-
恶性黑色素瘤(malignant melanoma,MM)是一种少见的、恶性程度极高的肿瘤,其发病率仅占皮肤恶性肿瘤的3%,但病死率却高达65%[1]。MM在西方人群中发病率较高,在中国、韩国、日本等亚洲国家较少见,但近40年来,亚洲人的发病率也呈稳定增长趋势[2]。约1/3的患者会出现局部复发或转移的风险[3],美国癌症联合委员会(AJCC)黑色素瘤分期数据库的统计结果显示,Ⅳ期患者的5年生存率仅为15%~20%[4]。因此,早期预测疾病的转归并采取积极有效的治疗迫在眉睫。
全身18F-FDG PET/CT是评估肿瘤葡萄糖代谢和增殖最常用的功能成像方法。转移性MM可能累及身体任何远离原发病变的器官,因此局部成像不能对疾病进行全方位地评估。已有研究结果证实,18F-FDG PET/CT在检测黑色素瘤局部复发和隐匿性远处转移、预后和对治疗的影响等方面的诊断准确率均优于CT [5-7]。
SUV已成为评估患者葡萄糖代谢水平的常用指标。大多数肿瘤的18F-FDG PET/CT研究都使用SUVmax单一指标评估肿瘤的代谢情况[8-9]。然而,SUVmax仅代表肿瘤最活跃的部分,不能反映肿瘤的整体代谢情况。Biehl等[10]和Larson等[11]已提出采用肿瘤代谢体积(metabolic tumour volume,MTV)和病灶糖酵解总量(total lesion glycolysis,TLG)克服SUVmax的局限性。目前,全身MTV和全身TLG对转移性MM患者预后的研究报道较少。本研究旨在评估基线18F-FDG PET/CT的代谢参数在预测转移性MM患者预后中的价值。
-
收集2011年8月至2018年12月经南京大学医学院附属鼓楼医院病理确诊的47例转移性MM患者的临床资料,其中男性20例、女性27例,中位年龄59(23~86)岁。对所有患者的临床数据,包括性别、年龄、血清乳酸脱氢酶(lactate dehydrogenase,LDH)水平、原发灶部位、转移灶部位、淋巴结转移情况、美国癌症联合委员会(AJCC)第8版[4]TNM分期及M分期和治疗方式等进行回顾性研究。纳入标准:经病理确诊的MM患者。排除标准:(1)治疗后失访;(2)临床资料不全;(3)无可测量病灶;(4)未进行任何治疗。
-
对所有患者行化疗、免疫或靶向治疗。单纯化疗者15例,化疗方案为安罗替尼+替莫唑胺+顺铂或替莫唑胺+顺铂;单纯免疫治疗者13例,其中大剂量干扰素治疗12例、帕博利珠单抗治疗1例;单纯靶向治疗者1例,治疗方案为达拉非尼口服治疗;联合治疗者18例,治疗方案为化疗+靶向或免疫治疗。在随访期间,通过影像学检查判断疾病是否进展,并对可疑病变进行活检。
-
随访时间为0.5~53.6个月,中位随访时间为15个月。随访期间,17例患者病死,其中2例(11.8%)患者在随访开始6个月内病死(1例0.5个月后病死,另1例0.7个月后病死);其他患者的随访时间均超过6个月。选择黑色素瘤特异性生存期(melanoma-specific survival,MSS)和无进展生存期(progression-free survival,PFS)作为随访的终点。患者的随访期定义为从18F-FDG PET/CT成像到最后1次临床回顾的时间;MSS定义为从18F-FDG PET/CT成像到病死的时间;PFS定义为从18F-FDG PET/CT成像到疾病进展或病死的时间。
-
所有患者检查前均禁食6 h以上,注射药物前空腹血糖≤10.0 mmol/L,按5.18×106 Bq/kg静脉注射18F-FDG(江原安迪科公司生产,放射化学纯度>95%)。1 h后开始采集全身图像,扫描仪为荷兰Philips公司生产的 GEMINI GXL PET/CT仪。先行CT定位扫描,扫描参数:管电压120 kV、管电流120 mA、层厚5.0 mm、间隔5.0 mm。然后行PET扫描,采用三维采集模式,脑部扫描范围自颅顶至下颌骨平面,采集时间为4 min/床位;身体扫描范围自颅底至股骨中下段,共8~9个床位(双上肢立正姿势放置于大腿两侧或平行抱于胸前),双下肢扫描范围自股骨中下段至足底,共7~8个床位,采集时间为1.2 min/床位。应用CT数据进行衰减校正,迭代法重建最终获得横断面、矢状面、冠状面的CT、PET及PET/CT融合图像。所有患者均于检查前签署了知情同意书。本研究符合《赫尔辛基宣言》的原则。
-
PET/CT图像由2位有PET/CT诊断经验的主治及以上职称的核医学医师独立阅片,意见不一致时协商取得一致。在MEDEX工作站对病灶进行勾画,以SUV>40% SUVmax的体素边界作为临界值[12],勾画出每个病灶的MTV,每个病灶的MTV之和为全身MTV。TLG为ROI内的平均标准化摄取值(SUVmean)与MTV的乘积,勾画后由系统自动得出结果,每个病灶的TLG之和为全身TLG。利用ROC曲线进行分析,计算SUVmax、全身MTV、全身TLG的最佳临界值。
-
在MSS和PFS生存分析中分别以SUVmax、全身MTV和全身TLG的最佳临界值为界,将患者分为6组:SUVmax≤临界值组和>临界值组、全身MTV≤临界值组和>临界值组、全身TLG≤临界值组和>临界值组。
-
采用SPSS 20.0软件对数据进行统计学分析。定性资料采用率表示。采用Kaplan-Meier法及Log-rank检验预测2组间MSS和PFS的差异。采用单因素分析法评估PET参数和临床变量的预后意义。采用Cox比例风险模型分析PET参数是否为MSS和PFS的独立预后危险因素。P<0.05为差异有统计学意义。
-
47例MM患者中,13例(27.7%)的血清LDH水平≥245 U/L(正常范围109~245 U/L);39例(83.0%)的肿瘤原发部位为四肢,7例(14.9%)的肿瘤原发部位非四肢,1例(2.1%)的肿瘤原发部位不明;40例(85.1%)伴有淋巴结转移。M分期结果:M0期20例(42.6%)、M1期27例(57.4%)(M1a期12例、M1c期13例、M1d期2例)。TNM分期结果:Ⅲ期20例(42.6%)、Ⅳ期27例(57.4%)。随访结果:22例(46.8%)病情进展、17例(36.2%)病死。
-
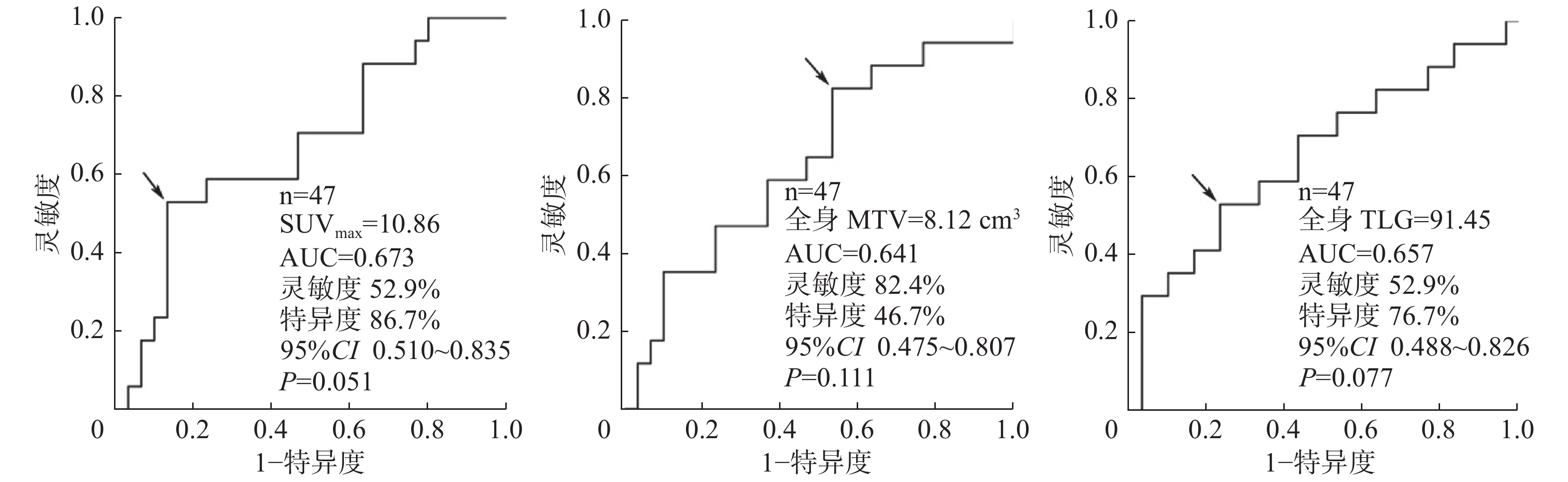
ROC曲线分析结果:SUVmax、全身MTV和全身TLG的最佳临界值分别为10.86、8.12 cm3和91.45(图1)。高于临界值的SUVmax、全身MTV和全身TLG的患者疾病进展率和病死率均上升,分别为92%、90%、94%和69%、47%、56%。7例病死患者的SUVmax、全身MTV和全身TLG均高于临界值。
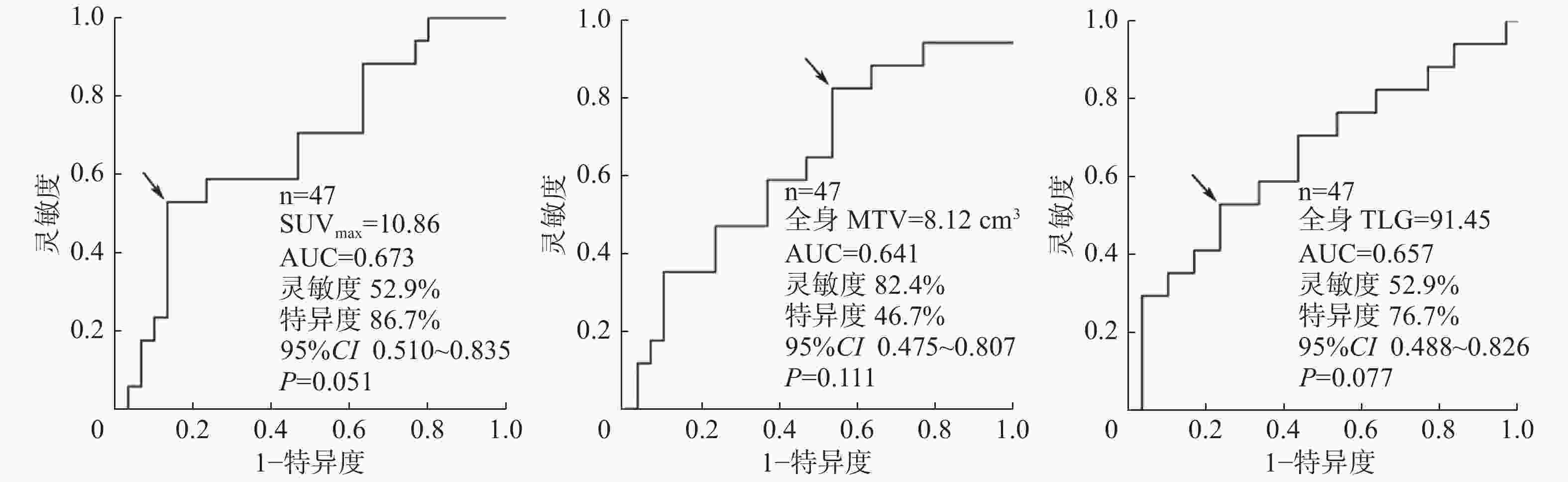
图 1 预测转移性恶性黑色素瘤PET/CT参数最佳临界值的ROC曲线
Figure 1. Receiver operating characteristic curve analysis showed the PET parameters optimal threshold of metastatic malignant melanoma
根据ROC曲线获得的PET参数的最佳临界值将患者重新分组,分别生成Kaplan-Meier生存曲线。由图2可见,通过Log-rank检验,全身MTV和全身TLG以最佳临界值为界的2组患者PFS的差异均有统计学意义(χ2=5.04、5.02,均P<0.05);SUVmax和全身TLG以最佳临界值为界的2组患者MSS的差异均有统计学意义(χ2=10.22、4.38,均P<0.05)。由表1可知,SUVmax>10.86、全身MTV>8.12 cm3、全身TLG>91.45组患者的中位生存时间、1年和2年生存率均低于SUVmax≤10.86、全身MTV≤8.12 cm3、全身TLG≤91.45组。
项目 SUVmax 全身MTV 全身TLG ≤10.86 >10.86 ≤8.12 cm3 >8.12 cm3 ≤91.45 >91.45 中位生存时间(个月) 39.0 11.0 15.0 7.3 36.0 12.2 1年生存率(%) 55.9 38.5 58.8 47.6 54.8 29.2 2年生存率(%) 29.4 15.4 41.2 20.0 32.3 8.3 注:表中,FDG:氟脱氧葡萄糖;PET:正电子发射断层显像术;CT:计算机体层摄影术;SUVmax:最大标准化摄取值;MTV:肿瘤代谢体积;TLG:病灶糖酵解总量 表 1 18F-FDG PET/CT参数临界值评估47例转移性恶性黑色素瘤患者的生存时间和生存率
Table 1. Survival time and survival rate evaluated by 18F-FDG PET/CT parameter threshold in 47 patients with metastatic malignant melanoma
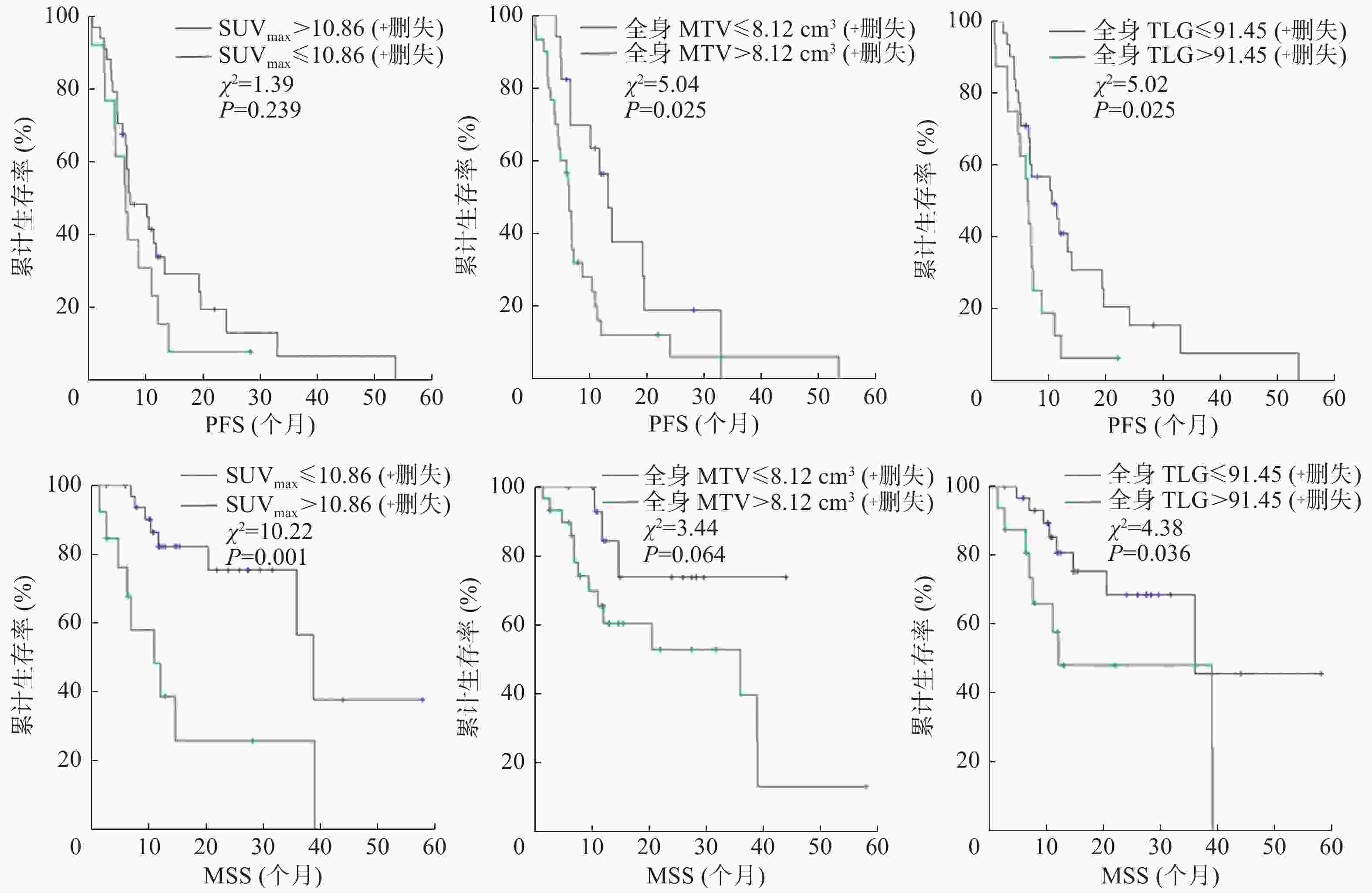
图 2 47例转移性恶性黑色素瘤患者PFS、MSS的Kaplan-Meier生存曲线
Figure 2. Kaplan-Meier survival curves of progression-free survival and melanoma-specific survival in 47 patients with metastatic malignant melanoma
应用临床变量(性别、年龄、LDH水平、原发灶部位、转移灶部位、淋巴结转移、M分期、TNM分期、治疗方式)和PET参数对PFS和MSS进行单因素分析,由表2可知,MSS的预后危险因素包括LDH≥245 U/L、淋巴结转移、SUVmax>10.86和全身TLG>91.45;PFS的预后危险因素包括M1期、全身MTV>8.12 cm3和全身TLG>91.45。
变量 例数 黑色素瘤特异性生存期 无进展生存期 危险比 95%CI P值 危险比 95%CI P值 性别 男 20 0.46 0.17~1.27 0.140 0.59 0.31~1.15 0.120 女 27 年龄(岁) ≤60 27 0.82 0.28~2.45 0.720 0.97 0.50~1.86 0.920 >60 20 LDH(U/L) <245 34 2.84 0.96~8.34 0.048 1.47 0.71~3.05 0.300 ≥245 13 原发灶部位 四肢/非四肢 46 0.81 0.20~3.23 0.770 1.40 0.73~2.69 0.300 原发灶不明 1 转移灶部位 单纯淋巴结/脏器转移 21 1.12 0.35~6.88 0.560 1.32 0.92~1.89 0.130 多发转移 26 淋巴结转移 否 7 1.56 0.97~9.85 0.045 1.78 0.68~4.63 0.240 是 40 M分期 0期 20 1.32 0.93~1.87 0.120 1.27 1.01~1.59 0.042 1期 27 TNM分期 Ⅲ期 20 1.38 0.50~3.77 0.530 1.60 0.83~3.01 0.160 Ⅳ期 27 治疗方式 化疗、靶向治疗 16 0.77 0.52~1.13 0.180 1.24 0.94~1.62 0.120 免疫治疗、联合治疗 31 SUVmax ≤10.86 34 4.22 1.62~11.05 0.001 1.51 0.76~3.00 0.239 >10.86 13 全身MTV(cm3) ≤8.12 17 3.08 0.87~10.80 0.064 2.17 1.08~4.35 0.025 >8.12 30 全身TLG ≤91.45 31 2.69 1.02~7.07 0.036 2.11 1.08~4.12 0.025 >91.45 16 注:表中,CI:可变区间;LDH:乳酸脱氢酶;TNM:肿瘤、淋巴结、转移;SUVmax:最大标准化摄取值;MTV:肿瘤代谢体积;TLG:病灶糖酵解总量 表 2 47例转移性恶性黑色素瘤患者预后危险因素的单因素分析
Table 2. Univariate analysis of prognostic risk factors in 47 patients with metastatic malignant melanoma
将单因素分析有意义的变量进行多因素Cox比例风险模型分析,评估转移性MM的独立预后危险因素,由表3可知,MSS的独立预后危险因素是SUVmax>10.86(危险比 4.11,95%CI:1.40~11.78,P=0.008)。
变量 例数 黑色素瘤特异性生存期 无进展生存期 危险比 95%CI P值 危险比 95%CI P值 SUVmax ≤10.86 34 4.11 1.40~11.78 0.008 − − − >10.86 13 全身MTV(cm3) ≤8.12 17 − − − 1.80 0.80~3.00 0.145 >8.12 30 全身TLG ≤91.45 31 2.14 0.75~6.15 0.157 1.79 0.80~3.69 0.116 >91.45 16 注:表中,CI:可变区间;SUVmax:最大标准化摄取值;MTV:肿瘤代谢体积;TLG:病灶糖酵解总量;−:无此项数据 表 3 47例转移性恶性黑色素瘤患者预后危险因素的多因素分析
Table 3. Multivariate analysis of prognostic risk factors in 47 patients with metastatic malignant melanoma
-
黑色素瘤是起源于皮肤黏膜及色素膜黑色素细胞的恶性肿瘤。目前,18F-FDG PET/CT在MM中主要用于疾病的诊断、分期和疗效评价[13-14],对于疾病预后评估的研究较少,且研究方向也局限在原发灶或局部病变[15-16]。由于MM具有侵袭程度高、极易转移和复发的特点,因此,对MM的预后及远期生存的准确评估迫在眉睫。在本研究中,我们应用基线PET参数对MM的预后进行评估。
本研究中,临床变量(性别、年龄、LDH水平、原发灶部位、转移灶部位、淋巴结转移、M分期、TNM分期、治疗方式)的单因素分析表明,LDH水平、淋巴结转移是MSS的预后因素,M1分期是PFS的预后因素。Agarwala等[17]对1085名受试者的大型前瞻性研究结果表明,LDH是转移性MM的临床预后因素,与我们的研究结果一致。2017年美国癌症联合委员会(AJCC)第8版[4]的MM分期显示,M分期和LDH水平是MM的预后危险因素,并将LDH水平升高视为M1a(1)期。
本研究结果表明,SUVmax>10.86的MM患者的不良预后及相关病死的风险更高。在临床诊疗中,SUVmax代表肿瘤侵袭性的程度,SUVmax升高与肿瘤细胞的增殖有关。本研究的单因素分析结果表明,SUVmax是转移性MM患者MSS的独立预后危险因素,但不是PFS的独立预后危险因素。Son等[16]的研究结果表明,SUVmax是原发性MM 特异性生存期的预测因子,但调整了溃疡这个因素后,SUVmax不再能预测MM的总生存期。Schmitt等[18]研究了24例接受抗鼠科肉瘤病毒癌基因同源物B1(BRAF)抑制剂和丝裂原活化蛋白激酶的激酶(MEK)1 抑制剂靶向治疗的MM患者,结果发现,SUVmax能够很好地预测PFS,但并不能预测总生存期,这与我们的研究结果不一致,分析其原因,可能与Schmitt等[18]的研究病例数较少(24例),随访时间较短(最长随访时间600 d)有关。
本研究PET参数的单因素分析结果显示,全身MTV和全身TLG能够反映疾病的复发和进展,虽然多因素分析结果提示全身MTV和全身TLG不是评估MM预后的最佳预测因素,但其仍是评估MM预后的重要预后因素,全身MTV和全身TLG越高,MM患者疾病进展或病死的概率越大,这与Son等[16]的研究结果相似,其团队发现全身MTV和全身TLG是原发性MM 特异性生存期的重要预测因子。Seban等[12]的一项关于抗程序性死亡受体1(PD-1)免疫治疗PET参数对MM预后的研究结果表明,全身MTV和全身TLG是不良预后的最佳预测因素,这与本研究结果不完全一致,分析其原因:(1)Seban等[12]的研究中入组患者的治疗方法为一致的抗程序性死亡受体1(PD-1)免疫治疗,而本研究的治疗方式有多种,到目前为止,不同的治疗方式是否影响全身MTV和全身TLG对预后的评估尚不明确,需要进一步的研究;(2)本研究的分组方式也可能是影响因素之一,Seban等[12]是以每个参数的中位数作为临界值,对其进行再次分组,而本研究是通过ROC曲线选择参数的临界值进行分组,PET参数临界值的不同会引起分组差异,其对预测效能的影响有待进一步研究;(3)由于本研究是通过MEDEX工作站进行MTV、TLG代谢参数的测量,其准确性可能较国际认可软件有一定差距,这有待进一步验证。
本研究仍有一些局限性。本研究是单中心试验研究,患者数相对较少而且是回顾性的。因此,仍然需要进行前瞻性、大规模、多中心的研究。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 来瑞鹤负责数据的获取与分析、论文的撰写;蒋冲负责研究过程的实施、数据的分析;滕月负责数据的采集与统计分析;孙一文负责论文的审阅与修订;李爱梅负责研究命题的提出、论文的修订。
基线18F-FDG PET/CT在转移性恶性黑色素瘤患者预后评估中的价值
Prognostic value of pretreatment 18F-FDG PET/CT in patients with metastatic malignant melanoma
-
摘要:
目的 探讨治疗前18F-氟脱氧葡萄糖(FDG)PET/CT代谢参数在转移性恶性黑色素瘤(MM)患者预后评估中的价值。 方法 回顾性分析2011年8月至2018年12月在南京大学医学院附属鼓楼医院确诊为转移性MM的47例患者的临床资料,其中男性20例、女性27例,中位年龄59(23~86)岁。对所有患者行化疗、免疫或靶向治疗。随访时间为0.5~53.6个月。黑色素瘤特异性生存期(MSS)和无进展生存期(PFS)分别定义为从18F-FDG PET成像到患者病死的时间和疾病进展或病死的时间。所有患者在治疗前均行18F-FDG PET/CT检查,测量最大标准化摄取值(SUVmax),并以SUV>40% SUVmax的体素边界作为临界值,分别测量并计算全身肿瘤代谢体积(MTV)和全身病灶糖酵解总量(TLG)。采用受试者工作特征(ROC)曲线分析得出PET参数的最佳临界值,并以SUVmax、全身MTV和全身TLG临界值为界分别将患者分为2组,共6组。采用Kaplan-Meier法及Log-rank检验预测2组间MSS和PFS的差异。采用单因素分析法评估PET参数和临床变量的预后意义。采用Cox比例风险模型多因素分析PET参数是否为MSS和PFS的独立预后危险因素。 结果 SUVmax、全身MTV和全身TLG的最佳临界值分别为10.86、8.12 cm3和91.45。全身MTV和全身TLG以临界值为界的2组患者PFS的差异均有统计学意义(χ2=5.04、5.02,均P<0.05);SUVmax和全身TLG以临界值为界的2组患者MSS的差异均有统计学意义(χ2=10.22、4.38,均P<0.05)。单因素分析结果表明,血清乳酸脱氢酶水平≥245 U/L、淋巴结转移、SUVmax>10.86和全身TLG>91.45是MSS的预后危险因素;M1期、全身MTV>8.12 cm3和全身TLG>91.45是PFS的预后危险因素。多因素分析结果表明,SUVmax>10.86是MSS的独立预后危险因素。 结论 18F-FDG PET/CT代谢参数SUVmax是转移性MM患者病死的最佳预测因素,而全身MTV和全身TLG对转移性MM患者的预后具有一定的预测价值。 -
关键词:
- 黑色素瘤 /
- 氟脱氧葡萄糖F18 /
- 正电子发射断层显像术 /
- 体层摄影术,X线计算机 /
- 肿瘤代谢体积 /
- 病灶糖酵解总量
Abstract:Objective To demonstrate whether 18F-FDG PET/CT metabolic parameters could predict prognosis in patients with metastatic maligant melanoma (MM). Methods A retrospective analysis was conducted on a dataset composed of 47 patients who were newly diagnosed with metastatic MM and currently undergoing pretreatment 18F-FDG PET/CT in the Affiliated Hospital of Nanjing University Medical School, Nanjing Drum Tower Hospital from August 2011 to December 2018. Of 47 patients, 20 were male, 27 were female and median age 59 (23−86) years. All patients were treated with chemotherapy, immunotherapy or targeted therapy and follow-up time was 0.5 to 53.6 months. Melanoma-specific survival (MSS) and progression-free survival (PFS) were defined as the time from 18F-FDG PET/CT imaging to the patient's death and the patient's death or progression of the disease, separately. All patients underwent 18F-FDG PET/CT imaging before treatment.The maximum standardized uptake value (SUVmax) was measured. Whole-body metabolic tumor volume (MTV) and whole-body total lesion glycolysis (TLG) were measured automatically and SUV>40% SUVmax voxel boundary was used as threshold. The optimal thresholds of PET parameters were obtained using the receiver operating characteristic (ROC) curve, and the patients were divided into two groups separately according to the optimal thresholds of SUVmax, whole-body MTV and whole-body TLG and six groups were obtained. The difference of MSS and PFS between the two groups were predicted by Kaplan-Meier method and Log-rank test. Univariate analysis was conducted to evaluate the prognostic value of PET parameters and clinical variables. The Cox proportional risk model multivariate analysis was used to determine whether the PET parameters can act as independent prognostic risk factors for MSS and PFS. Results The cut-off values for SUVmax, whole-body MTV, and whole-body TLG were 10.86, 8.12 cm3, and 91.45, respectively, as shown in the ROC curve analysis. The PFS was significantly different in two groups divided by optimal thresholds of whole-body MTV or whole-body TLG, separately (χ2=5.04, 5.02; both P<0.05). Similarly, the MSS was significantly different in two groups divided by optimal thresholds of SUVmax or whole-body TLG, separately (χ2=10.22, 4.38; both P<0.05). The univariate analysis results were as follows: the serum lactate dehydrogenase level≥245 U/L, lymphatic metastasis, SUVmax>10.86 and whole-body TLG>91.45, which were associated with predictors of MSS. M l stage, whole-body MTV>8.12 cm3 and whole-body TLG>91.45, which were associated with PFS. The multivariate analysis results showed that SUVmax>10.86, proving its potential as an independent prognostic risk factor for MSS. Conclusion The 18F-FDG PET/CT metabolic parameter SUVmax was the best predictive marker in metastatic MM patients, whole-body MTV and whole-body TLG helped for the prognosis of metastatic MM patients. -
表 1 18F-FDG PET/CT参数临界值评估47例转移性恶性黑色素瘤患者的生存时间和生存率
Table 1. Survival time and survival rate evaluated by 18F-FDG PET/CT parameter threshold in 47 patients with metastatic malignant melanoma
项目 SUVmax 全身MTV 全身TLG ≤10.86 >10.86 ≤8.12 cm3 >8.12 cm3 ≤91.45 >91.45 中位生存时间(个月) 39.0 11.0 15.0 7.3 36.0 12.2 1年生存率(%) 55.9 38.5 58.8 47.6 54.8 29.2 2年生存率(%) 29.4 15.4 41.2 20.0 32.3 8.3 注:表中,FDG:氟脱氧葡萄糖;PET:正电子发射断层显像术;CT:计算机体层摄影术;SUVmax:最大标准化摄取值;MTV:肿瘤代谢体积;TLG:病灶糖酵解总量 表 2 47例转移性恶性黑色素瘤患者预后危险因素的单因素分析
Table 2. Univariate analysis of prognostic risk factors in 47 patients with metastatic malignant melanoma
变量 例数 黑色素瘤特异性生存期 无进展生存期 危险比 95%CI P值 危险比 95%CI P值 性别 男 20 0.46 0.17~1.27 0.140 0.59 0.31~1.15 0.120 女 27 年龄(岁) ≤60 27 0.82 0.28~2.45 0.720 0.97 0.50~1.86 0.920 >60 20 LDH(U/L) <245 34 2.84 0.96~8.34 0.048 1.47 0.71~3.05 0.300 ≥245 13 原发灶部位 四肢/非四肢 46 0.81 0.20~3.23 0.770 1.40 0.73~2.69 0.300 原发灶不明 1 转移灶部位 单纯淋巴结/脏器转移 21 1.12 0.35~6.88 0.560 1.32 0.92~1.89 0.130 多发转移 26 淋巴结转移 否 7 1.56 0.97~9.85 0.045 1.78 0.68~4.63 0.240 是 40 M分期 0期 20 1.32 0.93~1.87 0.120 1.27 1.01~1.59 0.042 1期 27 TNM分期 Ⅲ期 20 1.38 0.50~3.77 0.530 1.60 0.83~3.01 0.160 Ⅳ期 27 治疗方式 化疗、靶向治疗 16 0.77 0.52~1.13 0.180 1.24 0.94~1.62 0.120 免疫治疗、联合治疗 31 SUVmax ≤10.86 34 4.22 1.62~11.05 0.001 1.51 0.76~3.00 0.239 >10.86 13 全身MTV(cm3) ≤8.12 17 3.08 0.87~10.80 0.064 2.17 1.08~4.35 0.025 >8.12 30 全身TLG ≤91.45 31 2.69 1.02~7.07 0.036 2.11 1.08~4.12 0.025 >91.45 16 注:表中,CI:可变区间;LDH:乳酸脱氢酶;TNM:肿瘤、淋巴结、转移;SUVmax:最大标准化摄取值;MTV:肿瘤代谢体积;TLG:病灶糖酵解总量 表 3 47例转移性恶性黑色素瘤患者预后危险因素的多因素分析
Table 3. Multivariate analysis of prognostic risk factors in 47 patients with metastatic malignant melanoma
变量 例数 黑色素瘤特异性生存期 无进展生存期 危险比 95%CI P值 危险比 95%CI P值 SUVmax ≤10.86 34 4.11 1.40~11.78 0.008 − − − >10.86 13 全身MTV(cm3) ≤8.12 17 − − − 1.80 0.80~3.00 0.145 >8.12 30 全身TLG ≤91.45 31 2.14 0.75~6.15 0.157 1.79 0.80~3.69 0.116 >91.45 16 注:表中,CI:可变区间;SUVmax:最大标准化摄取值;MTV:肿瘤代谢体积;TLG:病灶糖酵解总量;−:无此项数据 -
[1] Pavri SN, Clune J, Ariyan S, et al. Malignant melanoma: beyond the basics[J]. Plast Reconstr Surg, 2016, 138(2): 330e−340e. DOI: 10.1097/PRS.0000000000002367. [2] Qiu DM, Marugame T. Comparison of time trends in skin cancer incidence (1973-97) in East Asia, Europe and USA, from Cancer Incidence in Five Continents Vol. Ⅳ−Ⅷ[J]. Jpn J Clin Oncol, 2008, 38(3): 234−236. DOI: 10.1093/jjco/hyn012. [3] Tas F, Erturk K. Recurrence behavior in early-stage cutaneous melanoma: pattern, timing, survival, and influencing factors[J]. Melanoma Res, 2017, 27(2): 134−139. DOI: 10.1097/CMR.0000000000000332. [4] Gershenwald JE, Scolyer RA, Hess KR, et al. Melanoma staging: evidence-based changes in the American Joint Committee on Cancer eighth edition cancer staging manual[J]. CA Cancer J Clin, 2017, 67(6): 472−492. DOI: 10.3322/caac.21409. [5] Rohren EM. PET/computed tomography and patient outcomes in melanoma[J]. PET Clin, 2015, 10(2): 243−254. DOI: 10.1016/j.cpet.2014.12.006. [6] Youland RS, Packard AT, Blanchard MJ, et al. 18F-FDG PET response and clinical outcomes after stereotactic body radiation therapy for metastatic melanoma[J/OL]. Adv Radiat Oncol, 2017, 2(2): 204−210[2019-09-09]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5514257. DOI: 10.1016/j.adro.2017.02.003. [7] Hasebe M, Yoshikawa K, Nishii R, et al. Usefulness of 11C-methionine-PET for predicting the efficacy of carbon ion radiation therapy for head and neck mucosal malignant melanoma[J]. Int J Oral Maxillofac Surg, 2017, 46(10): 1220−1228. DOI: 10.1016/j.ijom.2017.04.019. [8] Malik D, Sood A, Mittal BR, et al. Role of 18F-fluorodeoxyglucose positron emission tomography/computed tomography in restaging and prognos is of recurrent melanoma after curative surgery[J]. World J Nucl Med, 2019, 18(2): 176−182. DOI: 10.4103/wjnm.WJNM_37_18. [9] Jiang C, Zhang X, Jiang M, et al. Assessment of the prognostic capacity of pretreatment, interim, and post-therapy 18F-FDG PET/CT in extranodal natural killer/T-cell lymphoma, nasal type[J]. Ann Nucl Med, 2015, 29(5): 442−451. DOI: 10.1007/s12149-015-0964-8. [10] Biehl KJ, Kong FM, Dehdashti F, et al. 18F-FDG PET definition of gross tumor volume for radiotherapy of non-small cell lung cancer: is a single standardized uptake value threshold approach appropriate?[J]. J Nucl Med, 2006, 47(11): 1808−1812. [11] Larson SM, Erdi Y, Akhurst T, et al. Tumor treatment response based on visual and quantitative changes in global tumor glycolysis using PET-FDG imaging. The visual response score and the change in total lesion glycolysis[J]. Clin Positron Imaging, 1999, 2(3): 159−171. DOI: 10.1016/s1095-0397(99)00016-3. [12] Seban RD, Nemer JS, Marabelle A, et al. Prognostic and theranostic 18F-FDG PET biomarkers for anti-PD1 immunotherapy in metastatic melanoma: association with outcome and transcriptomics[J]. Eur J Nucl Med Mol Imaging, 2019, 46(11): 2298−2310. DOI: 10.1007/s00259-019-04411-7. [13] Ravichandran S, Nath N, Jones DC, et al. The utility of initial staging PET-CT as a baseline scan for surveillance imaging in stage Ⅱ and Ⅲ melanoma[J]. Surg Oncol, 2020, 35: 533−539. DOI: 10.1016/j.suronc.2020.10.018. [14] Gellén E, Sántha O, Janka E, et al. Diagnostic accuracy of 18F-FDG-PET/CT in early and late stages of high-risk cutaneous malignant melanoma[J]. J Eur Acad Dermatol Venereol, 2015, 29(10): 1938−1944. DOI: 10.1111/jdv.13084. [15] Kang S, Ahn BC, Hong CM, et al. Can 18F-FDG PET/CT predict recurrence in patients with cutaneous malignant melanoma?[J]. Nuklearmedizin, 2011, 50(3): 116−121. DOI: 10.3413/Nukmed-0356-10-09. [16] Son SH, Kang SM, Jeong SY, et al. Prognostic value of volumetric parameters measured by pretreatment 18F FDG PET/CT in patients with cutaneous malignant melanoma[J]. Clin Nucl Med, 2016, 41(6): e266−e273. DOI: 10.1097/RLU.0000000000001205. [17] Agarwala SS, Keilholz U, Gilles E, et al. LDH correlation with survival in advanced melanoma from two large, randomised trials (Oblimersen GM301 and EORTC 18951)[J]. Eur J Cancer, 2009, 45(10): 1807−1814. DOI: 10.1016/j.ejca.2009.04.016. [18] Schmitt RJ, Kreidler SM, Glueck DH, et al. Correlation between early 18F-FDG PET/CT response to BRAF and MEK inhibition and survival in patients with BRAF-mutant metastatic melanoma[J]. Nucl Med Commun, 2016, 37(2): 122−128. DOI: 10.1097/MNM.0000000000000406. -





 下载:
下载: