-
随着医学科技水平的发展,医学检查方法愈趋多样化、医学仪器设备愈趋高端化,甲状腺结节在健康人群中的检出率也随之升高,达50%~60%[1]。目前细针穿刺活检被认为是术前诊断甲状腺结节良恶性的“金标准”,其诊断灵敏度达83%(65%~98%)、特异度达92%(72%~100%),但仍有15%~20%的患者不能被确诊,在这部分患者中,仅有20%~25%的患者经术后病理结果被诊断为恶性结节,75%~80%的患者接受了不必要的甲状腺手术[2]。另外,目前常用甲状腺球蛋白(thyroglobulin,Tg)水平预测转移情况,但其易受到抗甲状腺球蛋白抗体(thyroglobulin antibody,TgAb)的干扰,且部分术后行131I治疗的DTC患者在治疗中会出现Tg水平较高而全身碘扫描结果显示阴性的情况,这会对临床诊断造成一定的困扰[3-4]。针对这些临床困惑,本研究通过检测血清midkine(MK)和galectine-3(Gal-3)水平来进行补充,以便临床医师更好地制定治疗方案和评估患者的预后。
-
回顾性分析2016年11月至2017年11月于山西医科大学第一医院普外科拟行甲状腺切除术的甲状腺结节患者41例,其中男性11例、女性30例,年龄29~73(48.61±11.59)岁,根据手术病理结果分为恶性组和良性组。同时收集体检中心体检的32名健康者为健康对照组,其中男性15名、女性17名,年龄24~58(46.00±11.18)岁。
41例甲状腺结节患者中,恶性组有23例,男性6例、女性17例,年龄30~73(47.39±11.52)岁,其中甲状腺乳头状癌19例、甲状腺微小乳头状癌4例;良性组有18例,男性5例、女性13例,年龄29~65(46.00±11.18)岁,其中结节性甲状腺肿17例、甲状腺嗜酸性细胞腺瘤1例。纳入标准:甲状腺结节拟行手术切除者。排除标准:良恶性结节并存;合并有桥本甲状腺炎等其他甲状腺疾病;合并有其他部位肿瘤、冠心病、糖尿病、肝肾疾病、感染性疾病。
-
回顾性分析2016年11月至2017年11月于山西医科大学第一医院核医学科行2次131I治疗并于每次治疗5~7 d后行全身碘扫描显像的DTC术后患者51例,其中男性12例、女性39例,年龄25~72(44.41±10.21)岁。纳入标准:首次行131I治疗的甲状腺全切术后的DTC患者,且术后进行了2次131I治疗。排除标准:甲状腺全切术后未行2次131I治疗的DTC患者。
上述2项回顾性研究均符合《赫尔辛基宣言》的原则。
-
51例DTC术后患者行131I治疗前停服左旋甲状腺素钠片(德国默克公司) 3~4周,并低碘饮食2~4周。清甲前告知患者治疗目的、可能的不良反应及注意事项,并签署131I治疗同意书。第1次放射性131I(原子高科股份有限公司,放射化学纯度>95%)治疗时,空腹一次性口服3.7 GBq(对于存在摄取131I转移病灶的患者给予 3.7~7.4 GBq)。治疗后第3天开始口服左旋甲状腺素钠片替代治疗,5~7 d后使用德国Siemens公司的 Symbia SPECT/CT仪行131I全身扫描,配高能通用型平行孔准直器,参数:能峰364 keV、窗宽20%、矩阵256×1024、放大倍速1.0。3~6个月后行第2次 131I治疗,治疗后5~7 d行全身碘扫描。每次131I治疗后对 DTC患者的治疗后效果及转移情况进行评估。
-
由2位副主任及以上职称的核医学医师对全身碘扫描图像进行判读,意见不统一时,经讨论及第3位医师评定后达成一致意见。DTC患者淋巴结及肺转移判断标准:在2次131I治疗期间,经超声、CT、PET/CT等影像学检查结果明确提示有淋巴结、肺等部位转移,和(或)2次131I治疗后在全身碘扫描中有甲状腺外组织(淋巴结、肺等)的131I摄取。密切随访所有 DTC 患者,随访时间为3~6个月,具有患者两次 131I 治疗期间所有的采集数据。
-
甲状腺结节患者和DTC术后患者均于空腹时采集血样,甲状腺结节患者收集术前血样(采集前禁食8 h以上),测定血清MK和Gal-3水平;DTC术后患者收集首次服131I治疗前的血样,并测定血清MK和Gal-3水平。所有血样均以3000×g离心10 min后分离血清。
-
使用德国Siemens公司生产的IMMULITE 2000型全自动化学发光免疫分析仪测定Tg水平(参考范围 1.69~59.90 ng/mL,最高可测值 500.00 ng/mL)。
-
使用上海江莱生物科技有限公司生产的抗体及相关试剂进行检测,检测仪器为美国赛默飞世尔公司的 FC 型全自动酶标仪。检测流程:①将微孔酶标板及待测样品室内放置20 min;②先后往标准品孔、样品孔中加入50 μL不同浓度的标准品和待测样品及100 μL辣根过氧化物酶标记的检测抗体;③封住反应孔,在37℃恒温箱内放置60 min;④从恒温箱内取出后,倒掉液体,用吸水纸拍干,孔内加满350 μL洗涤液,放置1 min后倒掉拍干,重复上述步骤4~5次;⑤每孔加入底物MK、Gal-3各50 μL,37℃避光孵育15 min;⑥每孔加入50 μL终止液,在450 nm 波长下测定光密度值;⑦绘制标准曲线,计算各样品的浓度值。每个样品血清或标准品均重复测定2次。
-
采用SPSSl9.0软件进行统计学分析。符合正态分布的计量资料以
$\bar x $ ±s表示,2组间均值的比较采用两独立样本t检验(方差齐)、3组间均值的比较采用单因素方差分析;血清MK和Gal-3水平间的相关性采用Pearson相关性分析。采用ROC曲线确定MK和Gal-3诊断良恶性甲状腺结节的最佳阈值。以α=0.05为检验水准,P<0.05为差异有统计学意义。 -
根据2次治疗性131I全身扫描结果及期间其他影像学检查结果中是否有淋巴结和(或)肺转移,将51例DTC术后患者分为转移阳性组和转移阴性组,其中转移阳性组8例,包括男性2例、女性6例,年龄25~61(40.41±9.53)岁,具体转移部位见表1;转移阴性组43例,包括男性10例、女性33例,年龄26~72(48.41±10.89)岁。
编号 年龄 性别 转移部位 1 25 女 双侧颈Ⅱ区 2 28 女 左颈Ⅲ区及右颈Ⅵ区 3 33 男 右颈Ⅳ区 4 34 女 右颈Ⅲ、Ⅳ区 5 43 男 双肺多发转移 6 47 女 左颈Ⅵ区及右颈Ⅱ区 7 52 女 右颈Ⅲ、Ⅳ区 8 61 女 双肺多发转移 表 1 分化型甲状腺癌患者术后转移阳性组患者淋巴结和 (或)肺转移部位
Table 1. Locations of lymph node and/or lung metastasis in differentiated thyroid carcinoma positive group
-
甲状腺结节恶性组、良性组和健康对照组3组间的年龄差异无统计学意义(F=0.28,P>0.05)。恶性组的血清MK和Gal-3水平均高于良性组及健康对照组,且差异有统计学意义(F=48.40、5.08,均P<0.05),但良性组与健康对照组间血清MK和Gal-3水平的差异均无统计学意义(t=−0.90、0.63,均P>0.05)(表2)。
组别 MK(pg/mL) Gal-3(ng/mL) 恶性组(n=23) 385.79±198.62 2.97±1.45 良性组(n=18) 280.12±101.24 1.29±0.33 健康对照组(n=32) 270.93±104.18 1.05±0.39 F值 48.40 5.08 P值 <0.01 0.01 注:表中,MK:midkine;Gal-3:galectine-3 表 2 2组甲状腺结节患者和健康对照者的血清学指标比较 (
$\bar x $ Table 2. Comparison of serological indexes among malignant, benign and healthy thyroid nodules (
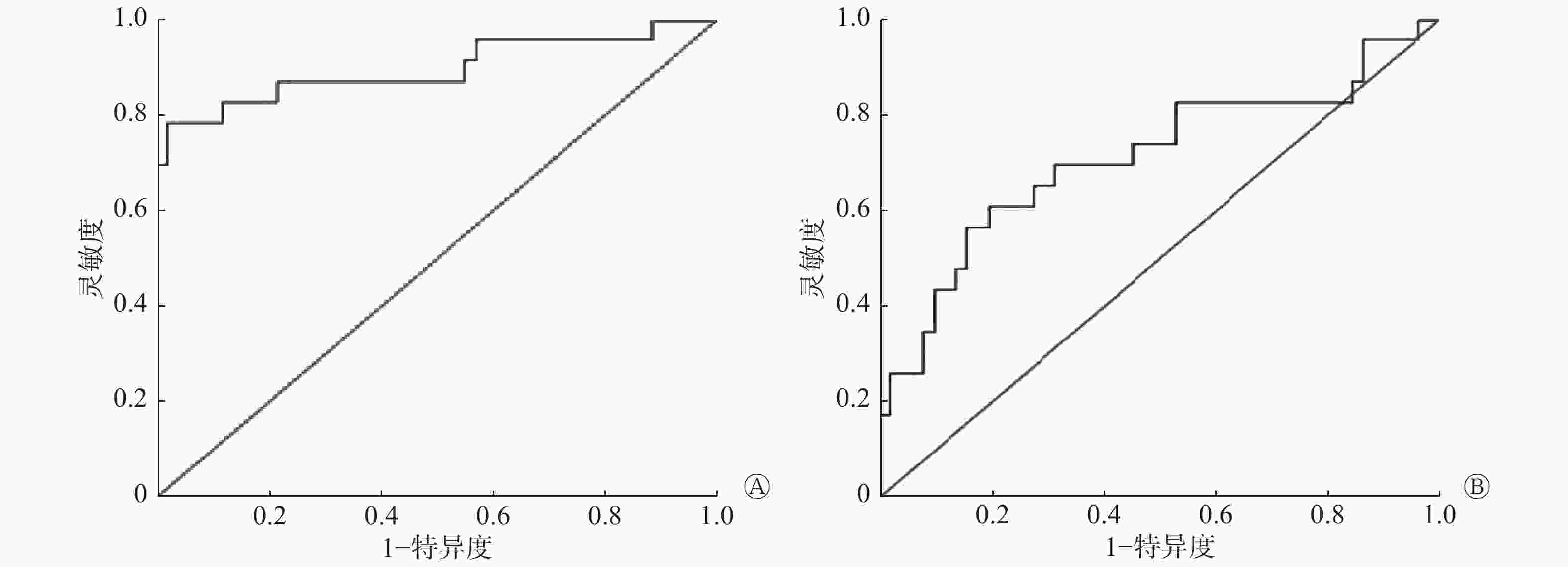
$ \bar x$ ROC曲线分析结果显示,MK的最佳阈值为318.87 pg/mL、AUC为0.91(95%CI: 0.81~0.99)、灵敏度为88.5%、特异度为78.1%;Gal-3的最佳阈值为1.61 ng/mL、AUC为0.72(95%CI: 0.57~0.86)、灵敏度为69.8%、特异度为70.4%(图1)。
-
转移阳性组的血清MK和Gal-3水平较转移阴性组均明显升高,转移阳性组的Tg水平明显高于转移阴性组,且差异均有统计学意义(t=−4.86~−2.89,均P<0.01)(表3)。
组别 MK(pg/mL) Gal-3(ng/mL) Tg(ng/mL) 转移阳性组
(n=8)489.93±124.69 2.82±1.51 176.30±131.94 转移阴性组
(n=43)277.41±115.24 1.74±0.66 15.10±14.38 t值 −4.01 −2.89 −4.86 P值 <0.01 <0.01 <0.01 注:表中,MK:midkine;Gal-3:galectine-3;Tg:甲状腺球蛋白 表 3 分化型甲状腺癌患者术后首次131I治疗前2组的血清 学指标比较(
$\bar x $ Table 3. Comparison of serum indexes between 2 groups in patients after differentiated thyroid carcinoma before the first 131I treatment (
$\bar x $ -
Pearson相关性分析结果显示,甲状腺结节患者术前(r=0.67,P<0.05)及DTC患者术后 (r=0.84,P<0.05)血清MK、Gal-3水平之间均存在显著相关性(图2)。
-
MK是一种相对分子质量较低的蛋白质,属于分泌型肝素结合生长因子[5],在成人体内,MK在特定的组织或器官中低水平表达,但是当机体在遭到肿瘤细胞、炎症因子的浸润以及进行组织细胞修复时,其表达水平会明显增加[2,6]。目前的研究结果显示,MK的生物学作用包括抗细胞凋亡、促进细胞分裂、促进血管生成和肿瘤侵袭转移等[7]。Gal-3是单基因LGALS3编码的蛋白质,位于14号染色体,是一种能识别糖蛋白和糖脂中特异性低聚糖结构的半乳糖结合蛋白,也称血清免疫球蛋白E结合蛋白,其主要在细胞黏附、生长、分化及凋亡、维持细胞内稳态、促进血管生成和肿瘤侵袭转移等方面发挥作用[8]。二者在肿瘤发生发展中的作用不容忽视。
Kuzu等[2]研究发现,在87%的恶性肿瘤中均可发现血清MK水平升高,包括消化系统肿瘤、泌尿系统肿瘤、恶性间皮瘤和乳腺癌等。Meng等[9]研究发现,甲状腺结节恶性组的血清MK水平明显高于良性组和健康对照组,良恶性结节鉴别诊断的最佳阈值为323.12 ng/L,AUC为0.83,诊断灵敏度和特异度分别为75.71%和75.00%。冯光勇和黄伟[10]研究发现,良恶性甲状腺结节鉴别诊断的最佳阈值为330.00 ng/mL,AUC为0.85,诊断灵敏度和特异度分别为81.30%和78.70%。本研究结果显示,恶性组的血清MK水平较良性组及健康对照组高,而良性组与健康对照组间的差异无统计学意义,良恶性结节组间最佳阈值为318.87 pg/mL,并有较高的灵敏度和特异度。Jing等[11]通过对大量文献综述得出,血清MK水平对恶性肿瘤(包括甲状腺癌)的诊断灵敏度和特异度可达78%和83%,总比值比为16.79,这表明血清MK水平对恶性肿瘤的诊断准确性具有较强的可信性。
目前对于DTC术后的患者,常以Tg水平作为判断131I疗效及甲状腺癌复发和转移的重要血清学检测指标,李田军等[12]的研究结果显示,DTC术后、首次131I治疗前的Tg水平升高可以预测肿瘤转移,当Tg水平的阈值为52.75 μg/L时,判断存在肺转移、骨转移或淋巴结转移等远处转移的灵敏度和特异度分别为78.90%和91.70%,但Tg易受TgAb的干扰,会在一定程度上干扰对患者病情的判断。在本研究中,转移阳性组中血清MK、Tg水平较阴性组明显升高,因转移阳性组样本量有限未能进一步筛选诊断阈值。MK具有与Tg相类似的评估转移灶的作用,其机制可能是因肿瘤切除后患者血清MK的水平会明显降低,但如果DTC发生转移或有残存转移灶,其可成为血清MK的另一个来源,导致体内MK水平再次升高[9]。Jee等[13]研究得出相似结果,而Meng等[9]研究发现,在DTC术后患者血清MK水平>500 ng/L和≤500 ng/L两组中, 1年无转移生存率分别为29.09%和94.97%,且血清MK水平高者,1年无转移生存率明显降低。而有关MK在肿瘤发生发展的基础研究中发现,人体中存在多种MK受体,包括受体酪氨酸蛋白磷酸酶ζ(PRTPζ)、间变性淋巴瘤激酶(ALK)、神经多糖C(NGC)、低密度脂蛋白受体相关蛋白(LRP)、蛋白多糖(SDC)、Notch、整合素和核仁素等[14]。目前研究结果表明,MK可能通过与受体ALK结合,使其磷酸化,进一步激活磷脂酰肌醇3激酶(PI3K)及丝裂素活化蛋白激酶(MAPK)信号通路,使细胞凋亡受抑,细胞分裂、增殖速度加快[14];通过间变性淋巴瘤激酶(ALK)磷酸化,激活磷脂酰肌醇3激酶/蛋白激酶B、核因子κB、酪氨酸受体激酶B信号传导途径的各个因素,导致细胞失巢凋亡受抑制,进一步对表皮细胞生长及细胞外基质的黏附也失去抑制作用,同时也可破坏肿瘤细胞转移的生理屏障,并与受体Notch 2结合诱导上皮间质转化,共同促进肿瘤细胞侵犯及转移[7]。
有关Gal-3的分子机制研究结果显示,Gal-3在肿瘤的发生发展中可能通过以下几个途径来发挥作用:①通过与受体结合进一步激活磷脂酰肌醇3激酶/蛋白激酶B信号传导通路,来达到减少线粒体膜内外电位差的目的,进而抑制半胱氨酸天冬氨酸蛋白酶1(Caspase-1)及半胱氨酸天冬氨酸蛋白酶3(Caspase-3)的活动调控细胞凋亡;②细胞凋亡刺激物被修饰为促细胞分裂素或调整细胞外基质间细胞黏附,促进细胞生长、迁徙及抑制凋亡;③通过β连黏蛋白/Wnt信号通路的活化促进肿瘤细胞生长加速[15]。在分子水平研究的基础上,佟杰等[16]发现,甲状腺乳头状癌高表达Gal-3,阳性率达58%~100%。在本研究中,我们检测了血清中Gal-3的水平,结果提示恶性结节组中的Gal-3水平明显高于良性组和健康对照组;良恶性结节鉴别的最佳阈值为1.61 ng/mL,AUC为0.71,具有较高的诊断特异度和灵敏度(分别为70.4%和69.8%)。这与薛刚等[8]的研究结果一致。Saussez等[17]研究了甲状腺良恶性结节中血清Gal-3的水平,结果发现其最佳阈值为3.2 ng/mL,诊断灵敏度为73%、特异度为74%。但Dobrescu等[18]的研究结果与上述研究不一致,他们发现,在良恶性结节中,血清Gal-3水平的差异无统计学意义,与肿瘤侵袭性也无相关性,这可能是因为其研究的样本量较上述研究少,所以结论有所不同。以上诸多研究结果显示,Gal-3水平在甲状腺结节的良恶性鉴别中可能也具有一定的诊断价值,但仍需要大样本量的研究来进一步证实。
有关血清Gal-3水平在肿瘤转移及预后的作用在结直肠癌等的相关研究中也有所体现[19],而在对DTC的研究中,也有学者得出与本研究类似的结果:转移阳性组中血清Gal-3水平明显高于阴性组[20]。相关研究结果显示,与淋巴结转移阴性组对比,转移阳性组中DTC患者血清中Gal-3水平的表达量明显增高,并可根据Gal-3水平表达的高低来初步判断甲状腺癌是否有转移灶的存在;血清Gal-3水平与肿瘤分期呈正相关,分期越高,血清中Gal-3的水平越高[21]。而其中分子机制可能是Gal-3通过在低氧环境中促进上皮间充质化,再进一步激活磷脂酰肌醇3激酶/蛋白激酶B信号传导通路以及丝裂素活化蛋白激酶、β连黏蛋白/Wnt和Src/黏着斑激酶信号通路这一途径来实现细胞转移[22]。
本研究结果发现,血清中MK和Gal-3水平对甲状腺良恶性结节的诊断有一定临床价值,并且对评估DTC术后是否还存在转移灶也有一定的意义。此外,我们还发现,血清MK与Gal-3之间有明显的相关性,这可能与两者在甲状腺癌中的分子机制有一定相关性,有待进一步深入研究。但本研究中的样本量有限(特别是转移阳性组与阴性组),未能将血清MK、Gal-3水平分别与Tg水平进行对比研究,另外随访日期较短,缺少对患者远期预后的评估,日后将进一步增加样本量,延长随访时间,更深一步地探索、求证两者在甲状腺疾病诊断中的应用价值。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 武晨负责研究方案的设计与实施、数据的采集与统计分析、论文的撰写;杜芬负责研究方案的实施、数据的采集、统计分析与指导;刘师伟、马浩华负责数据的采集、论文撰写的指导;张海涛负责数据的采集、技术支持;刘静负责研究方案的实施、数据的采集;张迎伟负责数据的分析与指导;罗丽萍负责协助研究的进行、数据的采集与分析;程艳负责研究命题的提出、论文的审阅。
血清MK和Gal-3水平在甲状腺结节及DTC术后转移灶中的诊断价值
Diagnostic value of serum MK and Gal-3 in thyroid nodules and postoperative DTC metastases
-
摘要:
目的 探讨血清midkine(MK)和galectine-3(Gal-3)水平在良恶性甲状腺结节鉴别诊断中的价值,及其在分化型甲状腺癌(DTC)术后患者首次131I治疗前转移灶中的评估价值。 方法 ①甲状腺结节鉴别诊断的回顾性研究:收集2016年11月至2017年11月在山西医科大学第一医院拟行手术治疗的41例甲状腺结节患者[男性11例、女性30例,年龄29~73(48.61±11.59)岁]的术前血清样本,根据病理结果分为恶性组(23例)和良性组(18例),同时收集32名健康者作为健康对照组。采用单因素方差分析术前血清MK和Gal-3水平在良恶性甲状腺结节中是否存在差异,并绘制受试者工作特征曲线分析两者对甲状腺结节的诊断价值。②DTC患者术后是否存在转移灶的回顾性研究:收集2016年11月至2017年11月于山西医科大学第一医院核医学科首次服131I治疗前的51例DTC术后患者[男性12例、女性39例,年龄25~72(44.41±10.21)岁]的血清样本及临床资料,所有患者行2次131I治疗,并于每次治疗后5~7 d行全身碘扫描,根据其结果及其他检查(超声、CT、PET/CT)结果,判断是否有淋巴结和(或)肺转移。采用两独立样本t检验比较组间血清MK和Gal-3水平的差异性,Pearson检验分析血清MK和Gal-3水平间的相关性。 结果 ①手术前恶性组的血清MK和Gal-3水平明显高于良性组及健康对照组,且差异有统计学意义(F=48.40、5.08,均P<0.05)。对于术前良恶性甲状腺结节的鉴别,MK的最佳阈值为318.87 pg/mL、曲线下面积(AUC)为0.91(95%CI: 0.81~0.99)、特异度为78.1%、灵敏度为88.5%;Gal-3的最佳阈值为1.61 ng/mL、AUC为0.72(95%CI: 0.57~0.86)、特异度为70.4%、灵敏度为69.8%。②51例DTC患者中,转移阳性组8例、转移阴性组43例。转移阳性组甲状腺球蛋白、MK、Gal-3水平均明显高于转移阴性组,且差异有统计学意义(t=−4.86、−4.01、−2.89,均P<0.05)。Pearson相关性分析结果显示,甲状腺结节患者术前(r=0.67,P<0.05)及DTC患者术后(r=0.84,P<0.05)血清MK、Gal-3水平之间均存在显著相关性。 结论 血清MK和Gal-3水平可用于良恶性甲状腺结节的鉴别诊断,并且可以评估DTC术后是否存在肿瘤转移。 -
关键词:
- 甲状腺结节 /
- 甲状腺肿瘤 /
- 肿瘤转移 /
- 血清midkine /
- 血清galectine-3
Abstract:Objective To investigate the value of serum midkine (MK) and galectine-3 (Gal-3) in the differential diagnosis of benign and malignant thyroid nodules, and to analyze the ability of serum MK and Gal-3 to evaluate metastases before the first 131I treatment in patients with differentiated thyroid cancer (DTC) after thyroidectomy. Methods ①Data on benign and malignant thyroid nodule differential diagnosis were collected from November 2016 to November 2017 in the First Hospital of Shanxi Medical University. In general surgery, 41 cases (11 males and 30 females, aged 29−73 (48.61±11.59) years) of surgical treatment of thyroid nodules in patients with preoperative blood specimens were determined. The patients were divided according to the result of pathology as follows: malignant group (23 cases) and benign group (18 cases). A total of 32 healthy subjects served as controls. One-way ANOVA was used to analyze whether preoperative serum MK and Gal-3 were different in benign and malignant thyroid nodules. Receiver operating characteristic curve was drawn to study the diagnostic value of the two in thyroid nodules. ②From November 2016 to November 2017, 51 patients (12 males and 39 females, aged 25−72 (44.41±10.21) years) with DTC before the first 131I treatment in the Nuclear Medicine Department of the First Hospital of Shanxi Medical University participated in the evaluation of postoperative metastasis. All patients underwent 131I treatment two times, and the whole body iodine scan was performed 5−7 days after each treatment. According to the results and other examination results (ultrasound, CT, PET/CT), the lymph node and/or lung metastasis was judged. Serum was collected before the first iodine treatment. The differences in MK and Gal-3 between groups were compared using two-sample t-test. Pearson test was used to analyze the correlation between serum MK and Gal-3.before the first iodine treatment. The differences in MK and Gal-3 between groups were compared using two-sample t-test. Pearson test was used to analyze the correlation between serum MK and Gal-3. Results ①Pre-surgical MK and Gal-3 levels were significantly higher in patients with malignant thyroid nodule than those in benign cases and in controls (F=48.40, 5.08, both P<0.05). The ROC curve analysis determined the optimal value of MK and Gal-3 for differential diagnosis between malignant and benign thyroid nodules. The diagnostic power of MK was the highest at 318.87 pg/mL, with an area under the ROC curve (AUC) of 0.91(95%CI: 0.81~0.99), specificity of 78.1%, and sensibility of 88.5%. The diagnostic power of Gal-3 was the highest at 1.61 ng/mL, with AUC of 0.72(95%CI: 0.57~0.86), specificity of 70.4%, and sensibility of 69.8%. ②The patients were grouped according to the presence of lymph node and bone and/or lung metastasis into metastasis positive group (8 cases) and negative group (43 cases). Thyroglobulin, MK(A), and Gal-3(A) in the positive group were significantly higher than those in the negative group (t=−4.86, −4.01, −2.89, all P<0.05). MK (r=0.67, P<0.05) and Gal-3 (r=0.84, P<0.05) were significantly correlated in pre-surgical and pre-131I-ablative. Conclusions Serum MK and Gal-3 can be used for the differential diagnosis of benign and malignant thyroid nodules and to evaluate whether metastasis is present in patients with DTC before 131I treatment. -
Key words:
- Thyroid nodule /
- Thyroid neoplasms /
- Neoplasm metastasis /
- Serum midkine /
- Serum galectine-3
-
表 1 分化型甲状腺癌患者术后转移阳性组患者淋巴结和 (或)肺转移部位
Table 1. Locations of lymph node and/or lung metastasis in differentiated thyroid carcinoma positive group
编号 年龄 性别 转移部位 1 25 女 双侧颈Ⅱ区 2 28 女 左颈Ⅲ区及右颈Ⅵ区 3 33 男 右颈Ⅳ区 4 34 女 右颈Ⅲ、Ⅳ区 5 43 男 双肺多发转移 6 47 女 左颈Ⅵ区及右颈Ⅱ区 7 52 女 右颈Ⅲ、Ⅳ区 8 61 女 双肺多发转移 表 2 2组甲状腺结节患者和健康对照者的血清学指标比较 (
$\bar x $ Table 2. Comparison of serological indexes among malignant, benign and healthy thyroid nodules (
$ \bar x$ 组别 MK(pg/mL) Gal-3(ng/mL) 恶性组(n=23) 385.79±198.62 2.97±1.45 良性组(n=18) 280.12±101.24 1.29±0.33 健康对照组(n=32) 270.93±104.18 1.05±0.39 F值 48.40 5.08 P值 <0.01 0.01 注:表中,MK:midkine;Gal-3:galectine-3 表 3 分化型甲状腺癌患者术后首次131I治疗前2组的血清 学指标比较(
$\bar x $ Table 3. Comparison of serum indexes between 2 groups in patients after differentiated thyroid carcinoma before the first 131I treatment (
$\bar x $ 组别 MK(pg/mL) Gal-3(ng/mL) Tg(ng/mL) 转移阳性组
(n=8)489.93±124.69 2.82±1.51 176.30±131.94 转移阴性组
(n=43)277.41±115.24 1.74±0.66 15.10±14.38 t值 −4.01 −2.89 −4.86 P值 <0.01 <0.01 <0.01 注:表中,MK:midkine;Gal-3:galectine-3;Tg:甲状腺球蛋白 -
[1] Gharib H, Papini E, Garber JR, et al. American association of clinical endocrinologists, american college of endocrinology, and associazione medici endocrinologi medical guidelines for clinical practice for the diagnosis and management of thyroid nodules-2016 up date[J]. Endocr Pract, 2016, 22(5): 622−639. DOI: 10.4158/EP161208.GL. [2] Kuzu F, Arpazi D, Unal M, et al. Midkine: a novel biomarker to predict malignancy in patients with nodular thyroid disease[J]. Int J Endocrinol, 2016, 2016: 6035024. DOI: 10.1155/2016/6035024. [3] Czepczyński R, Stangierski A, Oleksa R, et al. Incidental 18F-FDG uptake in the thyroid in patients diagnosed with PET/CT for other malignancies[J]. Nucl Med Rev Cent East Eur, 2011, 14(2): 68−72. DOI: 10.5603/NMR.2011.00018. [4] Barrio M, Czernin J, Yeh MW, et al. The incidence of thyroid cancer in focal hypermetabolic thyroid lesions: an 18F-FDG PET/CT study in more than 6000 patients[J]. Nucl Med Commun, 2016, 37(12): 1290−1296. DOI: 10.1097/MNM.0000000000000592. [5] Muramatsu T, Kadomatsu K. Midkine: an emerging target of drug development for treatment of multiple diseases[J]. Br J Pharmacol, 2014, 171(4): 811−813. DOI: 10.1111/bph.12571. [6] 李春红, 曹鸿艳, 蔡莉. Galectin-3的抗凋亡机制及其在肺癌中的研究[J]. 国际免疫学杂志, 2008, 31(3): 229−232. DOI: 10.3760/cma.j.issn.1673-4394.2008.03.019.
Li CH, Cao HY, Cai L. Research on anti-apoptosis mechanism of Galectin-3 and the expression in lung cancer[J]. Int J Immunol, 2008, 31(3): 229−232. DOI: 10.3760/cma.j.issn.1673-4394.2008.03.019.[7] Sun B, Hu CL, Yang ZB, et al. Midkine promotes hepatocellular carcinoma metastasis by elevating anoikis resistance of circulating tumor cells[J/OL]. Oncotarget, 2017, 8(20): 32523−32535[2019-06-01]. https://pubmed.ncbi.nlm.nih.gov/28430645/. DOI: 10.18632/oncotarget.15808. [8] 薛刚, 刘军超, 黄静, 等. 血清与组织glactin-3 联合检测在甲状腺癌早期诊断中的价值[J]. 南方医科大学学报, 2013, 33(7): 1027−1030. DOI: 10.3969/j.issn.1673-4254.2013.07.18.
Xue G, Liu JC, Huang J, et al. Detection of glactin-3 in both serum and tissue for early diagnosis of thyroid carcinoma[J]. J South Med Univ, 2013, 33(7): 1027−1030. DOI: 10.3969/j.issn.1673-4254.2013.07.18.[9] Meng ZW, Tan J, Zhang GZ, et al. Evaluation of serum midkine as a biomarker in differentiated thyroid cancer[J]. Life Sci, 2015, 130: 18−24. DOI: 10.1016/j.lfs.2015.02.028. [10] 冯光勇, 黄伟. 血清肝素结合生长因子对分化型甲状腺肿瘤诊断及临床预后的评估价值[J]. 临床血液学杂志, 2017, 30(8): 600−604. DOI: 10.13201/j.issn.1004-2806-b.2017.08.007.
Feng GY, Huang W. Diagnosis and prognosis prediction value of serum midkine in differentiated thyroid cancer[J]. J Clin Hematol, 2017, 30(8): 600−604. DOI: 10.13201/j.issn.1004-2806-b.2017.08.007.[11] Jing X, Cui XR, Liang HP, et al. Diagnostic accuracy of ELISA for detecting serum Midkine in cancer patients[J/OL]. PLoS One, 2017, 12(7): e0180511[2019-06-01]. https://pubmed.ncbi.nlm.nih.gov/28686647/. DOI:10.1371/journal.pone.0180511 [12] 李田军, 林岩松, 梁军, 等. 131I治疗前刺激性Tg对乳头状甲状腺癌远处转移的预测价值[J]. 中华核医学与分子影像杂志, 2012, 32(3): 189−191. DOI: 10.3760/cma.j.issn.2095-2848.2012.03.007.
Li TJ, Lin YS, Liang J, et al. The value of pre-ablation stimulated thyroglobulin in predicting distant metastasis of papillary thyroid cancer[J]. Chin J Nucl Med Mol Imaging, 2012, 32(3): 189−191. DOI: 10.3760/cma.j.issn.2095-2848.2012.03.007.[13] Jee YH, Celi FS, Sampson M, et al. Midkine concentrations in fine-needle aspiration of benign and malignant thyroid nodules[J]. Clin Endocrinol (Oxf), 2015: 83. DOI: 10.1111/cen.12676. [14] Sorrelle N, Dominguez ATA, Brekken RA, et al. From top to bottom: midkine and pleiotrophin as emerging players in immune regulation[J]. J Leukoc Biol, 2017, 102(2): 277−286. DOI: 10.1189/jlb.3MR1116-475R. [15] Dumic J, Dabelic S, Flögel M. Galectin-3: an open-ended story[J]. Biochim Biophys Acta, 2006, 1760(4): 616−635. DOI: 10.1016/j.bbagen.2005.12.020. [16] 佟杰, 王岩, 笪冀平. CK19 HBME-1 Galectin-3等表达在甲状腺乳头状微小癌与良性病变鉴别诊断中的意义[J]. 中华肿瘤杂志, 2011, 33(8): 599−604. DOI: 10.3760/cma.j.issn.0253-3766.2011.08.010.
Tong J, Wang Y, Da JP. Usefulness of CK19, HBME-1 and Galectin-3 expressions in differential diagnosis of thyroid papillary microcarcinoma from benign lesions[J]. Chin J Oncol, 2011, 33(8): 599−604. DOI: 10.3760/cma.j.issn.0253-3766.2011.08.010.[17] Saussez S, Glinoer D, Chantrain G, et al. Serum galectin-1 and galectin-3 levels in benign and malignant nodular thyroid disease[J]. Thyroid, 2008, 18(7): 705−712. DOI: 10.1089/thy.2007.0361. [18] Dobrescu R, Schipor S, Manda D, et al. Does serum Galectin-3 add value in thyroid cancer diagnosis?[J/OL]. Endocrine Abstracts, 2016, 41: GP232[2019-06-01]. https://www.endocrine-abstracts.org/ea/0041/ea0041GP232.htm. DOI: 10.1530/endoabs.41.GP232. [19] 任丽丽, 耿建. 老年结肠癌病人循环与肿瘤组织中Galectin-3水平及临床意义[J]. 实用老年医学, 2017, 31(2): 153−156. DOI: 10.3969/j.issn.1003-9198.2017.02.015.
Ren LL, Geng J. Clinical and prognostic effects of Galectin-3 in elderly patients with colorectal cancer[J]. Pract Geriatr, 2017, 31(2): 153−156. DOI: 10.3969/j.issn.1003-9198.2017.02.015.[20] 尚培中, 柳勇, 李永庆, 等. COX-2和Gal-3表达与甲状腺乳头状癌浸润转移的关系[J]. 河北北方学院学报: 自然科学版, 2016, 32(8): 1−4. DOI: 10.3969/j.issn.1673-1492.2016.08.001.
Shang PZ, Liu Y, Li YQ, et al. Expressions of COX-2 and Gal-3 and invasion and metastasis in papillary thyroid carcinoma[J]. J Hebei North Univ (Natur Sci), 2016, 32(8): 1−4. DOI: 10.3969/j.issn.1673-1492.2016.08.001.[21] 刘燕青. 甲状腺癌患者血清中galectin-3、midkine含量测定对肿瘤病情评估的价值[J]. 海南医学院学报, 2017, 23(3): 418−421. DOI: 10.13210/j.cnki.jhmu.20161124.001.
Liu YQ. Value of serum galectin-3 and midkine level for assesing tumor severity in patients with thyriod cancer[J]. J Hainan Med Univ, 2017, 23(3): 418−421. DOI: 10.13210/j.cnki.jhmu.20161124.001.[22] Zheng JJ, Lu WH, Wang C, et al. Galectin-3 induced by hypoxia promotes cell migration in thyroid cancer cells[J/OL]. Oncotarget, 2017, 8(60): 101475−101488[2019-06-01]. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5731889/. DOI:10.18632/oncotarget.21135. -





 下载:
下载:



