 cm的病灶中。 -
关键词:
- 结直肠肿瘤 /
- 肝 /
- 肿瘤转移 /
- 氟脱氧葡萄糖F18 /
- 正电子发射断层显像术 /
- 体层摄影术,X线计算机 /
- 磁共振成像
Abstract:Objective To compare the diagnostic values of 18F-fluorodeoxyglucose (FDG) PET/CT and PET/MRI in colorectal cancer liver metastases. Methods A retrospective analysis was performed on data collected from 27 patients with suspected colorectal cancer liver metastases, including 19 males and 8 females aged 28–78 (60.6±12.2) years old who underwent whole-body 18F-FDG PET/CT and upper abdomen 18F-FDG PET/MRI examination in Ningbo Mingzhou Hospital from September 2018 to September 2019. The result of histopathological examination and imaging follow-up served as the reference standards. The sensitivity, specificity, accuracy, positive predictive value (PPV) and negative predictive value (NPV) of both methods in the diagnosis of liver metastases were calculated. Liver metastases lesions were divided into three groups according to size: lesion length≤1 cm, 1 cm< lesion length≤2 cm, and lesion length>2 cm. The sensitivity of 18F-FDG PET/CT and PET/MRI in each group were calculated. McNemar test was used to compare the sensitivity, specificity, and accuracy of the two methods in the diagnosis of liver metastases. χ2 test was used to compare the PPV and NPV of the two methods in the diagnosis of liver metastases. Results Based on 27 patients with colorectal cancer analysis, the sensitivity and accuracy of 18F-FDG PET/MRI in the diagnosis of liver metastases (94% (15/16) and 96% (26/27)) were higher than those of PET/CT (81% (13/16) and 89% (24/27)), but the difference was not statistically significant (McNemar test, both P>0.05). Based on lesion analysis, mong the 118 liver lesions found in the 27 patients, 72 lesions were liver metastases and 46 lesions were benign liver lesions. The sensitivity, accuracy, and NPV of 18F-FDG PET/MRI in the diagnosis of liver metastases (85% (61/72), 91% (107/118) and 81% (46/57)) were significantly higher than those of PET/CT (63% (45/72), 76% (90/118) and 63% (45/72); McNemar test, χ2=5.072, all P<0.05). The sensitivity of 18F-FDG PET/MRI (63% (17/27)) for lesions with length≤1 cm was significantly higher than that of PET/CT (7% (2/27); McNemar test, P<0.05). Conclusion Compared with 18F-FDG PET/CT, 18F-FDG PET/MRI has higher diagnostic value in colorectal cancer liver metastases, especially for lesion with length≤1 cm. -
表 1 27例结直肠癌肝转移患者18F-FDG PET/CT与PET/MRI 诊断效能的比较 (%)
Table 1. Comparison of diagnostic efficacy between 18F-FDG PET/CT and PET/MRI in colorectal cancer liver meastases based on 27 patients (%)
影像学方法 灵敏度 特异度 准确率 阳性预测值 阴性预测值 PET/CT 81(13/16) 100(11/11) 89(24/27) 100(13/13) 79(11/14) PET/MRI 94(15/16) 100(11/11) 96(26/27) 100(15/15) 92(11/12) 检验值 − − − χ2=0.000 χ2=0.851 P值 0.500 1.000 0.500 1.000 0.598 注:FDG为氟脱氧葡萄糖;PET为正电子发射断层显像术;CT为计算机体层摄影术;MRI为磁共振成像;−为McNemar检验,无检验值 表 2 18F-FDG PET/CT与PET/MRI在27例结直肠癌患者肝 转移病灶(72个)中诊断效能的比较 (%)
Table 2. Comparison of diagnostic efficacy between 18F-FDG PET/CT and PET/MRI in colorectal cancer patients(27) liver meastases lesion(72) (%)
影像学
方法灵敏度 特异度 准确率 阳性
预测值阴性
预测值PET/CT 63(45/72) 98(45/46) 76(90/118) 98(45/46) 63(45/72) PET/MRI 85(61/72) 100(46/46) 91(107/118) 100(61/61) 81(46/57) 检验值 − − − χ2=1.339 χ2=5.072 P值 <0.001 1.000 <0.001 0.430 0.024 注:表中,FDG为氟脱氧葡萄糖;PET为正电子发射断层显像术;CT为计算机体层摄影术;MRI为磁共振成像;−为McNemar检验,无检验值 -
[1] Mao YT, Chen B, Wang HF, et al. Diagnostic performance of magnetic resonance imaging for colorectal liver metastasis: a systematic review and meta-analysis[J/OL]. Sci Rep, 2020, 10(1): 1969[2020-03-07]. https://www.nature.com/articles/s41598-020-58855-1. DOI: 10.1038/s41598-020-58855-1. [2] Chakedis J, Squires MH, Beal EW, et al. Update on current problems in colorectal liver metastasis[J]. Curr Probl Surg, 2017, 54(11): 554−602. DOI: 10.1067/j.cpsurg.2017.10.002. [3] Jones RP, Kokudo N, Folprecht G, et al. Colorectal liver metastases: a critical review of state of the art[J]. Liver Cancer, 2016, 6(1): 66−71. DOI: 10.1159/000449348. [4] Engstrand J, Nilsson H, Strömberg C, et al. Colorectal cancer liver metastases — a population-based study on incidence, management and survival[J/OL]. BMC Cancer, 2018, 18(1): 78[2020-03-07]. https://bmccancer.biomedcentral.com/articles/10.1186/s12885-017-3925-x. DOI: 10.1186/s12885-017-3925-x. [5] Gurusamy K, Corrigan N, Croft J, et al. Liver resection surgery versus thermal ablation for colorectal liver metastases (LAVA): study protocol for a randomised controlled trial[J/OL]. Trials, 2018, 19(1): 105[2020-03-07]. https://trialsjournal.biomedcentral.com/articles/10.1186/s13063-018-2499-5. DOI: 10.1186/s13063-018-2499-5. [6] Okuno M, Hatano E, Nishino H, et al. Does response rate of chemotherapy with molecular target agents correlate with the conversion rate and survival in patients with unresectable colorectal liver metastases?: a systematic review[J]. Eur J Surg Oncol, 2017, 43(6): 1003−1012. DOI: 10.1016/j.ejso.2016.08.019. [7] Beiderwellen K, Gomez B, Buchbender C, et al. Depiction and characterization of liver lesions in whole body 18F-FDG PET/MRI[J]. Eur J Radiol, 2013, 82(11): e669−e675. DOI: 10.1016/j.ejrad.2013.07.027. [8] Kirchner J, Sawicki LM, Deuschl C, et al. 18F-FDG PET/MR imaging in patients with suspected liver lesions: value of liver-specific contrast agent Gadobenate dimeglumine[J/OL]. PLoS One, 2017, 12(7): e0180349[2020-03-07]. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0180349. DOI: 10.1371/journal.pone.0180349. [9] Lee DH, Lee JM, Hur BY, et al. Colorectal cancer liver metastases: diagnostic performance and prognostic value of PET/MR imaging[J]. Radiology, 2016, 280(3): 782−792. DOI: 10.1148/radiol.2016151975. [10] Lee KH, Lee JM, Park JH, et al. MR imaging in patients with suspected liver metastases: value of liver-specific contrast agent gadoxetic acid[J]. Korean J Radiol, 2013, 14(6): 894−904. DOI: 10.3348/kjr.2013.14.6.894. [11] 谭继初, 曾鹏程, 陶辉, 等. 结直肠癌肝转移瘤的CT成像特点及规律分析[J]. 广州医药, 2019, 50(1): 64−67. DOI: 10.3969/j.issn.1000-8535.2019.01.016.
Tan JC, Zeng PC, Tao H, et al. CT imaging features and regularity analysis of colorectal cancer liver metastases[J]. Guangzhou Med J, 2019, 50(1): 64−67. DOI: 10.3969/j.issn.1000-8535.2019.01.016.[12] Morsing A, Hildebrandt MG, Vilstrup MH, et al. Hybrid PET/MRI in major cancers: a scoping review[J]. Eur J Nucl Med Mol Imaging, 2019, 46(10): 2138−2151. DOI: 10.1007/s00259-019-04402-8. [13] Ishii S, Shimao D, Hara T, et al. Comparison of integrated whole-body PET/MR and PET/CT: is PET/MR alternative to PET/CT in routine clinical oncology?[J]. Ann Nucl Med, 2016, 30(3): 225−233. DOI: 10.1007/s12149-015-1050-y. [14] Maffione AM, Lopci E, Bluemel C, et al. Diagnostic accuracy and impact on management of 18F-FDG PET and PET/CT in colorectal liver metastasis: a meta-analysis and systematic review[J]. Eur J Nucl Med Mol Imaging, 2015, 42(1): 152−163. DOI: 10.1007/s00259-014-2930-4. [15] Brendle C, Schwenzer NF, Rempp H, et al. Assessment of metastatic colorectal cancer with hybrid imaging: comparison of reading performance using different combinations of anatomical and functional imaging techniques in PET/MRI and PET/CT in a short case series[J]. Eur J Nucl Med Mol Imaging, 2016, 43(1): 123−132. DOI: 10.1007/s00259-015-3137-z. [16] Berger KL, Nicholson SA, Dehdashti F, et al. FDG PET evaluation of mucinous neoplasms: correlation of FDG uptake with histopathologic features[J]. AJR Am J Roentgenol, 2000, 174(4): 1005−1008. DOI: 10.2214/ajr.174.4.1741005. [17] Nasu K, Kuroki Y, Minami M. Diffusion-weighted imaging findings of mucinous carcinoma arising in the ano-rectal region: comparison of apparent diffusion coefficient with that of tubular adenocarcinoma[J]. Jpn J Radiol, 2012, 30(2): 120−127. DOI: 10.1007/s11604-011-0023-x. [18] Donati OF, Hany TF, Reiner CS, et al. Value of retrospective fusion of PET and MR images in detection of hepatic metastases: comparison with 18F-FDG PET/CT and Gd-EOB-DTPA-enhanced MRI[J]. J Nucl Med, 2010, 51(5): 692−699. DOI: 10.2967/jnumed.109.068510. [19] Beiderwellen K, Geraldo L, Ruhlmann V, et al. Accuracy of [18F]FDG PET/MRI for the detection of liver metastases[J/OL]. PLoS One, 2015, 10(9): e0137285[2020-03-07]. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0137285. DOI: 10.1371/journal.pone.0137285. [20] Hong SB, Choi SH, Kim KW, et al. Diagnostic performance of [18F]FDG-PET/MRI for liver metastasis in patients with primary malignancy: a systematic review and meta-analysis[J]. Eur Radiol, 2019, 29(7): 3553−3563. DOI: 10.1007/s00330-018-5909-x. [21] 张占文, 吕清湖, 陈斐妮, 等. 18氟-脱氧葡萄糖正电子发射计算机断层成像联合同机增强CT对结直肠癌肝转移的诊断价值[J]. 中华胃肠外科杂志, 2015, 18(3): 238−242. DOI: 10.3760/cma.j.issn.1671-0274.2015.03.009.
Zhang ZW, Lyu QH, Chen FN, et al. Diagnostic value of 18F-fluorodexyglucose positron emission tomography combined with contrast enhanced computed tomography in colorectal cancer liver metastasis[J]. Chin J Gastrointest Surg, 2015, 18(3): 238−242. DOI: 10.3760/cma.j.issn.1671-0274.2015.03.009.[22] 梁家绪, 周依, 贺祥. Gd-EOB-DTPA MRI增强和Gd-EOB-DTPA MRI增强联合DWI对结直肠癌肝转移诊断价值比较的meta分析[J]. 河南医学研究, 2019, 28(22): 4036−4041. DOI: 10.3969/j.issn.1004-437X.2019.22.002.
Liang JX, Zhou Y, He X. Comparison of Gd-EOB-DTPA-enhanced MRI combined with DWI and Gd-EOB-DTPA-enhanced MRI along in detection of colorectal cancer liver metastases: a meta analysis[J]. Henan Med Research, 2019, 28(22): 4036−4041. DOI: 10.3969/j.issn.1004-437X.2019.22.002.[23] 毛武剑, 石洪成. 18F-FDG PET/CT在结直肠癌肝转移诊治中的应用价值[J]. 中华核医学与分子影像杂志, 2019, 39(9): 560−563. DOI: 10.3760/cma.j.issn.2095-2848.2019.09.014.
Mao WJ, Shi HC. Value of 18F-FDG PET/CT in the diagnosis and treatment of colorectal cancer liver metastases[J]. Chin J Nucl Med Mol Imaging, 2019, 39(9): 560−563. DOI: 10.3760/cma.j.issn.2095-2848.2019.09.014. -






 下载:
下载: