-
急性心肌梗死(acute myocardial infarction,AMI)患者的存活心肌与坏死心肌并存,明确心肌受损的程度及范围对于临床治疗决策具有重要意义[1]。研究表明,AMI后患者的心肌收缩功能改变,特别是存活心肌的比例是影响再发心血管事件的重要因素[2]。核素心肌血流灌注/代谢显像具有简单无创、可量化和易于获得等优点,是判断心肌损伤的部位、范围和程度最理想的方法[3]。本研究应用99Tcm-MIBI SPECT心肌灌注显像(myocardial perfusion imaging,MPI)与18F-FDG PET/CT心肌代谢显像,并分析AMI患者行经皮腔内冠状动脉成形术(percutaneous transluminal coronary angioplasty,PTCA)治疗后左心室收缩功能的变化,为临床治疗方案的选择及预后评估等提供参考。
-
回顾性分析2015年6月至2017年12月于山西医科大学附属第一医院行PTCA治疗的AMI患者(均为首次发病)36例,其中男性27例、女性9例,年龄(57.9±12.3岁)。所有患者均在AMI发病1周内行PTCA治疗,且在PCI治疗前3 d和治疗后6~8个月内行MPI。纳入标准按照2012年国际心脏病学会及世界卫生组织关于AMI的诊断标准[4],心肌生化标志物[心肌肌钙蛋白I (cardiactroponini,cTnI)]水平升高且超过参考值上限99百分位值,同时至少伴有下述心肌缺血症状之一:①缺血症状;②心电图提示新发缺血性改变;③心电图提示病理性Q波形成;④影像学证据提示新发局部室壁运动异常或存活心肌丢失。排除标准:其他因素引起的心功能不全,如:心肌病、瓣膜病、严重心律失常和先天性心脏病等。所有患者在检查前均签署了知情同意书。
-
(1)99Tcm-MIBI SPECT的静息MPI图像采集:患者静脉注射99Tcm-MIBI 925 MBq(25 mCi)后20 min进食脂餐,1~1.5 h后行静息MPI。采用德国Siemens公司的Symbia T16双探头SPECT显像仪及IQ-SPECT程序,低能高分辨锥形孔准直器进行1:1断层采集。双探头呈90°,自右前斜45°开始到左后斜45°,顺时针旋转180°,每25 s采集1帧,共采集34帧图像。采集矩阵为128×128,能峰为140 keV,窗宽正反各20%。(2)门控18F-FDG PET/CT心肌代谢显像图像采集:采用美国GE公司的discovery VCT型PET/CT仪,于99Tcm-MIBI静息MPI后次日行心肌代谢显像。显像前患者禁食8~12 h,于显像前1 h口服葡萄糖50~75 mg,测量并控制血糖水平在5.0~8.8 mmol/L,静脉注射18F-FDG 1 h后行PET/CT显像。患者18F-FDG注射剂量按体重(公斤)乘以0.1计算。心电门控PET图像采集前先进行10 s CT透射扫描(管电压120 mV,管电流35 mA),后采集图像10 min,采集矩阵256×256,放大倍数1.0,每个心动周期采集8帧图像。(3)图像重建与处理:利用仪器自带的图像处理软件(syngo MIApplications VA60A)对99Tcm-MIBI静息MPI及门控18F-FDG PET心肌代谢显像图像进行重建,重建方法选择Flash 三维,迭代次数为16次,子集个数为8个。利用仪器软件获得所有重建图像及定量参数。
-
所有图像均由2位核医学副高以上职称医师独立分析,诊断一致时方可认为有效;诊断不一致时请另1位主任医师对图像进行判读。18F-FDG心肌代谢PET图像重建后与静息MPI图像进行对比,采用美国心脏协会17节段4分评分法评估[5],0分:放射性分布正常,心肌对18F-FDG的摄取≥75%;1分:放射性分布轻度减低,50%≤心肌对18F-FDG的摄取<75%;2分:放射性分布严重减低,25%≤心肌对18F-FDG的摄取<50%;3分:放射性分布缺损,心肌对18F-FDG的摄取<25%;4分:无放射性分布,心肌无18F-FDG摄取。根据各节段心肌血流灌注/代谢的特点,将左心室分为正常心肌(灌注、代谢均正常)、存活心肌(灌注减低或缺损、代谢正常;或灌注减低且灌注/代谢不匹配)和梗死心肌(灌注、代谢均减低且灌注/代谢匹配)[6]。若同一心肌节段灌注显像比代谢显像评分高1分及1分以上,则判断为灌注/代谢不匹配,即心肌存活;若灌注显像与代谢显像均示放射性缺损,则判断为灌注/代谢匹配,即心肌坏死[7]。统计患者存活心肌/坏死心肌节段数(surviving myocardium/necrotic myocardium,S/N)的比值。左心室心功能参数利用QGS软件分析得出,记录患者左心室射血分数(left ventricular ejection fraction,LVEF)及高峰射血率(peak ejection rate,PER),6个月后患者LVEF增加5%及以上判断为左心室收缩功能改善[8-9]。
-
根据所有患者6个月后LVEF的变化值(ΔLVEF)分为2组,A组(24例):心功能改善组(ΔLVEF≥5%);B组(12例):心功能未改善组(ΔLVEF<5%)。常规记录患者年龄、性别、体重指数、吸烟史、饮酒史、高血压病史、糖尿病史、高脂血症病史、心绞痛病史、胸痛时间及心梗部位等病史资料。
-
使用SPSS21.0软件对数据进行统计学分析。符合正态分布的计量资料以
$\bar x$ ±s表示,计数资料以率和百分比表示。符合正态分布的组间数据、方差齐及满足独立性,差异的比较采用独立样本t检验。采用独立样本t检验和χ2检验分析患者的一般临床资料、S/N比值、LVEF等指标间的差异;应用Logistic回归分析S/N比值、PER与ΔLVEF之间的关系;采用ROC曲线寻找预测左心室收缩功能改善(ΔLVEF≥5%)的S/N界值。P<0.05表示差异有统计学意义。 -
由表1可见,A组与B组患者间年龄、性别、体重指数、吸烟史、饮酒史、高血压史、糖尿病史、高脂血症病史和心绞痛病史等差异均无统计学意义。
组别 男性/女性(例) 年龄(岁) 体重指数(kg/m2) 吸烟史 高血压病史 糖尿病史 高血脂症病史 心绞痛病史 A组(n=24) 18/6 53.6±9.3 24.75±3.60 16(66.7%) 17(70.8%) 13(54.2%) 12(50.0%) 16(66.7%) B组(n=12) 9/3 62.2±9.1 25.01±3.79 7(58.3%) 10(83.3%) 7(58.3%) 8(66.7%) 7(58.3%) 检验值 χ2=0.000 t=−2.828 t=−0.201 χ2=0.241 χ2=0.667 χ2=0.056 χ2=0.900 χ2=0.241 P值 1.000 0.110 0.842 0.429 0.403 0.819 0.103 0.429 注:表中,A组:心功能改善组(ΔLVEF≥5%);B组:心功能未改善组(ΔLVEF<5%)。ΔLVEF:左心室射血分数的变化值 表 1 2组急性心肌梗死患者一般临床特征的比较
Table 1. Comparison of general clinical characteristics between the two groups of patients with acute myocardial infarction
-
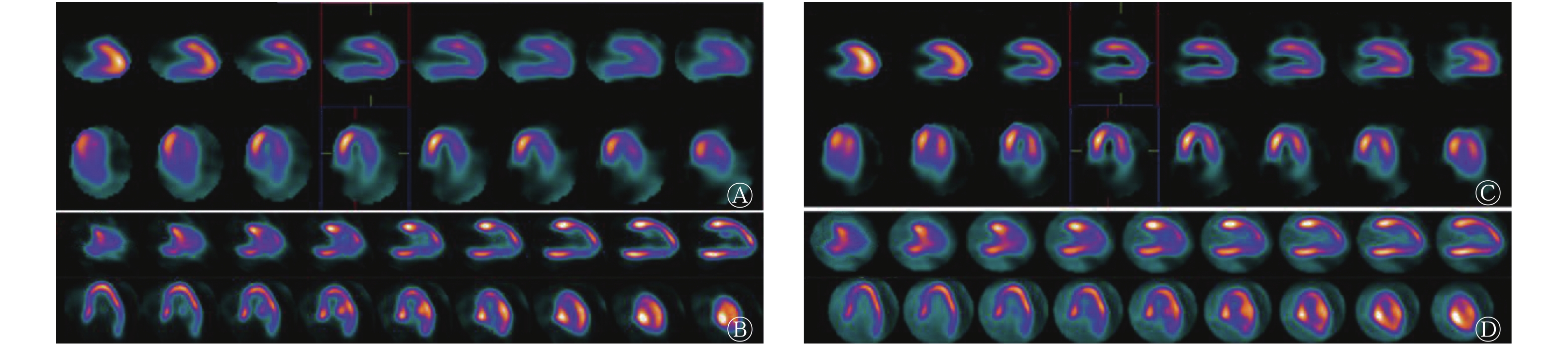
PTCA治疗前,A组与B组的LVEF、PER、cTnI比较,差异无统计学意义(t=−1.075~1.824,P=0.141~0.573),S/N比值的差异有统计学意义(t=0.824,P=0.042);PTCA治疗后,2组LVEF、PER、S/N比值的比较,差异有统计学意义(t=−2.528~−2.366,P=0.016~0.024),而cTnI的差异无统计学意义(t=−1.229,P=0.259)。具体数据见表2。典型病例的心肌灌注/代谢显像见图1。
图 1 急性心肌梗死患者(男性,43岁)PTCA治疗前后的心肌灌注/代谢显像图
Figure 1. Myocardial perfusion/metabolism imaging in patients with acute myocardial infarction (male, 43 years old) before and after percutaneous transluminal coronary angioplasty treatment
组别 LVEF(%) PER(%) S/N比值 cTnI(μg/L) 治疗前 治疗后 治疗前 治疗后 治疗前 治疗后 治疗前 治疗后 A组(n=24) 36±12 47±12 1.54±0.35 2.11±0.48 1.24±1.06 0.73±0.47 1.52±0.41 1.39±0.39 B组(n=12) 39±10 38±10 1.79±0.44 1.71±0.50 0.58±0.37 0.62±0.39 1.02±0.20 1.18±0.67 t值 0.569 −2.528 −1.075 −2.366 0.824 −2.514 −0.844 −1.229 P值 0.573 0.016 0.292 0.024 0.042 0.017 0.141 0.259 注:表中,A组:心功能改善组(ΔLVEF≥5%);B组:心功能未改善组(ΔLVEF<5%)。LVEF:左心室射血分数;PER:高峰射血率;S/N比值:存活心肌/坏死心肌节段数的比值;cTnI:心肌肌钙蛋白I 表 2 2组急性心肌梗死患者的左心室心功能参数的比较(
$\scriptstyle \bar x $ Table 2. Comparison of left ventricular cardiac function parameters between the two groups of patients with acute myocardial infarction (
$\scriptstyle \bar x$ -
对影响患者左心室功能改善的因素进行Logistic回归分析,结果筛选出与治疗后LVEF改善相关的因素,包括患者胸痛时间、心梗位置、存活心肌比例(S/N比值)及首次检查与PTCA治疗的间隔时间,其中S/N比值是患者左心室功能改善的独立影响因素(OR=2.164,P=0.018)。
-
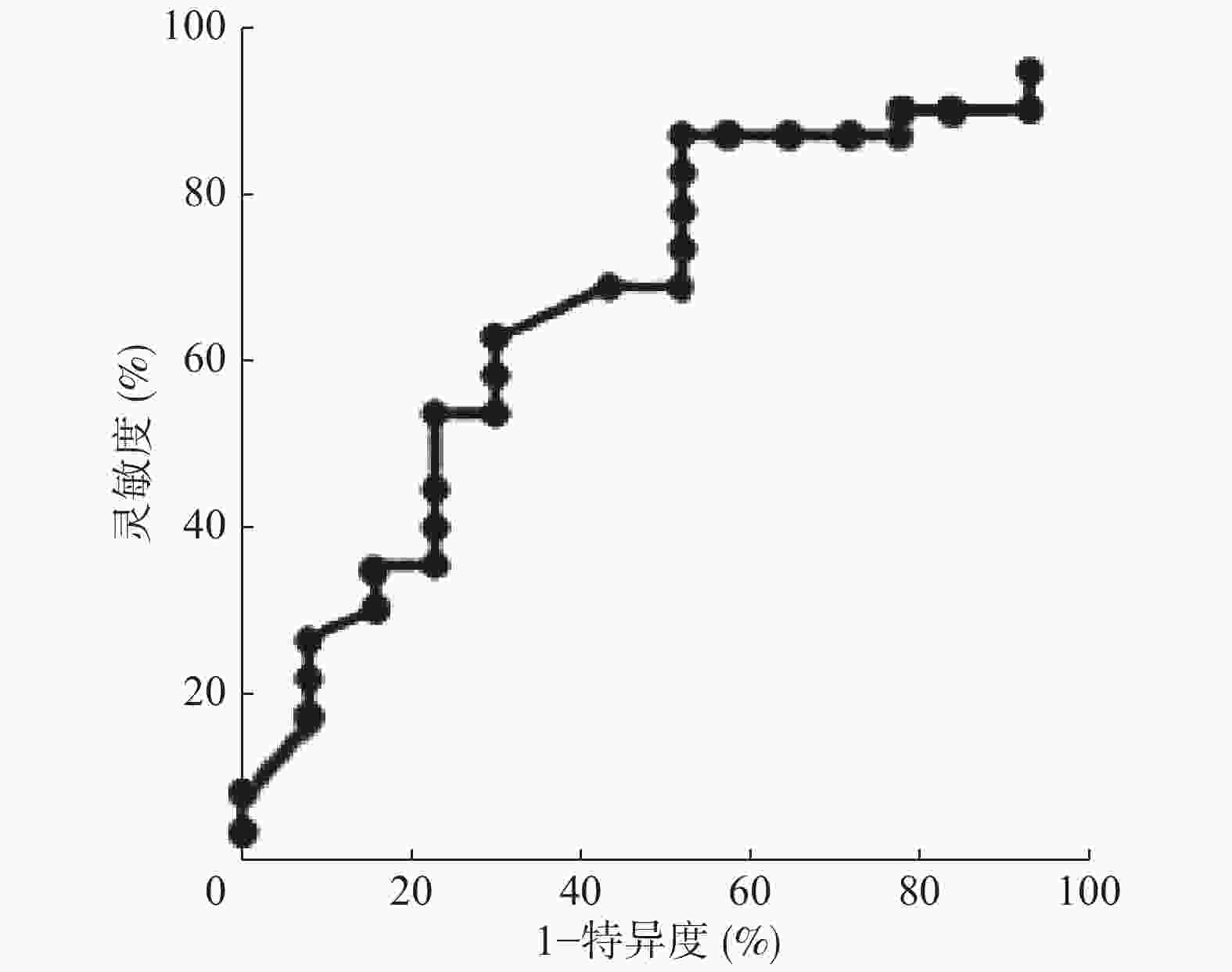
由图2的ROC曲线显示,以S/N比值预测PTCA治疗后LVEF改善的界值为0.62,AUC为0.823(95%CI:0.661~0.985),灵敏度为91.54%,特异度为85.71%。PTCA治疗前S/N比值≥0.62的患者(n=25),治疗后平均LVEF明显高于S/N比值<0.62的患者(n=11),且差异有统计学意义(48±10 vs. 39±13,t=−2.315,P=0.015);而治疗前二者的比较,差异无统计学意义(37±12 vs. 38±11,t=−2.315,P=0.015)。
-
AMI是在冠状动脉病变的基础上出现的缺血事件,最终导致左心室心肌细胞的损伤和(或)死亡。以往研究证实,心肌肌钙蛋白是早期诊断AMI的特异性标志物,灵敏度高、特异性强[10]。心肌肌钙蛋白作为早期诊断AMI的生化指标,对AMI患者的诊断具有一定提示作用。本研究结果显示,PTCA治疗前后cTnI的差异无统计学意义,这说明心肌肌钙蛋白对于AMI提供的临床价值有限[11]。但最新研究结果表明,应用心肌灌注/代谢显像评估左心室存活心肌数量可预测患者的预后,具有重要的临床价值[6]。
患者发生AMI后若不尽早实现闭塞血管再通,将会使存活心肌进一步受到损害,极易引起心功能不全和恶性心律失常[12]。本研究结果显示,AMI患者PTCA治疗前的LVEF、PER差异无统计学意义,而治疗后LVEF、PER较治疗前增加、S/N比值较治疗前降低,且差异有统计学意义。其原因可能在于PTCA早期再灌注治疗能及时疏通闭塞及狭窄动脉,给血管床以“支持”,使AMI患者冬眠心肌和顿抑心肌功能有效恢复,故早期PTCA治疗挽救存活心肌对AMI患者左心室功能的恢复十分必要。但目前国内外对PTCA治疗后左心室心功能改善程度与存活心肌占比的相关性研究较少。Arbel等[13]研究结果发现,PTCA治疗后AMI患者再发心血管意外事件的概率与LVEF的改善密切相关。本研究多因素Logistic回归分析亦发现S/N比值是患者远期LVEF改善的独立影响因素。左心室的S/N比值不同,对左心室重构影响不一。我们还通过ROC曲线对左心室的S/N比值与LVEF改善进行半定量分析,结果发现S/N比值预测AMI患者PTCA治疗后LVEF改善的界值为0.62(灵敏度85.71%、特异度91.54%)。在S/N比值≥0.62的AMI患者中,PTCA治疗后平均LVEF明显高于S/N比值<0.62的患者。因此,在PTCA治疗前评估患者存活心肌占比对预测患者远期预后具有重要价值。应用心肌灌注/代谢显像半定量分析S/N比值可预测AMI患者左心室功能的变化,对于S/N比值≥0.62的患者,应积极给予血运重建阻止存活心肌进一步损伤,可为其他心肌梗死区开辟侧支循环并提供充足血氧,使患者LVEF明显改善,降低患者远期心力衰竭和心血管意外事件的发生率,同时也可为临床治疗提供参考,最终使患者成为最佳获益者。
Slart等[14]以心肌细胞对18F-FDG的摄取百分比≥50%和室壁增厚率≥10%作为判断心肌存活的标准,运用18F-FDG PET心肌代谢显像及心脏MRI对行PTCA治疗的患者进行研究分析,结果发现存活心肌节段数是左心室收缩功能变化的最佳预测指标。其研究结果与本研究得出的S/N比值是AMI患者PCI治疗后LVEF改善的独立影响因素这一结果基本一致,但本研究预测LVEF改善的S/N界值与其结果存在差异,这可能与患者入选标准和评估心肌存活的方法不同有关。本研究使用18F-FDG PET心肌代谢显像联合MPI是目前评估心肌存活的“金标准”[15],其对AMI患者的临床价值更高。
本研究的不足之处为样本量较少,研究的影响因素不全面,在今后研究中需行多中心的大样本研究,进行更细致的节段分组研究和多因素分析,进一步验证本研究的可靠性。有关PTCA治疗后患者的远期预后尚需大样本临床研究进一步证实。
利益冲突 本研究由署名作者按以下贡献声明独立开展,不涉及任何利益冲突。
作者贡献声明 崔雅丽、秦锐锐、胡光负责现场试验的实施、论文的撰写等;刘海燕、晋建华负责方法的建立、论文的审阅。
核素心肌显像对急性心肌梗死患者 PTCA后左心室收缩功能变化的评估价值
Evaluation value of radionuclide myocardial imaging on left ventricular systolic function changes in patients with acute myocardial infarction after PTCA
-
摘要:
目的 探讨核素心肌灌注/代谢显像对急性心肌梗死(AMI)患者经皮腔内冠状动脉成形术(PTCA)后左心室收缩功能变化的评估价值。 方法 回顾性分析2015年6月至2017年12月于山西医科大学附属第一医院行PTCA治疗的AMI患者36例,其中男性27例、女性9例,年龄(57.9±12.3)岁。所有患者均在AMI发病1周内行PTCA治疗,并于治疗前3天和治疗后6~8个月行99Tcm-甲氧基异丁基异腈SPECT心肌灌注显像(MPI)和18F-氟脱氧葡萄糖PET/CT心肌代谢显像。根据治疗后左心室射血分数(LVEF)的变化值(ΔLVEF)将患者分为2组:A组为心功能改善组(ΔLVEF≥5%);B组为心功能未改善组(ΔLVEF<5%)。采用t检验和χ2检验分析比较2组患者的临床资料、LVEF、高峰射血率(PER)、存活心肌/坏死心肌节段数(S/N)比值及心肌肌钙蛋白I(cTnI)等指标间的差异;采用Logistic回归分析影响左心室收缩功能的相关因素;采用受试者特征工作(ROC)曲线分析预测LVEF改善(ΔLVEF≥5%)的S/N界值。 结果 (1)2组患者的性别、年龄、体重指数、吸烟史、高血压病史、糖尿病史、高脂血症病史、心绞痛病史等差异均无统计学意义(均P>0.05)。(2)PTCA治疗前,A组和B组间LVEF、PER、cTnI的差异无统计学意义,而S/N比值(A组:1.24±1.06,B组:0.58±0.37)的差异有统计学意义(t=0.824,P=0.042);PTCA治疗后,2组间LVEF(A组:47±12,B组:38±10)、PER(A组:2.11±0.48,B组:1.71±0.50)、S/N比值(A组:0.73±0.47,B组:0.62±0.39)的差异有统计学意义(t=−2.528、−2.366、−2.514,P=0.016、0.024、0.017),但cTnI间的差异无统计学意义。(3)Logistic回归分析显示,S/N比值是ΔLVEF的独立影响因素(OR=2.164,P=0.018)。(4)ROC曲线结果显示,以S/N比值预测AMI患者PTCA治疗后ΔLVEF≥5%的界值为0.62,曲线下面积为0.823(95%CI:0.661~0.985),特异度为85.71%,灵敏度为91.54%。 结论 核素心肌灌注/代谢显像对AMI患者行PTCA治疗后左心室收缩功能变化的评估具有重要的临床价值。 -
关键词:
- 心肌梗死 /
- 心肌灌注显像 /
- 正电子发射断层显像计算机体层摄影术 /
- 血管成形术,气囊,冠状动脉 /
- 存活心肌 /
- 坏死心肌
Abstract:Objective To evaluate the value of radionuclide myocardial perfusion/metabolic imaging in the assessment of left ventricular systolic function after percutaneous coronary intervention and percutaneous transluminal coronary angioplasty (PTCA) in patients with acute myocardial infarction (AMI). Methods The present study was a retrospective analysis of 36 patients (male/female, 27/9; age, 57.9±12.3 years) with AMI who were treated with PTCA in the First Affiliated Hospital of Shanxi Medical University from June 2015 to December 2017. All patients received PTCA treatment within 1 week of onset of AMI. SPECT myocardial perfusion imaging and PET/CT myocardial metabolic imaging were performed 3 days before and 6–8 months after treatment. Patients were then divided into two groups, namely, group A (left ventricular ejection fraction (LVEF) change value (ΔLVEF), ΔLVEF≥5%) and group B (ΔLVEF<5%). The t- and χ2 tests were used to compare differences between the two groups in terms of clinical data (LVEF, peak ejection rate (PER), ratio of surviving myocardium/necrotic myocardium (S/N) and cardiac troponini (cTnI) Logistic regression was used to analyze the factors affecting left ventricular systolic function. Receiver operating characteristic (ROC) curve analysis determined the S/N ratio cutoff for improved LVEF values (ΔLVEF≥5%). Results No statistically significant differences in gender, age, body mass index, history of smoking, hypertension, diabetes, hyperlipidemia, and angina were found between the two groups (all P>0.05). Before PTCA treatment, differences in LVEF, PER, and troponin cTnI between groups A and B were not statistically significant but differences in S/N ratio (group A: 1.24±1.06, group B: 0.58±0.37) were statistically significant (t=0.824, P=0.042). After PTCA treatment, differences in LVEF (group A: 47±12, group B: 38±10), PER (group A: 2.11±0.48, group B: 1.71±0.50), and S/N ratio (group A: 0.73±0.47, group B: 0.62±0.39) between the two groups were statistically significant (t=−2.528, −2.366, −2.514; P=0.016, 0.024, 0.017) but the difference in cTnI was not statistically significant. Multivariate Logistic regression analysis showed that the S/N ratio is an independent influencing factor of ΔLVEF (OR=2.164, P=0.018). ROC curve results showed that the S/N ratio could be used to predict the cut-off value (0.62) of ΔLVEF≥5% after PTCA treatment in AMI patients. The area under the curve was 0.823 (95%CI: 0.661–0.985), the specificity was 85.71%, and the sensitivity was 91.54%. Conclusion Radionuclide myocardial perfusion/metabolic imaging has important clinical value in evaluating left ventricular systolic function after PTCA treatment in AMI patients. -
表 1 2组急性心肌梗死患者一般临床特征的比较
Table 1. Comparison of general clinical characteristics between the two groups of patients with acute myocardial infarction
组别 男性/女性(例) 年龄(岁) 体重指数(kg/m2) 吸烟史 高血压病史 糖尿病史 高血脂症病史 心绞痛病史 A组(n=24) 18/6 53.6±9.3 24.75±3.60 16(66.7%) 17(70.8%) 13(54.2%) 12(50.0%) 16(66.7%) B组(n=12) 9/3 62.2±9.1 25.01±3.79 7(58.3%) 10(83.3%) 7(58.3%) 8(66.7%) 7(58.3%) 检验值 χ2=0.000 t=−2.828 t=−0.201 χ2=0.241 χ2=0.667 χ2=0.056 χ2=0.900 χ2=0.241 P值 1.000 0.110 0.842 0.429 0.403 0.819 0.103 0.429 注:表中,A组:心功能改善组(ΔLVEF≥5%);B组:心功能未改善组(ΔLVEF<5%)。ΔLVEF:左心室射血分数的变化值 表 2 2组急性心肌梗死患者的左心室心功能参数的比较(
$\scriptstyle \bar x $ Table 2. Comparison of left ventricular cardiac function parameters between the two groups of patients with acute myocardial infarction (
$\scriptstyle \bar x$ 组别 LVEF(%) PER(%) S/N比值 cTnI(μg/L) 治疗前 治疗后 治疗前 治疗后 治疗前 治疗后 治疗前 治疗后 A组(n=24) 36±12 47±12 1.54±0.35 2.11±0.48 1.24±1.06 0.73±0.47 1.52±0.41 1.39±0.39 B组(n=12) 39±10 38±10 1.79±0.44 1.71±0.50 0.58±0.37 0.62±0.39 1.02±0.20 1.18±0.67 t值 0.569 −2.528 −1.075 −2.366 0.824 −2.514 −0.844 −1.229 P值 0.573 0.016 0.292 0.024 0.042 0.017 0.141 0.259 注:表中,A组:心功能改善组(ΔLVEF≥5%);B组:心功能未改善组(ΔLVEF<5%)。LVEF:左心室射血分数;PER:高峰射血率;S/N比值:存活心肌/坏死心肌节段数的比值;cTnI:心肌肌钙蛋白I -
[1] 李树恒, 田月琴, 孙晓昕, 等. 应用心肌灌注/代谢显像探讨心肌损伤与左室机械运动不同步化的相关性[J]. 中华核医学与分子影像杂志, 2017, 37(10): 623−626. DOI: 10.3760/cma.j.issn.2095−2848.2017.10.005.
Li SH, Tian YQ, Sun XX, et al. Evaluation of relationship between myocardial injury and left ventricular mechanical dyssynchrony using 99Tcm-MIBI myocardial perfusion SPECT and 18F-FDG myocardial metabolic PET imaging[J]. Chin J Nucl Med Mol Imaging, 2017, 37(10): 623−626. DOI: 10.3760/cma.j.issn.2095−2848.2017.10.005.[2] Qaderdan K, Vos GJA, McAndrew T, et al. Outcomes in elderly and young patients with ST-segment elevation myocardial infarction undergoing primary percutaneous coronary intervention with bivalirudin versus heparin: Pooled analysis from the EUROMAX and HORIZONS-AMI trials[J]. Am Heart J, 2017, 194: 73−82. DOI: 10.1016/j.ahj.2017.08.009. [3] 中华医学会心血管病学分会心血管病影像学组, 稳定性冠心病无创影像检查路径的专家共识写作组. 稳定性冠心病无创影像检查路径的专家共识[J]. 中国介入心脏病学杂志, 2017, 25(10): 541−549. DOI: 10.3969/j.issn.1004−8812.2017.10.001.
Cardiovascular Imaging Group, Cardiovascular Branch, Chinese Medical Association, Expert Consensus Writing Group on the Path of Noninvasive Imaging of Stable Coronary Heart Disease. Expert Consensus of Stable Coronary Heart Disease Noninvasive Imaging Path[J]. Chin J Interv Cardiol, 2017, 25(10): 541−549. DOI: 10.3969/j.issn.1004−8812.2017.10.001.[4] 徐济民. 缺血性心脏病诊断的命名及标准——国际心脏病学会和协会/世界卫生组织临床命名标准化专题组的联合报告[J]. 国外医学: 心血管疾病分册, 1979, (6): 365−366.
Xu JM. Nomenclature and Standards for the Diagnosis of Ischemic Heart Diseases—The Joint Report of the International Association of Cardiology and the Association / WHO Clinical Nomenclature Standardization Task Force[J]. For Med Sci Cardiovasc Dis, 1979, (6): 365−366.[5] 姜睿, 胡盛寿, 田月琴, 等. SPECT存活心肌评分对冠状动脉旁路移植手术远期心脏不良事件的预测作用[J]. 中华胸心血管外科杂志, 2010, 26(3): 154−157. DOI: 10.3760/cma.j.issn.1001−4497.2010.03.006.
Jiang R, Hu SS, Tian YQ, et al. Prognosis for SPECT test of myocardial viability of the long-term adverse cardiovascular events following the coronary artery bypass graft[J]. Chin J Thorac Cardiovasc Surg, 2010, 26(3): 154−157. DOI: 10.3760/cma.j.issn.1001−4497.2010.03.006.[6] 杨彦松. 存活心肌数量预测冠状动脉旁路移植术后左心室射血分数改善和左心室容积减小的价值[D]. 苏州: 苏州大学, 2016.
Yang YS. Amount of viable myocardium assessed by combining 99mTc-MIBI SPECT and 18F-FDG PET enable to predict left ventricular ejection fraction improvement and left ventricular volume reduction after coronary artery bypass surgery[D]. Suzhou: Soochow University, 2016.[7] Regenfus M, Schlundt C, von Erffa J, et al. Head-to-head comparison of contrast-enhanced cardiovascular magnetic resonance and 201Thallium single photon emission computed tomography for prediction of reversible left ventricular dysfunction in chronic ischaemic heart disease[J]. Int J Cardiovasc Imaging, 2012, 28(6): 1427−1434. DOI: 10.1007/s10554−011−9941−z. [8] Glaveckaite S, Valeviciene N, Palionis D, et al. Prediction of long-term segmental and global functional recovery of hibernating myocardium after revascularisation based on low dose dobutamine and late gadolinium enhancement cardiovascular magnetic resonance[J]. J Cardiovasc Magn Reson, 2014, 16: 83. DOI: 10.1186/s12968−014−0083−z. [9] Peovska I, Maksimovic J, Vavlukis M, et al. Functional outcome and quality of life after coronary artery bypass surgery in patients with severe heart failure and hibernated myocardium[J]. Nucl Med Commun, 2008, 29(3): 215−221. DOI: 10.1097/mnm.0b013e3282f3add9. [10] 浦春, 陶青松, 朱翔, 等. 三种心肌损伤标志物在急性心肌梗死早期诊断中的应用价值[J]. 中国临床药理学与治疗学, 2017, 22(1): 82−86.
Pu C, Tao QS, Zhu X, et al. Value of three cardiac markers in the early diagnosis of acute myocardial infarction[J]. Chin J Clin Pharmacol Ther, 2017, 22(1): 82−86.[11] Lewis EF, Li YH, Pfeffer MA, et al. Impact of Cardiovascular Events on Change in Quality of Life and Utilities in Patients After Myocardial Infarction: A VALIANT Study (Valsartan In Acute Myocardial Infarction)[J]. JACC Heart Fail, 2014, 2(2): 159−165. DOI: 10.1016/j.jchf.2013.12.003. [12] 莫东洪. PCI与药物溶栓治疗AMI后患者心电图运动平板试验对心功能的观察[J]. 中国现代药物应用, 2016, 10(14): 137−139. DOI: 10.14164/j.cnki.cn11−5581/r.2016.14.097.
Mo DH. Observation of cardiac function in patients with AMI after electrocardiogram exercise test after PCI and drug thrombolysis for AMI[J]. Chin J Med Drug Appl, 2016, 10(14): 137−139. DOI: 10.14164/j.cnki.cn11−5581/r.2016.14.097.[13] Arbel Y, Ko DT, Yan AT, et al. Long-term Follow-up of the Trial of Routine Angioplasty and Stenting After Fibrinolysis to Enhance Reperfusion in Acute Myocardial Infarction (TRANSFER-AMI)[J]. Can J Cardiol, 2018, 34(6): 736−743. DOI: 10.1016/j.cjca.2018.02.005. [14] Slart RHJA, Bax JJ, van Veldhuisen DJ, et al. Prediction of functional recovery after revascularization in patients with chronic ischaemic left ventricular dysfunction: head-to-head comparison between 99mTc-sestamibi/18F-FDG DISA SPECT and 13N-ammonia/18F-FDG PET[J]. Eur J Nucl Med Mol Imaging, 2006, 33(6): 716−723. DOI: 10.1007/s00259−005−0016−z. [15] Murthy VL, Bateman TM, Beanlands RS, et al. Clinical Quantification of Myocardial Blood Flow Using PET: Joint Position Paper of the SNMMI Cardiovascular Council and the ASNC[J]. J Nucl Med, 2018, 59(2): 273−293. DOI: 10.2967/jnumed.117.201368. -





 下载:
下载: