-
心肌灌注显像(myocardial perfusion imaging,MPI)是临床常用的无创性诊断心肌缺血的影像学方法,在冠心病诊断、疗效评价和预后判断中发挥着重要作用,是国内外认可的一项重要的检查技术[1]。目前国内主要采用SPECT进行MPI检查,常用显像剂为99Tcm-MIBI和99Tcm-替曲膦[2]。众多研究结果表明,SPECT MPI有一定的局限性,与PET相比其灵敏度较低,受膈肌和乳腺的衰减伪影影响较大,且传统SPECT无法进行心肌血流的绝对定量分析[3-4]。此外,PET MPI检查中患者所受辐射剂量更小[5]。但由于PET MPI中常用的显像剂为13N-NH3·H2O(13N-氨水),需要配备价格昂贵的医用回旋加速器,并且因其物理半衰期较短(9.98 min),导致患者显像的通过量低;另外,由于运动负荷显像很难进行,导致PET MPI的临床应用受到很大的限制,当前国内只有少数临床机构具备开展PET MPI的能力。因此,开发半衰期相对较长、适于配送的正电子心肌灌注显像剂已成为当前研究的热点之一[6],其中一种新型18F标记的心肌灌注显像剂18F-Flurpiridaz(BMS-747158)在国外已进入临床Ⅲ期研究阶段,其应用前景广阔[7-8]。目前国内尚未见此类显像剂在动物或人体显像的研究报道,为此,本研究探讨18F-Flurpiridaz的合成工艺,并初步研究其在正常巴马小型猪体内的MPI图像质量和体内分布特点。
-
Discovery Elite PET/CT、MINItrac医用回旋加速器、TracerLab FN正电子药物合成器均购自美国通用电气公司;CRC-25R放射性活度计购自美国CAPINTEC公司;MINIscan薄层扫描仪购自美国BIOSCAN公司。
-
H218O为以色列RCTEM公司产品;2-叔丁基-4-氯-5 - [4-(2-甲基磺酰基-乙氧基甲基)苯基甲氧基]哒嗪-3-哒嗪酮(简称前体)为天津希恩斯奥普德科技有限公司产品;Sep-Pak QMA色谱柱(简称QMA柱)、Sep-Pak C18固相萃取柱(简称C18柱)均为美国Waters公司产品;Maxi-Clean色谱柱为美国Alltech公司产品;氨基聚醚(K2.2.2)和去离子水均为德国ABX公司产品;K2CO3、无水乙腈、无水乙醇等试剂均为美国Sigma公司产品。
-
12~14月龄、体重30~35 kg的普通级实验用正常巴马小型猪5头,雌雄不限。实验前禁食12 h、禁饮4 h。实验动物由天津市百农实验动物繁育科技有限公司提供[SCXK(津)2015-0002],饲养于普通环境内,室温18℃~23℃,湿度60%~80%,常规饲养,饲养条件符合国家要求。实验中涉及动物的操作已经得到我院实验动物福利伦理审查委员会批准,批准号为:TICH-JY-20180426-1。
-
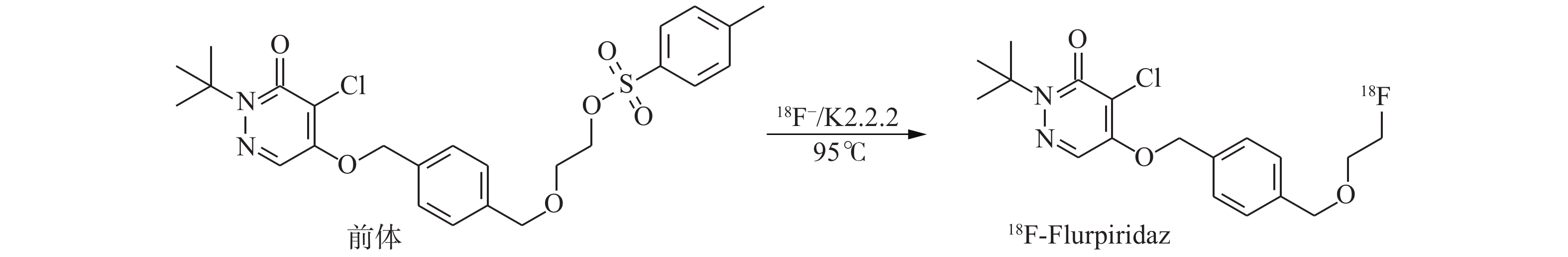
18F-Flurpiridaz的合成是基于文献[9]报道的方法并进行优化处理的。使用回旋加速器轰击H18O,通过(p,n)核反应产生18F−,结束后将靶水传送至合成器,18F−通过并富集在QMA柱上,用K2CO3溶液将18F−洗脱到反应瓶中,加入氨基聚醚(K2.2.2)溶液,负压加热蒸干除水。将前体溶液加入反应瓶中,95℃密闭反应20 min,反应结束并冷却至室温。反应液注入半制备型高效液相色谱仪(制备柱:NM C18,250 mm×16 mm,紫外检测波长254 nm,流动相:乙腈和水的比例为75:25,流速:8 mL/min)进行纯化,收集目标组分,将此含有产品的乙腈溶液通过C18柱,富集产品18F-Flurpiridaz,用10 mL注射用水洗涤C18柱,用0.5 mL乙醇将18F-Flurpiridaz从C18柱洗脱至产品瓶,向产品瓶中加入9.5 mL生理盐水。根据上述条件在TracerLab FN正电子药物合成器上建立自动合成程序生产18F-Flurpiridaz,最终产物经0.22 μm无菌过滤器过滤后备用。吸取少量样品点样于硅胶板上,展开后取出晾干,置于放射性薄层扫描仪上进行放射化学纯度测定。在室温下分别测定18F-Flurpiridaz放置1、2、4 h后的放射化学纯度。18F-Flurpiridaz的合成路线如图1所示。
-
将5头正常巴马小型猪利用吹管麻醉技术完成基础麻醉,耳部备皮消毒,使用留置针,建立耳中静脉通路[10]。采用丙泊酚诱导麻醉后以仰卧位固定在PET/CT检查床上,先行CT衰减校正采集(电压120 kV,电流15 mAs),经静脉通道快速推注18F-Flurpiridaz约37 MBq(3 mL),10 min后行心脏部位断层显像,采用心电门控三维采集模式(8 min/床位,8帧/心动周期,心率窗为±15%)。采用有序子集最大期望值法行图像重建,图像以短轴、垂直长轴、水平长轴表示,所有数据均在Xeleris工作站进行图像融合及后处理。所有实验猪一周内行13N-NH3·H2O PET/CT显像,采集注射显像剂后10 min的心脏部位图像,注射剂量和采集条件同前所述。
-
上述5头正常巴马小型猪维持麻醉,于注射18F-Flurpiridaz后30和60 min进行PET/CT全身显像,采集条件:CT扫描电压120 kV,自动毫安秒;PET采集1.5 min/床位,层厚3.75 mm,视野50 cm。所有数据均在Xeleris工作站进行图像融合及后处理。在靶与非靶器官上勾画ROI,测定放射性计数。
-
自18F−传输起至产品传输到产品瓶,用时约50 min,非衰变校正后的放射化学产率约为40%,比活度>5.6×108 Bq/mL,放射化学纯度>97%,在室温中放置4 h后,放射化学纯度仍>95%。
-
5头正常巴马小型猪全部成功显像,18F-Flurpiridaz与13N-NH3·H2O PET/CT MPI显像结果对比见图2,由上及下依次为短轴、垂直长轴和水平长轴断层图。结果显示,18F-Flurpiridaz显像心肌显影清晰,各节段心肌显像剂分布均匀,心腔本底计数低,无心外放射性干扰,与13N-NH3·H2O显像相比具有优良的成像质量。
-
18F-Flurpiridaz PET/CT全身显像结果如图3所示,心肌始终有很高的放射性摄取,心脏显影清晰,心脏周边的非靶器官无明显摄取。30 min显像时,心脏和肾脏摄取明显,肝脏有少量摄取(心/肝>3),肺接近本底水平(心/肺>15)。60 min显像时,放射性分布大致同前,心脏依然具有较高的放射性摄取,肾脏摄取较前减低(30和60 min显像时的心/肾分别为0.7 和 1.2)。两次显像四肢肌肉均有放射性摄取,但变化不大,心/肌肉均>7。
-
Purohit等[9]报道了18F-Flurpiridaz的制备及纯化方法,该方法的放射化学产率约为5%,用时90 min。本研究对该方法进行了部分优化,放射化学产率约为40%,用时仅为50 min。主要从两个方面进行了改进。(1)标记过程:文献中条件为加热90℃,反应30 min;我们采用的条件为加热95℃,反应20 min,同样可获得较好的标记结果,并且缩短了反应时间。(2)分离纯化过程:文献中采用的方法较为繁琐,对实验设备要求较高(标记反应结束后,反应液先通过C18柱除去部分杂质,再进入高效液相色谱系统进行分离纯化,收集目标组分后加热去除流动相,终产品用90%乙醇溶解);我们优化后的方法相对简化(反应结束后反应液直接转入高效液相色谱系统,收集目标组分后,采用固相萃取富集产品)。优化后的方法操作更加简便,且用时减少了40 min,有利于减少放射性衰变损失,提高放射化学产率。此外,文献中采用梯度洗脱的方式进行纯化,该方法需配备双泵,而国内常用合成模块通常配备单泵,只能进行等度洗脱。本研究采用75%乙腈作为流动相进行等度洗脱,分离效果较好。Tracerlab合成器具备程序编辑功能,我们根据制备条件建立了18F-Flurpiridaz的自动合成程序,在制备过程中无需手工进行放射性操作,且能更加准确地控制反应条件(温度、压力、时间等),避免了人为原因导致的合成失败,对于该显像剂的临床应用推广非常有利。
根据临床应用的实际情况,传统的PET MPI显像剂难以满足需求,15O-H2O(T1/2=2 min)和13N-NH3·H2O(T1/2=9.98 min)的半衰期均较短,无法实现区域配送,需要临床机构配备回旋加速器,极大地限制了PET MPI在临床中的应用[11-12]。82Rb(T1/2=1.25 min)需从发生器获得,价格昂贵,国内尚无供货,光子成像计数低、本底噪声大、影像质量不理想[13]。18F显像的空间分辨率优于15O、13N和82Rb,其半衰期为108 min,可以方便地进行区域间的短距离运输[14]。由于F的范德华半径与H接近,以F取代化合物分子中非生物活性位点处的H,对其生物活性无影响。心肌细胞中含有丰富的线粒体,其中线粒体复合物Ⅰ是线粒体内膜电子传递链的开端,参与能量代谢过程。Flurpiridaz以线粒体复合物Ⅰ为靶点,具有较高的亲和力,且对心肌细胞的功能和活性无影响[15]。Maddahi等[16]研究结果表明,健康志愿者进行18F-Flurpiridaz静息和负荷(运动负荷或药物负荷)显像安全,受到的辐射剂量低,未发现药物相关性不良反应。本研究中MPI结果显示了18F-Flurpiridaz的优良特性,心肌摄取明显,显影清晰,周边的非靶组织基本无摄取。注射后60 min内,心肌细胞均有很高的放射性摄取,显像的时间窗口期长,并可以进行运动负荷显像,极大地方便了临床使用。PET/CT全身显像提示肌肉组织存在一定的放射性摄取,这与肌肉组织中含有丰富的线粒体有关;肾脏内有放射性浓聚,这可能与肾小管中也含有较多的线粒体有关。
本研究结果表明,经过优化合成的18F-Flurpiridaz的放射化学产率较为理想,体外稳定性较好,初步动物实验结果显示了优良的成像质量,较长的成像时间窗,心脏周围放射性本底影响小,非常适合作为能够配送的正电子显像剂。在此基础上,我们还计划对18F-Flurpiridaz进行深入的研究:进一步优化其标记和分离纯化的条件,提高产率;参照《中国药典》对其进行质量控制,以符合临床使用要求[17];在正常动物体内进行生物学分布研究;建立心肌梗死和心肌缺血的动物模型并进行显像实验;探究其定量血流分析的能力[18]。这些研究将为国内开展18F-Flurpiridaz的应用研究奠定基础。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 李帅负责现场实验、论文的撰写和最终版本的修订;刘天文、王帆负责现场实验;汪娇负责数据的处理与图像的分析;张杰民、李剑明负责命题的提出和论文的审阅。
18F-Flurpiridaz的制备及其在正常巴马小型猪PET/CT心肌灌注显像中的初步实验研究
Primary experimental study on 18F-flurpiridaz synthesis and its myocardial perfusion imaging through PET/CT in normal miniature pigs
-
摘要:
目的 探讨合成18F-Flurpiridaz的优化方法,评估其在正常巴马小型猪PET/CT心肌灌注显像(MPI)中的分布情况。 方法 以18F取代2-叔丁基-4-氯-5-[4-(2-甲基磺酰基-乙氧基甲基)苯基甲氧基]哒嗪-3-哒嗪酮中的苯磺酸离去基团进行标记,通过高效液相色谱非梯度洗脱的方法纯化产物。5头正常巴马小型猪经耳缘静脉注射37 MBq 18F-Flurpiridaz后10 min行PET/CT MPI,并于注射后30和60 min进行PET/CT全身显像,观察显像剂在其体内主要脏器的摄取情况。 结果 18F-Flurpiridaz的制备时间约为50 min,非校正放射化学产率为40%,放射化学纯度>97%。MPI图像显示心肌细胞摄取明显,肝脏仅有少量摄取,肺基本无摄取,心肌图像质量好。全身显像中,显像剂注射后30 min,心肌和肾脏摄取明显,肌肉组织中有少量摄取,而其他组织器官均无明显摄取;显像剂注射后60 min,心脏内仍有较高摄取。 结论 实现并优化了18F-Flurpiridaz的自动化合成,为其临床应用奠定了基础。 -
关键词:
- 心肌灌注显像 /
- 氟放射性同位素 /
- Flurpiridaz /
- 正电子发射断层显像计算机体层摄影术 /
- 放射合成
Abstract:Objectve To explore the synthesis method of 18F-flurpiridaz and evaluate its ability for myocardial perfusion imaging (MPI) through PET/CT and bio-distribution in normal miniature pigs. Methods 18F-Flurpiridaz was prepared through the substitution of the toluene sulfonate leaving group of tert-butyl-4-chloro-5(4-(2-methyl(sulfonyl-ethoxymethyl)phenyl)methyl)pyridazin-3-one followed by radiolabeling with 18F. The product was isolated and purified by high performance liquid chromatography (HPLC) non-gradient elution. PET/CT MPI was performed 10 min after intravenous injection of 37 MBq 18F-flurpiridaz in five normal miniature pigs, and PET/CT whole-body scans were performed 30 and 60 min after injection to observe the bio-distribution. Results The total synthesis time (including HPLC separation) of 18F-flurpiridaz was approximately 50 min. The radiochemical yield was 40% (decay uncorrected), and the radiochemical purity was >97% (after HPLC purification). MPI results demonstrated that the radioactive uptake primarily accumulated in heart muscles, whereas little radioactivity was distributed throughout the liver and lungs. Moreover, the image quality was good. The whole-body PET/CT data showed high uptake in heart muscles and kidneys, whereas the skeleton muscle only had little radioactive uptake. No evident accumulations of activity in other organs were observed. 18F-Flurpiridaz had higher retention in the myocardium 60 min after injection. Conclusion The automatic synthesis of 18F-flurpiridaz is realized and optimized, which lays a foundation for its clinical application. -
-
[1] 孙沫沫, 李剑明. PET心肌灌注显像及其定量分析的研究进展[J]. 国际放射医学核医学杂志, 2017, 41(6): 423−429. DOI: 10.3760/cma.j.issn.1673−4114.2017.06.008.
Sun MM, Li JM. The research progress of myocardial perfusion and its quantitative analysis with PET[J]. Int J Radiat Med Nucl Med, 2017, 41(6): 423−429. DOI: 10.3760/cma.j.issn.1673−4114.2017.06.008.[2] 谢文晖, 蔡小佳, 黄钢. 常见心肌灌注显像剂及其显像程序[J]. 实用医学杂志, 2009, 25(9): 1524−1526. DOI: 10.3969/j.issn.1006−5725.2009.09.077.
Xie WH, Cai XJ, Huang G. The current myocardial perfusion imaging agents and its imaging procedures[J]. J Pract Med, 2009, 25(9): 1524−1526. DOI: 10.3969/j.issn.1006−5725.2009.09.077.[3] Bateman TM. Advantages and disadvantages of PET and SPECT in a busy clinical practice[J]. J Nucl Cardiol, 2012, 19(S1): 3−11. DOI: 10.1007/s12350−011−9490−9. [4] Beanlands RS, Youssef G. Diagnosis and prognosis of coronary artery disease: PET is superior to SPECT: Pro[J]. J Nucl Cardiol, 2010, 17(4): 683−695. DOI: 10.1007/s12350−010−9253−z. [5] Stabin MG. Radiopharmaceuticals for nuclear cardiology: Radiation dosimetry, uncertainties, and risk[J]. J Nucl Med, 2008, 49(9): 1555−1563. DOI: 10.2967/jnumed.108.052241. [6] 李帅, 李剑明. 18F标记新型正电子心肌灌注显像剂的研究进展[J]. 中国医学影像技术, 2014, 30(6): 926−929. DOI: 10.13929/j.1003−3289.2014.06.036.
Li S, Li JM. Progresses of new myocardial perfusion imaging agents labeled with 18F[J]. Chin J Med Imaging Technol, 2014, 30(6): 926−929. DOI: 10.13929/j.1003−3289.2014.06.036.[7] Maddahi J, Czernin J, Lazewatsky J, et al. Phase I, First-in-Human Study of BMS747158, a Novel 18F-Labeled Tracer for Myocardial Perfusion PET: Dosimetry, Biodistribution, Safety, and Imaging Characteristics After a Single Injection at Rest[J]. J Nucl Med, 2011, 52(9): 1490−1498. DOI: 10.2967/jnumed.111.092528. [8] Berman DS, Maddahi J, Tamarappoo BK, et al. Phase Ⅱ Safety and Clinical Comparison With Single-Photon Emission Computed Tomography Myocardial Perfusion Imaging for Detection of Coronary Artery Disease: Flurpiridaz F 18 Positron Emission Tomography[J]. J Am Coll Cardiol, 2013, 61(4): 469−477. DOI: 10.1016/j.jacc.2012.11.022. [9] Purohit A, Radeke H, Azure M, et al. Synthesis and Biological Evaluation of Pyridazinone Analogues as Potential Cardiac Positron Emission Tomography Tracers[J]. J Med Chem, 2008, 51(10): 2954−2970. DOI: 10.1021/jm701443n. [10] 刘天文, 王帆, 张杰民, 等. 小型猪吹管麻醉方法介绍[J]. 中国比较医学杂志, 2017, 27(4): 68, 88.
Liu TW, Wang F, Zhang JM, et al. The introduction of blow tube anesthesia for miniature pigs[J]. Chin J Comp Med, 2017, 27(4): 68, 88.[11] Werner RA, Chen XY, Rowe SP, et al. Moving into the next era of PET myocardial perfusion imaging: introduction of novel 18F-labeled tracers[J]. Int J Cardiovasc Imaging, 2019, 35(3): 569−577. DOI: 10.1007/S10554−018−1469−z. [12] Salata BM, Singh P. Role of cardiac PET in clinical practice[J]. Curr Treat Options Cardiovasc Med, 2017, 19(12): 93. DOI: 10.1007/s11936−017−0591−x. [13] Salerno M, Beller GA. Noninvasive assessment of myocardial perfusion[J]. Circ Cardiovasc Imaging, 2009, 2(5): 412−424. DOI: 10.1161/CIRCIMAGING.109.854893. [14] Iskandrian AE, Dilsizian V, Garcia EV, et al. Myocardial perfusion imaging: Lessons learned and work to be done-update[J]. J Nucl Cardiol, 2018, 25(1): 39−52. DOI: 10.1007/s12350−017−1093−7. [15] Yalamanchili P, Wexler E, Hayes M, et al. Mechanism of uptake and retention of F-18 BMS-747158-02 in cardiomyocytes: A novel PET myocardial imaging agent[J]. J Nucl Cardiol, 2007, 14(6): 782−788. DOI: 10.1016/J.NUCLCARD.2007.07.009. [16] Maddahi J, Bengel F, Czernin J, et al. Dosimetry, biodistribution, and safety of flurpiridaz F 18 in healthy subjects undergoing rest and exercise or pharmacological stress PET myocardial perfusion imaging[J]. J Nucl Cardiol, 2019, 26(6): 2018−2030. DOI: 10.1007/s12350−018−01484−z. [17] 国家药典委员会. 中华人民共和国药典2015年版. 第四部[M]. 北京: 中国医药科技出版社, 2015.
Chinese Pharmacopoeia Commission. Pharmacopoeia of the People's Republic of China 2015. Volume Ⅳ[M]. Beijing: China Medical Science Press, 2015.[18] Schindler TH, Quercioli A, Valenta I, et al. Quantitative assessment of myocardial blood flow−clinical and research applications[J]. Semin Nucl Med, 2014, 44(4): 274−293. DOI: 10.1053/j.semnuclmed.2014.04.002. -






 下载:
下载: